fuels
VCE 3/4 CHEMISTRY 2023
K.B Tutoring Notes
Lesson 1: Fuels
Learning Objectives
How fossil fuels are obtained
Use of fossil fuels
Production of biofuels
Distinction between fossil fuels and renewable fuels
Distinction between biodiesel and petrodiesel
Environmental implications of fuels
Units of Energy:
The SI unit for energy is known as a joule (J)
By extension:
1 kilojoule (kJ) = 103 Joules
1 megajoule (MJ) = 106 Joules
1 gigajoule (GJ) = 109 Joules
1 terajoule (TJ) = 1012 Joules
Conservation of Energy:
During a chemical reaction, atoms are neither created nor destroyed. They are merely rearranged to form new substances.
During a chemical reaction, mass is neither gained nor lost. In other words, the mass of the reactants is equal to the mass of the products. This is the Law of Conservation of Mass.
Likewise, during such a reaction, energy is neither created nor destroyed, it is merely converted to another form of energy. The total amount of energy remains unchanged. This is the Law of Conservation of Energy.
Fossil Fuels
Fossil Fuels are fuels that have been formed from the decomposition of organic matter over a long period of time in anoxic (without oxygen), pressurised and hot conditions.
These fuels are currently the prevalent sources of energy in society, with key examples being coal, oil and natural gas.
Societies are now looking to move away from these sources of fuel because:
Concern over the particulates being released into the atmosphere due to their combustion and usage, i.e. CO2 (contributes to greenhouse effect and global warming), SO2 (can cause acid rain)
The reserves of fossil fuels are finite and will eventually run out
Key Question: Why are fossil fuels considered to be non-renewable despite being formed through natural processes?
Answer:
They are not an energy resource that can be replaced by natural processes in a relatively short period of time (they are consumed at a rate that is far greater than the rate they are produced).
Coal
As plant material such as wood starts to decay, some gradual chemical changes occur
This leads to the carbon content of this material increasing, and the hydrogen and oxygen content decreasing with water evaporating during this process.
This process of decay involves this matter turning into peat , then brown coal and finally black coal.
As this material decays further and further into black coal, the energy content (energy released per mass of fuel) increases, making black coal a better fuel than the other two.
This is due to two factors:
The carbon content per gram increases, the substance which releases the energy when combusted
The water content per gram is reduced, meaning that energy is not consumed to vaporise this water and more energy is available for the desired usage.
The carbon dioxide released per unit of energy is less than that of peat and brown coal.
Worst Fuel Best Fuel
Peat Brown Coal Black Coal
Significant quantities of ash and other pollutants are released in the combustion of coal, making natural gas and oil cleaner fuels by comparison.
Oil
Refined oils such as petrol are initially obtained from crude oil
Crude oil is a mixture of hydrocarbons, predominantly alkanes
This crude oil is then put through fractional distillation to obtain fractions of hydrocarbons with similar molar masses (for example, crude oil can be separated into diesel which contains larger hydrocarbons and petrol, which has shorter hydrocarbons)
Each fraction would have similar boiling points due to the similar molar mass and similar strength of dispersion forces
Natural Gas
Natural gas consists of predominantly methane (CH4), with traces of other hydrocarbon gases
Methane is available in two forms: as a biofuel and as a fossil fuel
Methane is considered to be a non-renewable fossil fuel when it is obtained from gas reservoirs trapped between rocks, in coal seam gas and shale gas.
As these are the ways methane is obtained naturally, natural gas is considered non-renewable.
Extracting these forms of natural gas involves fracking, where the rock or coal is fractured to release the gas.
Energy Transformations
Combusting fuels releases the stored energy, which can be converted to other forms of energy such as kinetic or electric energy to use in various situations
Energy efficiency is the proportion of stored chemical energy that is converted to the desired form of energy. Usually, the more transformation steps, the lesser the efficiency.
Energy Transformations in Coal-fired power station
Coal is burnt in the furnace: chemical energy to thermal energy
Heat from the coal is used to boil water: thermal energy (coal) to thermal energy (steam). Usually don’t have to mention this, good to know.
Steam then turns a turbine: thermal energy to mechanical energy
Electricity produced from a generator connected to the turbine: mechanical energy to electrical energy
Energy Transformations in Gas-fired power station
Gas is burnt in the furnace: chemical energy to thermal energy
The hot gases then turns a turbine: thermal energy to mechanical energy
Electricity produced from a generator connected to the turbine: mechanical energy to electrical energy
This power station is more efficient that the coal-powered station as there are less conversion steps and hence less energy wasted
This greater efficiency means that gas powered stations emit significantly less carbon dioxide than coal powered station
Energy Efficiency: Useful Energy/Total Energy x 100 (units percentage)
The Need for Renewable Fuels:
Currently, around the world the prevalent fuels being used are finite fossil fuels.
