Elements, Atoms, and Ions
Elements
- 118 known: 88 found in nature, rest in lab
- You need to know the names and symbols before you can do chemistry
- Could mean a single atom of an element (Ar or H)
- Could mean many molecules of the same element (H2)
- Or atoms of an element in some form (sodium in the human body)
Symbols for Elements
- Each has 1 or 2 letters
- 1st letter is capitalized, 2nd is not
- Usually consists of 1 or 2 letters from names
* Oxygen = O - Sometimes taken from Greek or Latin name
* Gold = Au (aurum)
Dalton’s Atomic Theory
- Most natural materials are mixtures of pure substances
- Pure substances are either elements or combinations of elements called compounds
- A given compound always contains the same mass of the elements
Law of Constant Composition
A given compound always has the same composition, regardless where it came from.
Dalton’s Atomic Theory (1808)
- Elements are made up of tiny things called atoms
- All atoms of the same element are identical (proven to be false)
- Atoms are unique from other atoms of a different element
- Atoms of one element can combine with atoms from another to create a compound
- Atoms are indivisible in chemical processes. They are not created or destroyed in a chemical reaction, it simply changes the way they are grouped (proven to be wrong)
Formula of Compounds
- Compound - distinct substance of 2 or more elements, has the same masses of those elements
- Chemical Formulas - expresses the types of atoms and the number of each
Rules for Formulas
- Each atom is represented by its element symbol
- The number of each is indicated by a subscript written to the right of the symbol
- No subscript is written if there is just one atom
