Chapter 4: Atomic Structure
4.1-Developing the Model of the Atom
- In 1804, John Dalton agreed with Democritus that matter was made up of tiny spheres that couldn’t be broken up, but he reckoned that each element was made up of a different type of atom
- However, in 1909, scientists in Rutherford’s lab tried firing a beam of alpha particles at thin gold foil-this was the alpha scattering experiment.
- From the plum pudding model, they expected the particles to pass straight through the gold sheet, or only be slightly deflected.
- But although most of the particles did go straight through the sheet, some were deflected more than expected, and a few were deflected back the way they had come-something the plum pudding model couldn’t explain
- Nearly 100 years later, J.J.Thompson discovered particles called electrons that could be removed from atoms. * So Dalton’s theory wasn’t quite right.
- Thomson suggested atoms were spheres of positive charge with tiny negative electrons stuck in them like fruit in a plum pudding
- the plum pudding model
- Because a few alpha particles were deflected back, the scientists realised that most of the mass of the atom must be concentrated at the centre in a tiny nucleus. * This nucleus must also have a positive charge, since it repelled the positive alpha particles
- They also realised that because nearly all the alpha particles passed straight through, most of an atom is just empty space.
- This was the first nuclear model of the atom.
\
- The nuclear model that resulted from the alpha particle scattering experiment was a positively charged nucleus surrounded by a cloud of negative electrons
- Niels Bohr said that electrons orbiting the nucleus do so at certain distances called energy levels.
- His theoretical calculations agreed with experimental data
- Evidence from further experiments changed the model to have a nucleus made up of a group of particles which all had the same positive charge that added up to the overall charge of the nucleus
- About 20 years after the idea of a nucleus was accepted, in 1932, James Chadwick proved the existence of the neutron, which explained the imbalance between the atomic and mass numbers
\
- The model is constantly being changed, but currently * It contains protons and neutrons, which gives it an overall positive charge * The rest of mostly empty space, negative electrons more around the outside of the nucleus really fast * Radius of atom is 1x10(-10) * Number of protons = number of electrons * If they gain energy by absorbing EM radiation they move to a higher energy level
4.2-Isotopes and Nuclear Radiation
- All atoms of each element have a set number of protons.
- The number of protons in an atoms is its atomic number * The mass number is the number of protons+neutrons
- Isotopes are atoms with same number of protons different number of neutrons * All elements have different isotopes, but there are usually only one or two stable ones
- The other unstable isotopes tend to decay into other elements and give out radiation as they try to become more stable. * This process is called radioactive decay
- Radioactive substances spit out one or more types of ionising radiation from their nucleus-the ones you need to know are alpha, beta and gamma radiation
- They can also release neutrons when they decay, as they rebalance their atomic and mass numbers * Ionising radiation is radiation that knocks electrons off atoms, creating positive ions, * The ionising power of a radiation source is how easily it can do this
\
- Alpha radiation is when an alpha particle is emitted from the nucleus.
- A a-particle is two neutrons and two protons
- They don’t penetrate very far into materials and are stopped quickly-they can only travel a few cm in air and are absorbed by a sheet of paper * Because of their size they are strongly ionising
\
- A beta particles, is simply a fast-moving electron released by the nucleus. Beta particles have virtually no mass and a charge of -1 * They are moderately ionising. * They penetrate moderately far into materials before colliding and have a range in air of a few meters. * They are absorbed by a sheet of aluminium
- For every beta particle emitted, a neutron in the nucleus has turned into a proton
\
- Gamma rays are waves of electromagnetic radiation released by the nucleus
- They penetrate far into materials without being stopped and will travel a long distance through air * This means they are weakly ionising because they tend to pass through rather than collide with atoms. * Eventually they hit something and do damage
- They can be absorbed by thick sheets of lead or metres of concrete
\
4.3-Nuclear Equations
- Nuclear equations are a way of showing radioactive decay by using element symbols
- They’re written in the form:atom before decay - atom after decay+radiation emitted
- There is one golden rule to remember:
- the total mass and atomic number must be equal on both sides
\
- Remember, alpha particles are made up of two protons and two neutrons.