The necessity for fuels that have less of an environmental impact are leading to a push for widespread adoption of renewable and other less harmful fuels.
However, in order to meet this criteria, replacement energy sources need to be reliable, sustainable and cost effective.
This has led to research into and small-scale implementation of biofuels such as bioethanol, biodiesel and biogas, and other energy sources such as fuel cells (to be explored later).
Biofuels
Biofuels represent an alternative to fossil fuels in many common daily usages, such as household usages and transport usages
Biofuels are derived from plant matter. Commonly, these are plant oils, carbohydrate heavy plants such as sugar cane, and plant waste.
Biofuels get the bio suffix as opposed to their fossil fuel equivalents because they are produced directly from biological sources
Plant oils are a key ingredient in the production of biodiesel, the carbohydrate heavy plants are commonly used to produce bioethanol, and the plant waste is used to produce biogas.
These fuels, while renewable, also have less of a carbon footprint, as the carbon content in the biofuel initially came from carbon dioxide in the air and was converted via photosynthesis.
This leads to a theoretical zero net release, and hence can sometimes be said to be carbon neutral. However, this is not always the case, with fuel being expended to help grow the plants, convert the plant matter and transport it, among other things.
They are also not necessarily sustainable on a large scale, as the sheer quantity of plant matter necessary to keep up with society would be very large.
A conflict also arises with the uses of land to grow these biofuel crops as this comes into direct completion with land reserved for food crops, which is already stressed with a growing human population. This means that on a large scale, the land can be used for food or biofuel, but not both.
Bioethanol
Produced by the anaerobic fermentation of glucose by yeast
Yeast contains the biological catalysts (enzymes) needed to catalyse this fermentation
Commonly used as an alternative or supplementary fuel to petrol
Equation: C6H12O6 (aq) -> 2CH3CH2OH (aq) + 2CO2 (g)
Less energy available per litre opposed to petrol, and causes greater engine wear, but is renewable
This lower energy content is due to the ethanol molecule being partly oxidised, that is, partially containing oxygen
Less particulates are released due to the presence of ethanol
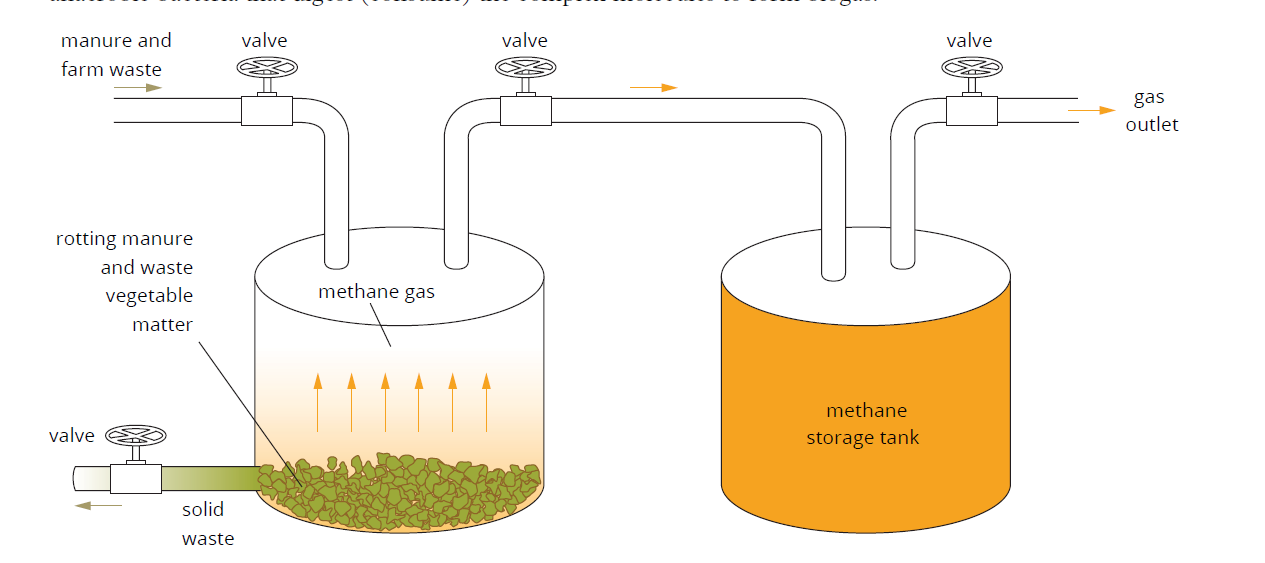
Biogas
A gaseous mixture containing a large proportion of methane gas and carbon dioxide
Primarily produced by anaerobic digestion of organic waste matter by microorganisms (i.e specific bacteria)
Not fully efficient, about 60% efficient
Inputs for the decomposition are widely available, such as from rubbish tips or on farms
The methane produced is predominantly used for domestic use (cooking, heating, etc.)