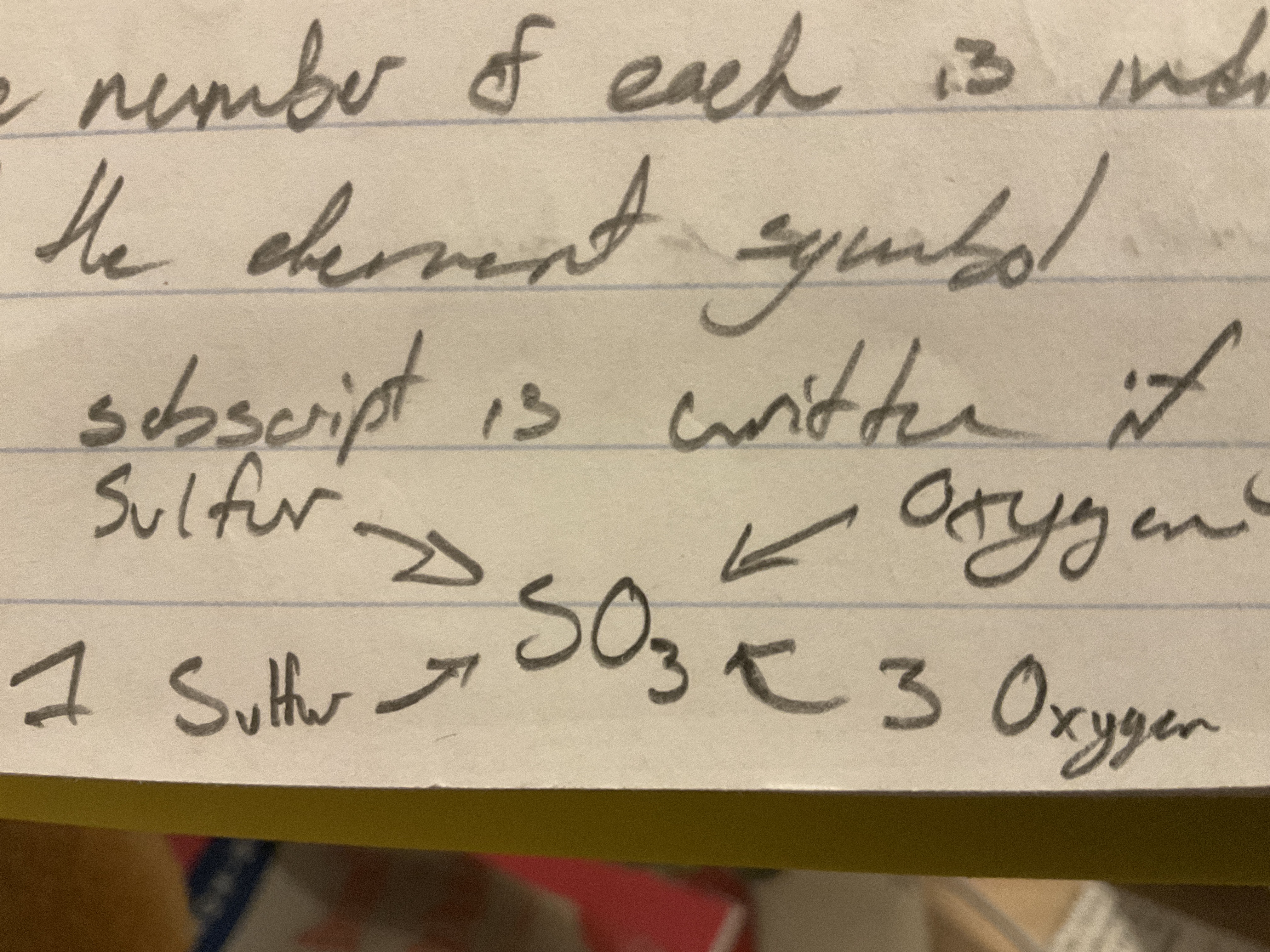 J.J. Thomson (1898-1903)
J.J. Thomson (1898-1903)
Postulated the existence of electrons using cathode-ray tubes
The atoms must also contain positive particles that balance exactly the negative change carried by particles we now call electrons
William Thomson (Plum Pudding Model)
Reasoned that the atom might be thought of as a uniform pudding of positive charge with enough negative electrons scattered within to counterbalance the positive charge
Ernest Rutherford (1911)
Explained the nuclear atom
Atom has a dense center of positive charge called the nucleus
Electrons travel around the nucleus at a relatively large distance
A proton has the same magnitude of charge as the electron, but its charge is positive
Rutherford and Chadwick (1932)
Mostly nuclei; contains neutron
A neutron is slightly more massive than a proton, but has no charge
Modern Atom Structure
Electrons: tiny, negatively charged equal to protons, outside nucleus
Protons: positively charged, same size as neutron, inside nucleus
Neutron: no charge, found in nucleus
Different Chemical Properties in Atoms
Caused by electrons
Electrons intermingle when atoms combine into molecules
Number of electrons determines chemical behavior
Isotopes
Atoms with same number of protons, ==different number of neutrons==
Shows almost identical chemical properties
Most elements contain isotopes in nature
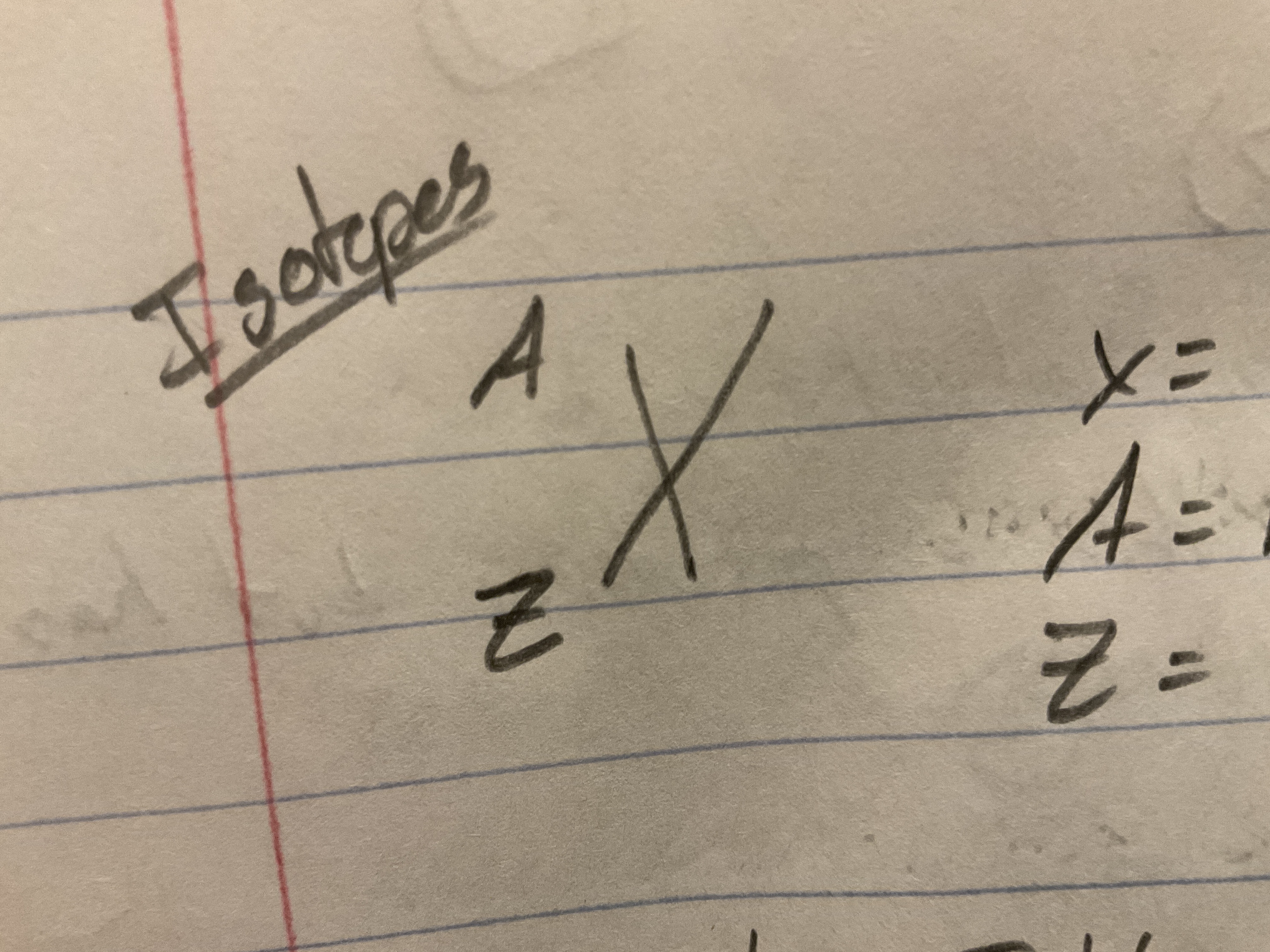
X = symbol of element
A = mass number (number of protons and neutrons)
Z = atomic number (number of protons)
Periodic Table
In order of increasing atomic number
Elements with similar properties are in same column, called groups or families