- So when an atom emits an alpha particles, its atomic number reduces by 2 and its mass number reduces by 4
- A proton is positively charges and a neutron is neutral, so the charge of the nucleus decreases
- In nuclear equations, an alpha particles can be written as a helium nucleus
\
- When beta decay occurs, a neutron in the nucleus turns into a proton and releases a fast-moving electron
- The number of protons in the nucleus has increased by 1.
- This increases the positive charge of the nucleus
- Because the nucleus has lost a neutron and gained a proton during beta decay, the mass of the nucleus doesn’t charge * A beta particle is written as 0/-1e in nuclear equations
\
- Gammas rays are a way of getting rid of excess energy from a nucleus
- This means that there is no change to the atomic mass or atomic number of the atom
4.4-Half-life
- Radioactive substances give out radiation from the nuclei of their atoms-no matter what * This radiation can be measured with a Geiger-Muller tube and counter, which records the count-rate-the number of radiation counts reaching it per second
- Radioactive decay is entirely random. So you can’t predict exactly which nucleus in a sample will decay next, or when any one of them will decay * But you can find out the time it takes for the amount of radiation emitted by a source to halve, this is known as the half-life. * It can be used to make predictions about radioactive sources, even though their decays are random
- Half-life can be used to find the rate at which a source decays-its ACTIVITY. Activity is measured in becquerels, Bq
\
- Each time a radioactive nucleus decays to become a stable nucleus, the activity as a whole will decrease
- For some isotopes it takes just a few hours before nearly all the unstable nuclei have decayed, whilst others last for millions of years * The problem with trying to measure this is that the activity never reaches zero, which is why we have to use the idea of half-life to measure how quickly the activity drops off
- The half-life is the time taken for the number of radioactive nuclei in an isotope to halve
- It is also the time taken for the activity, and so count-rate, to halve.
- A short half-life means the activity falls quickly, because the nuclei are very unstable and rapidly decay.
- Sources with a short half-life are dangerous because of the high amount of radiation they emit at the start, but they quickly become safe * A long half-life means the activity falls more slowly because most of the nuclei don’t decay for a long time-the source just sits there, releasing small amounts of radiation for a long time. * This can be dangerous because nearby areas are exposed to radiation for millions of years
\
- If you plot a graph of activity against time, it will always be shaped like the one to the right * The half-life is found from the graph by finding the time interval on the bottom axis corresponding to a halving of the activity on the vertical axis
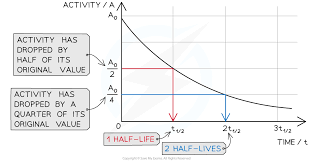
4.5-Background Radiation and Contamination
Background radiation is the low-level radiation that’s around us all the time. You should always measure and subtract the background radiation from your results. It comes from:
- Radioactivity of naturally occuring unstable isotopes which are all around us-in the air, in food, in building materials and in the rocks under our feet
- Radiation from space, which is known as cosmic rays.
- These come mostly from the sun.
- Luckily, the earth’s atmosphere protects us from much of this radiation * Radiation due to human activity or nuclear waste exists. * But this represents a tiny proportion of the total background radiation * The radiation doss tells you the risk of harm to body tissues due to exposure to radiation. It’s measured in sieverts. * The dose from background radiation is small, so millisieverts are often used. * Your radiation dose varies depending on where you live or if you have a job that involves radiation
\
- Objects near a radioactive source are irradiated by it.
- This simply means they’re exposed to it
- Irradiating something does not make it radioactive
- Keeping sources in lead-in boxes, standing behind barriers or being in a different room and using remote-controlled arms are all ways of reducing the effects of irradiation
\
- If unwanted radioactive atoms get onto or into an object, the object is said to be contaminated.
