Atoms, Molecules, Ions
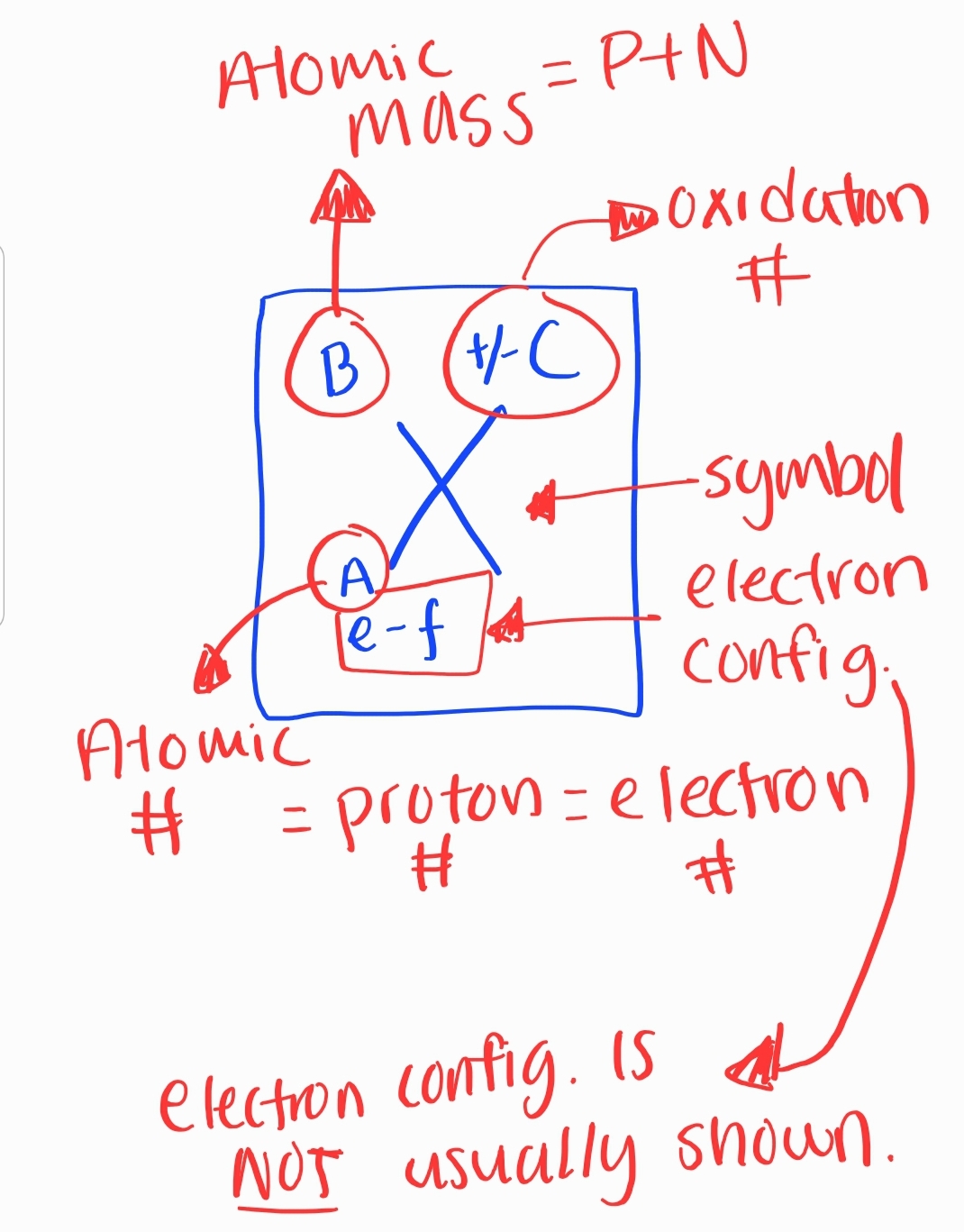
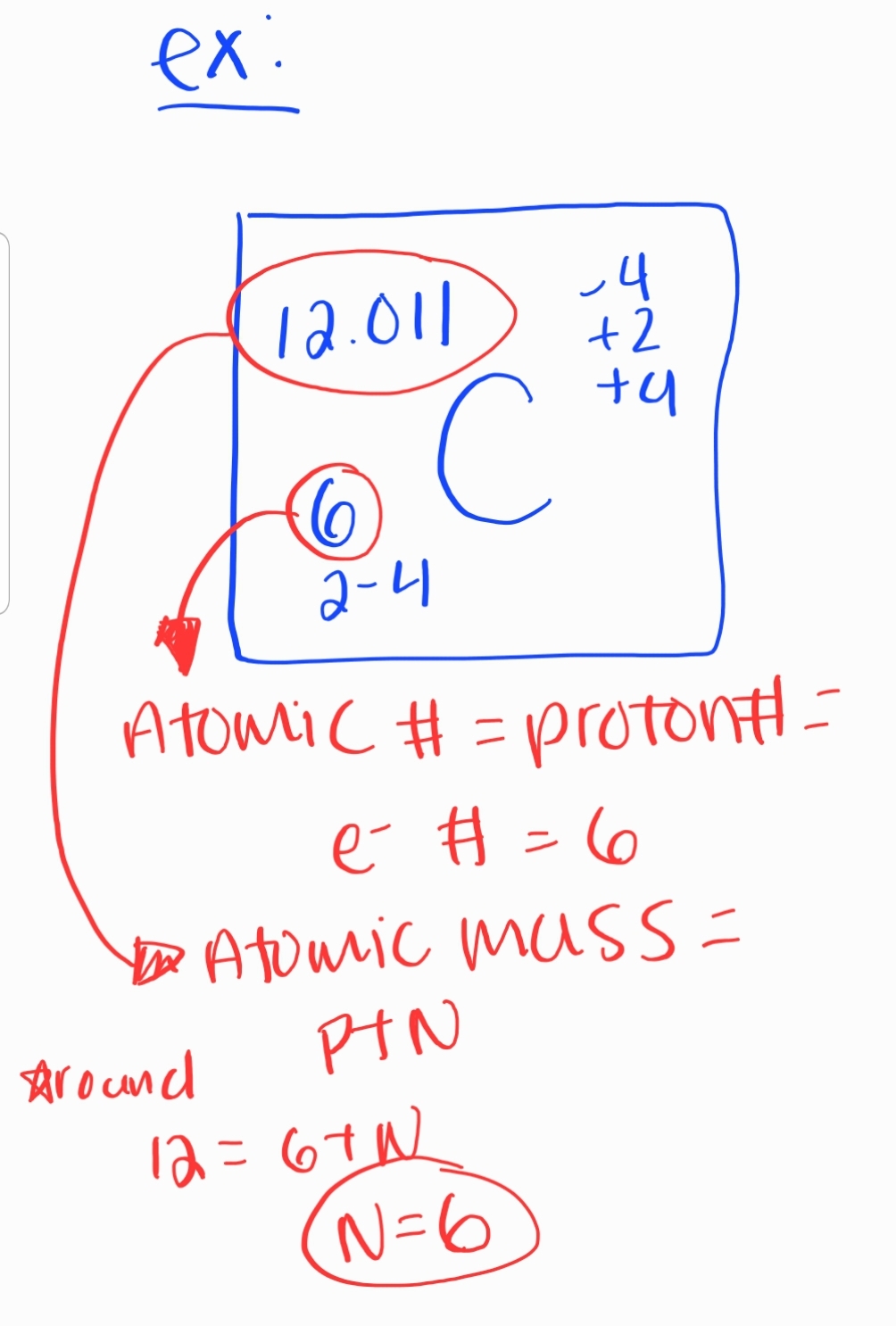
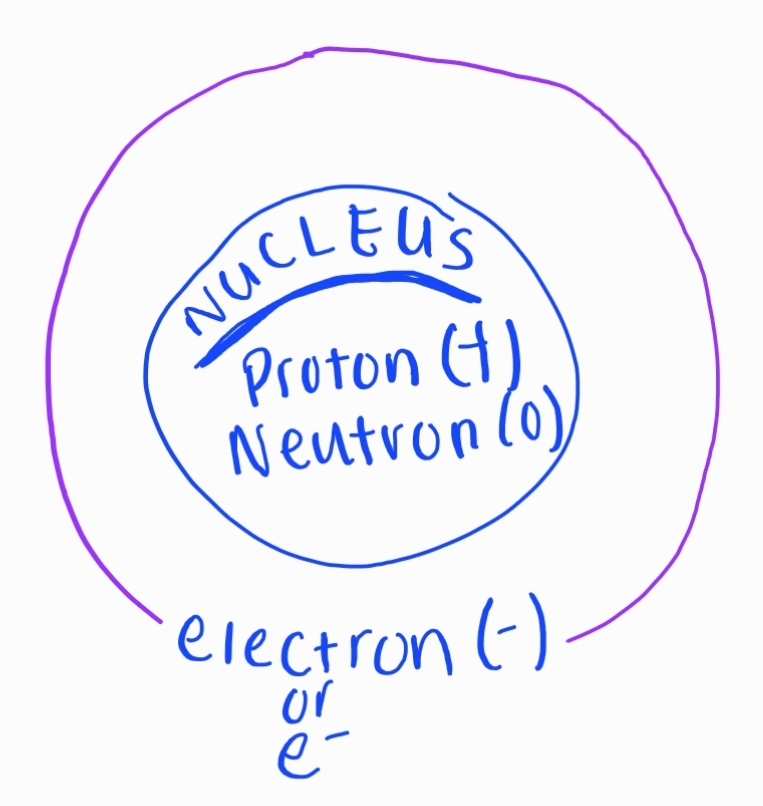
- All atoms of an element must have the same ATOMIC #
# Mind Map: Atoms, Molecules, Ions
## Central Idea: Atoms * Definition: Basic building blocks of matter * Composed of: Protons, neutrons, and electrons
## Main Branches
### 1. Atomic Structure * Protons * Positive charge * Located in the nucleus * Neutrons * No charge * Located in the nucleus * Electrons * Negative charge * Orbit around the nucleus
### 2. Elements * Definition: Pure substances made up of only one type of atom * Examples: Hydrogen, Oxygen, Carbon
### 3. Atomic Number and Mass * Atomic