🧪 Adv. Chem: Info on Solution, Solutes, and Solvents.
The solvent is what dissolves the solute. For example, in salt water the solvent is water because it is dissolving the salt (which is the solute).
Soluble means something can be dissolved.
Insoluble means something cannot be dissolved.
Examples include: Sugar is soluble in water, while oil is insoluble in water.
When a solution is saturated, it cannot dissolve more solute (it’s full).
When a solution is unsaturated, it can dissolve/hold more solute.
When a solution is supersaturated, it has more solute than it can theoretically hold (over full). In such circumstance, the solute will eventually fall out of the solution, settling at the bottom.
If a solution is too concentrated, you can dilute it by adding more of the solvent.
A solution with more solute, is more concentrated. For example, a 46% solution > 35% solution.
An alloy is a solution of two metals melted together. One example of an alloy is steel.
Types of non solution mixtures
(Non solution): A suspension is a temporary mixture in which particles eventually settle. It can be easily filtered
(Non solution): A colloid is a mixture with larger particles, as seen in milk, mayonnaise, and egg whites. The particles in this result come in clusters, not like single molecules seen in actual solutions. *Some Colloids may exhibit the Tyndall effect, which causes the scattering of light as it passes through. One instance of this can be seen in the scattering of light in fog.
Review Summary of Test📝
A solution is a mixture that is homogeneous at the molecular level, thus a mixture is a homogeneous solution.
Opposite of soluble is insoluble, meaning cannot be dissolved [further].
Solubility is the how many grams of solute dissolves in 100mL of water.
For most solid solutes, as temperature goes up, solubility goes up. ←Direct Relationship
For most gas solutes, as temperature goes up, solubility goes down. ←Inverse Relationship.
The five factors that affect solubility are temperature, pressure (especially in gases), inter-molecular forces, agitation, and surface area.
You must keep a carbonated beverage cool to prevent it from going flat, because gas solubility decreases as temperature rises. As the beverage gets warmer, the CO2 molecules are able to escape the drink more easily.
Non-electrolytes don’t ionize in water or conduct electricity, whereas weak electrolytes can partially iodize in water, conducting energy weakly. On the other hand, strong electrolytes fully iodize in water and conduct electricity effectively.
In order to conduct electricity, a solution must contain freely-moving ions, helping to move it along and complete the electrical circuit.
Water is known as the universal solvent because it has the ability to dissolve more substances than other solvents, due to its polar nature, which enables it to pull ions and other molecules into the solution.
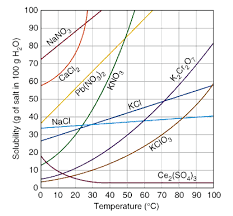
Review Graph
What is the solublity of KNO3 at 70 degrees C?
Molarity tells the concentration of a solution, identifying how many moles of solute per liter of the solution.
Molarity Practice Problems
Calculate the molarity of a solution containing 10 g of KNO3 dissolved in enough water to make 500 mL of solution.
If you have a 2.0 M NaCl solution, how many grams of NaCl are present in 250 mL of this solution?
What is the molarity of a solution prepared by dissolving 5.0 moles of glucose in 2.0 L of water?
Molar Dilutions (m1v1=m2v2)
You have a 3.0 M solution of hydrochloric acid (HCl) and need 250 mL of a 1.0 M solution. How much of the 3.0 M solution will you need to dilute?
- Given: M1 = 3.0 M, V1 = ?, M2 = 1.0 M, V2 = 250 mL
- Using the formula M1V1 = M2V2, we have:
(3.0M)(V1)=(1.0)(250mL)
Solving for V1 gives V1 = 83.3 mL.
If you want to prepare 1.5 L of a 0.5 M sodium chloride (NaCl) solution from a stock solution of 2.5 M NaCl, how much of the stock solution do you need to dilute?
- Given: M1 = 2.5 M, V1 = ?, M2 = 0.5 M, V2 = 1500 mL
- Using the formula M1V1 = M2V2, we have:
Solving for V1 gives V1 = 300 mL.A laboratory needs to prepare 500 mL of a 0.2 M potassium nitrate (KNO3) solution from a concentrated solution of 1.0 M. How much of the concentrated solution is required?
- Given: M1 = 1.0 M, V1 = ?, M2 = 0.2 M, V2 = 500 mL
- Using the formula M1V1 = M2V2, we have:
Solving for V1 gives V1 = 100 mL.