- These contaminating atoms might then decay, releasing radiation which could cause you harm
- Contamination is especially dangerous because radioactive particles could get inside your body
- Gloves and tongs should be used when handling sources, to avoid particles getting stuck to your skin or under your nails.
- Some industrial workers wear protective suits to stop them breathing in particles
\
Contamination or irradiation can cause different amounts of harm
- Outside the body, beta and gamma sources are the most dangerous.
- This is because beta and gamma can penetrate the body and get to the delicate organs.
- Alpha is less dangerous because it can’t penetrate the skin and is easily blocked by a small air gap.
- High levels of irradiation from all sources are dangerous, but especially from ones that emit beta and gamma
- Inside the body, alpha sources are the most dangerous, because they do all their damage in a very localised area.
- So contamination, rather than irradiation, is the major concern when working with alpha sources.
- Beta sources are less damaging inside the body, as radiation is absorbed over a wider area, and some passes out of the body although.
- Gamma sources are the least dangerous inside the body, as they mostly pass straight out-they have the lowest ionising power
- The more we understand about how radiation affects our bodies, the better we can protect ourselves when using it.
4.6-Uses and Risk
- Radiation can enter living cells and ionise atoms and molecules within them, this can lead to tissue damage * Lower doses tend to cause minor damage without killing the cells. * This can give rise to mutant cells which divide uncontrollably. * This is cancer. * Higher doses tend to kill cells completely, causing radiation sickness if a lot of cells all get blatted at once
\
- Certain radioactive isotopes can be injected into people and their progress around the body can be followed using an external detector.
- A computer converts the reading to a display showing where the strongest reading is coming from * One example is the use of iodine-123, which is absorbed by the thyroid gland just like normal iodine-127, but it gives out radiation which can be detected to indicate whether the thyroid gland is taking in iodine as it should
- Isotopes which are taken into the body like this are usually GAMMA, so that the radiation passes out of the body without causing much ionisation.
- They should have a short half-life so the radioactivity inside the patient quickly disappears
\
- Since high doses of ionising radiation will kill all living cells, it can be used to treat cancers * Gamma rays are directed carefully and at just the right dosage to kill the cancer cells without damaging too many normal cells. * Radiation-emitting implants can also be put next to or inside tumours
- However, a fair bit of damage is inevitably done to normal cells, which makes the patient feel very ill.
- But if the cancer is successfully killed off in the end, then it’s worth it
\
- Risks: prolonged exposure to radiation poses future risks and causes side effects * Benefits: can get rid of cancer entirely * Perceived risk can vary from person to person
4.7-Fission and Fusion
- Nuclear fission is a type of nuclear reaction used to release energy from large and unstable atoms by splitting them into smaller atoms * Spontaneous fission rarely happens. * Usually, the nucleus has to absorb a neutron before it will split * When the atom splits it forms two new lighter elements that are roughly the same size * Two or three neutrons are also released when an atom splits. * If any of these neutrons are moving slow enough to be absorbed by another nucleus, they cause more fission to occur, which is a chain reaction
- The energy not transferred to the kinetic energy is carried away by gamma rays * Energy carried away can be used to heat water making steam to turn turbines and generators
- The amount of energy produced by fission in a nuclear reactor is controlled by changing how quickly the chain reaction can occur, which is done by control rods
- Uncontrolled chain reactions quickly lead to lots of energy being released as an explosion-this is how nuclear weapons work
\
- Nuclear fusion is the opposite of nuclear fission * Two light nuclei collide at high speed and join to create larger, heavier nucleus
- Heavier nucleus produced does not have as much mass as the two separate light nuclei did.
- Some of the mass is converted to energy, which is released as radiation
- Fusion released a lot of energy
- Scientists haven’t found a way of using fusion to generate energy, and would be to hard and expensive to do at the moment
\