Number * Number of protons in an atom * Determines the element * Atomic Mass * Sum of protons and neutrons in an atom * Measured in atomic mass units (amu)
## Sub-Branches
### 1. Molecules * Definition: Two or more atoms chemically bonded together * Types of Bonds * Covalent Bonds * Sharing of electrons between atoms * Examples: H2, O2, CO2 * Ionic Bonds * Transfer of electrons between atoms * Examples: NaCl, MgO
### 2. Ions * Definition: Charged particles formed by * Cations * Positively charged ions (more protons than electrons) * Formed by losing electrons * Anions * Negatively charged ions (more protons than electrons) * Formed by gaining electrons * electrically charged particle w 2+ atoms linked together so it behaves as a unit instead of separate atoms * charge belongs to the ion as a WHOLE
### 3. Compound * : 2+ diff elements chemically bonded together * Examples: Water (H2O), Sodium Chloride (NaCl)
### 4. Chemical Reactions * : Process where atoms are rearranged to form new substances * Reactants * Substances present before the reaction * Products * Substances formed after the reaction
### 5. Isotopes * : Atoms of the same element with different numbers of neutrons * Ex: Carbon-12, Carbon-13, Carbon-14
### 6. Atomic Models * Dalton's Model * Thomson's Model * Rutherford's Model * Bohr's Model * Quantum Mechanical Model
### 7. Periodic Table * Organizes elements in an increasing atomic #
\