Biodiesel
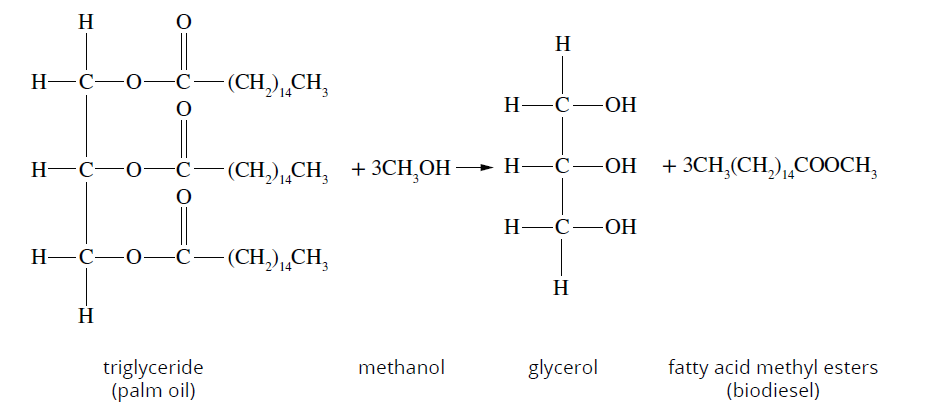
Biodiesel comprises of a mixture of esters
These esters are produced by reacting a vegetable fat/oil molecule with an alcohol molecule
A fat molecule includes a glycerol molecule bonded to 3 different fatty acids (carboxylic acids) via 3 ester links
An alcohol is added to the presence of this triglyceride and potassium hydroxide catalyst and is lightly heated.
This results in the formation of 3 ester molecules and a glycerol molecule, the ester molecules of which are separated out for use as a biofuel.
Used as a transport fuel for larger vehicles
Petrodiesel vs Biodiesel
Name | Petrodiesel | Biodiesel |
|---|---|---|
How is it obtained | Fossil fuel, produced from crude oil. (Hydrocarbon) | Renewable ester, produced from reaction between triglyceride and alcohol |
Chemical properties | Non-polar, with only dispersion forces between molecules. Hence lower viscosity and melting point than biodiesel. | Have a polar ester link and are usually larger molecules than Biodiesel.Hence dispersion forces and dipole-dipole forces exist.These stronger intermolecular forces result in higher melting point and viscosity than petrodiesel. |
Ability to flow in fuel lines | Flow better at lower temperatures due to lower viscosity due to weaker intermolecular forces. | Does not flow well in cold climates and low temperatures due to higher viscosity due to stronger intermolecular forces. |
Hygroscopic properties | Does not attract water | Absorbs water in storage due to polar ester ends of molecules.Reduces efficiency of combustion as this water must be evaporated. |
Energy Content | 40MJ L-1 | 36MJ L-1 |
Environmental Impacts | Energy used to extract and produce the fuelGreenhouse gases emitted to produce and combust | Production process does use energy, and thus release greenhouse gases, but biodiesel is more carbon neutral as the carbon dioxide released in combustion originates through photosynthesis. |
Definitions
Kinetic Energy: Energy associated with movement
Potential Energy: Energy stored in chemical bonds
Fuel: A substance with stored energy that can be easily released to be converted to another form of energy. e.g. Coal, biodiesel, etc.
Non-Renewable: When a substance is used up at a rate faster than it can be replaced
Renewable: When a substance is continually replenished at a rate faster than its consumption
Shale Gas: Methane trapped in shale rock
Coal Seam Gas: Methane bonded to the surface of the coal
Alkanes: A hydrocarbon with general formula (CnH2n+2)
Transesterification: A process in which the triglyceride reacts with an alcohol to form esters
Triglyceride: An ester molecule comprised of a glycerol molecule bonded to three fatty acids
Viscosity: A measurement of a liquid’s resistance to flow
Cloudpoint: The temperature below which diesel or biodiesel appear cloudy owing to the formation of small crystals. The presence of these crystals thickens the fuel and clogs filters and injectors.