Chapter 3 - Chemsity
Matter
anything occupying space and having mass
matter exists in three states
soild
defined volume and shape
liquid
assumes the shape of container
has a defined volume
water, milk,
gas
no defined volume or shape
takes the shape and volume of its container
air, helium, oxygen
States of water
solid - ice
liquid - water
gas - steam
Physical and chemical properties and changes
Physical: characteristics that are directly observable and unique to a substance
ex. odor, color, volume, state, boiling, and melting point
Chemical: substrate's ability to form a new substance
Characteristics that determine how the composition of matter changes as a result of contact with other matter or the influence of energy
Ex. flammability, rusting of steel, digestion of food
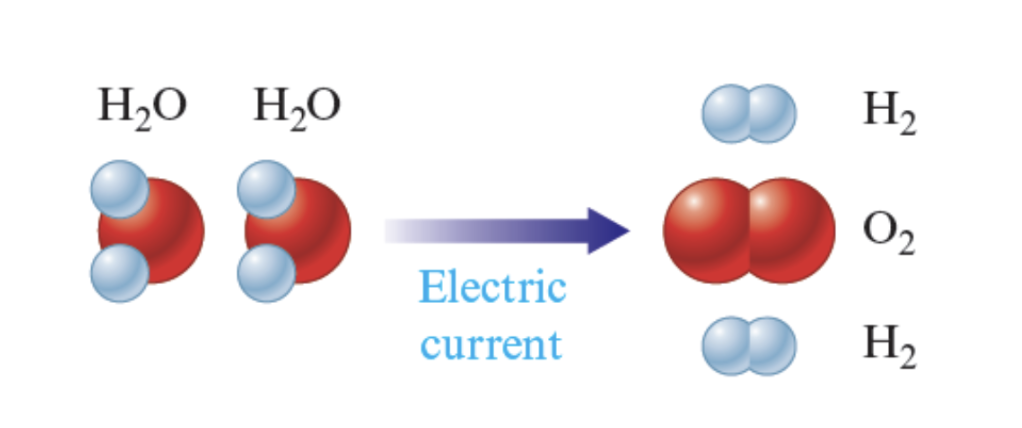
Electrolysis of water
Elements and compounds
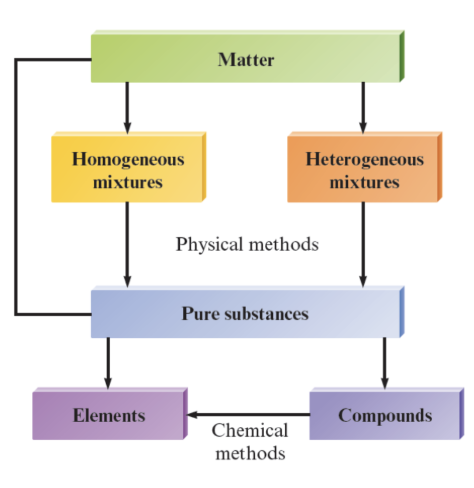
Element: a substance that cannot be broken down into other substances by chemical methods
Ex: Iron (Fe) Alumminun (AI)
All matter in the world around us contains elements
Compound: a substance composed of a given combination of elements that can be broken down into those elements by chemical methods
Ex: Water (H2O), carbon dioxide (CO2)
Always contains atoms of different elements
always has the same composition or the same combination of atmos
Mixed and Pure Substances
Pure substances
always have the same composition
are either elements or compounds
Ex: pure water, hydrogen, gold
Mixtures
have variable composition
Ex: wood, wine, and coffee
two or more pure substances
Homogeneous mixture
substance that is the same throughout
sometimes called a solution
consists of visibly indistinguishable parts
doesn’t vary in composition from one region to another
Ex: air around you, brass, table salt in water
Heterogeneous Mixture
visibly distinguishable parts
contain regions that have different properties from other regions
Ex: oil and vinegar dressing, sand stirred into water
Separation of mixtures
Separation based on physical properties
Mixtures can be separated based on different physical properties of the components
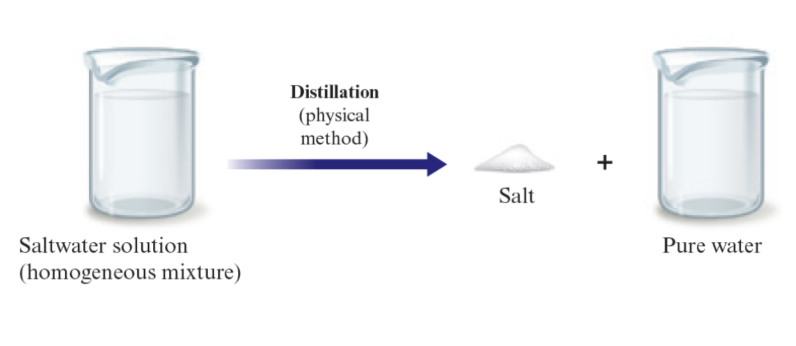
Boiling point → distillation
Salt water: no chemical change occurs when salt water is distilled
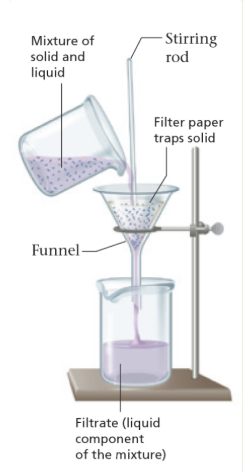
State of matter (solid/liquid/gas) → filtration
separates a liquid from a solid
Adherence to a surface → Chromatography
Volatility → Evaporation
Organization of matter