Periods - horizontal rows of elements
Physical Properties of Metal
Conductivity of heat and electricity
Malleability
Ductility
Lustrous
Physical Properties of Nonmetals
Lack of metal properties
Variations in properties
Can be solid, gas, or liquid
Physical Properties of Metalloids
Exhibits properties of metals and nonmetals, i.e. silicon
Natural States of the Elements
Most are very reactive
Usually not found in unconfined form
Exceptions are: noble metals - gold, platinum, silver, and noble gases - Group 8
Allotropes
Different forms of the same element
Ex: Solid carbon occurs in 3 forms - diamond, graphite, and buckminsterfullerene
Ions
Atoms can form ions by gaining or losing electrons
Metals tend to lose one or more electrons to form positive ions called cations
Cations are generally named by using the parent name followed by ion
Nonmetals want to receive electrons to form negative ions called anions
Anions are named using the root of the atom name followed by the suffix -ide, i.e. Oxygen → Oxide ion
Periodic Table Locations
The ion that a particular atom will form can be predicted from the periodic table
| Group Name | Ion Charge |
|---|---|
| Alkali Metals (1A) | +1 |
| Alkaline Earth Metals (2A) | +2 |
| Halogens (7A) | -1 |
| Noble Gases (8A) | 0 |
Compounds that Contain Ions
Ions combine to form ionic compounds
Properties: high melting points, conduct electricity (if melted or dissolved in water)
Electrically neutral
The charges the anion and cation in the compound must equal to zero
Formula for Compounds
Write the cation element symbol followed by the anion
The number of cations and anions must be correct for their charges to equal to zero
Mg(2+) + Cl(1-) + Cl(1-) = 0 charge
Cation Anions