Chapter 14: Carboxylic Acids, Esters, Amines, and Amides
14.1: Carboxylic Acids
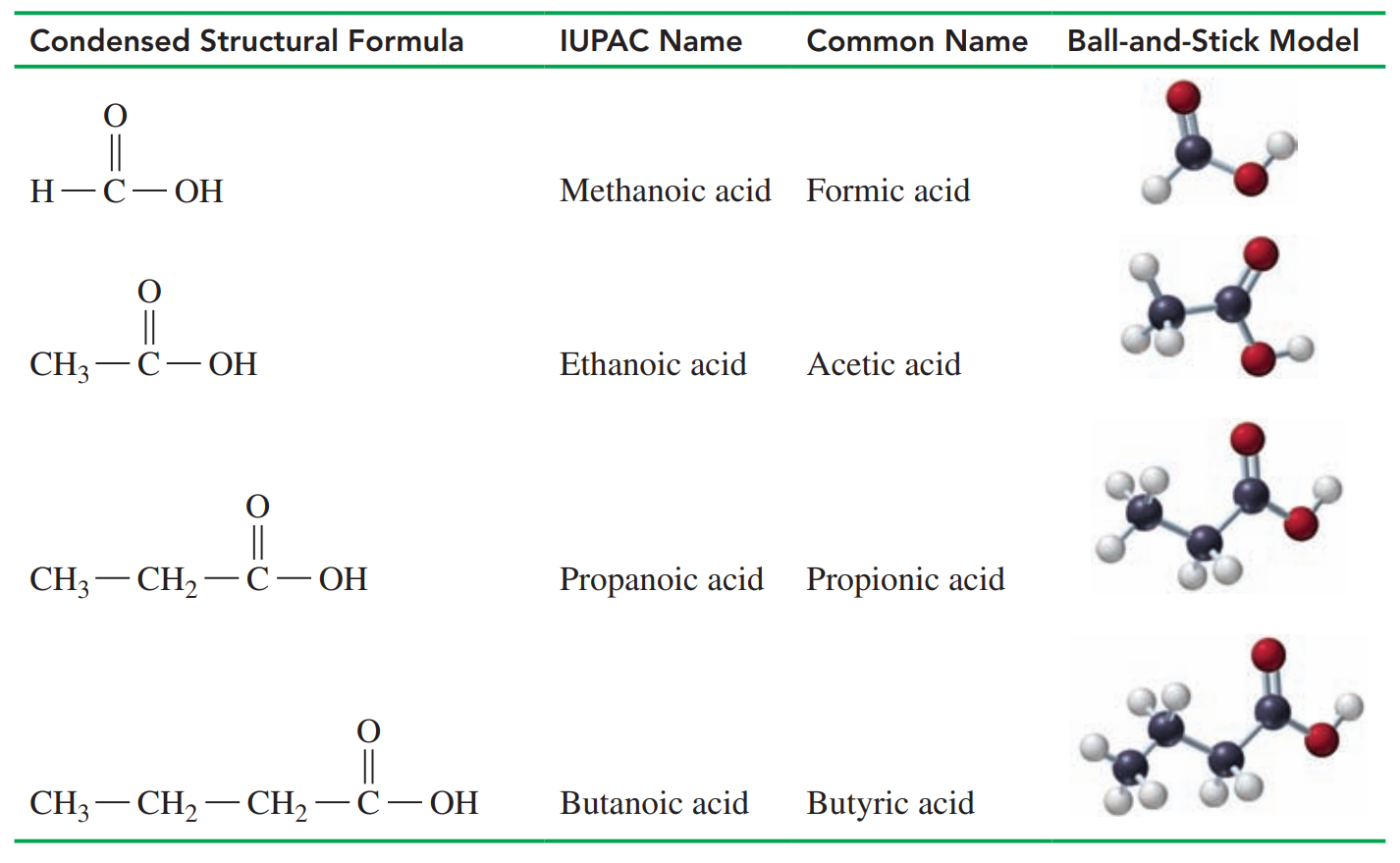
- Carboxylic Acids
- These are weak acids. They have a sour or tart taste, produce hydronium ions in water, and neutralize bases.
- The carbon atom of a carbonyl group is attached to a hydroxyl group that forms a carboxyl group.
Common Names of Carboxylic Acids
14.2: Properties of Carboxylic Acids
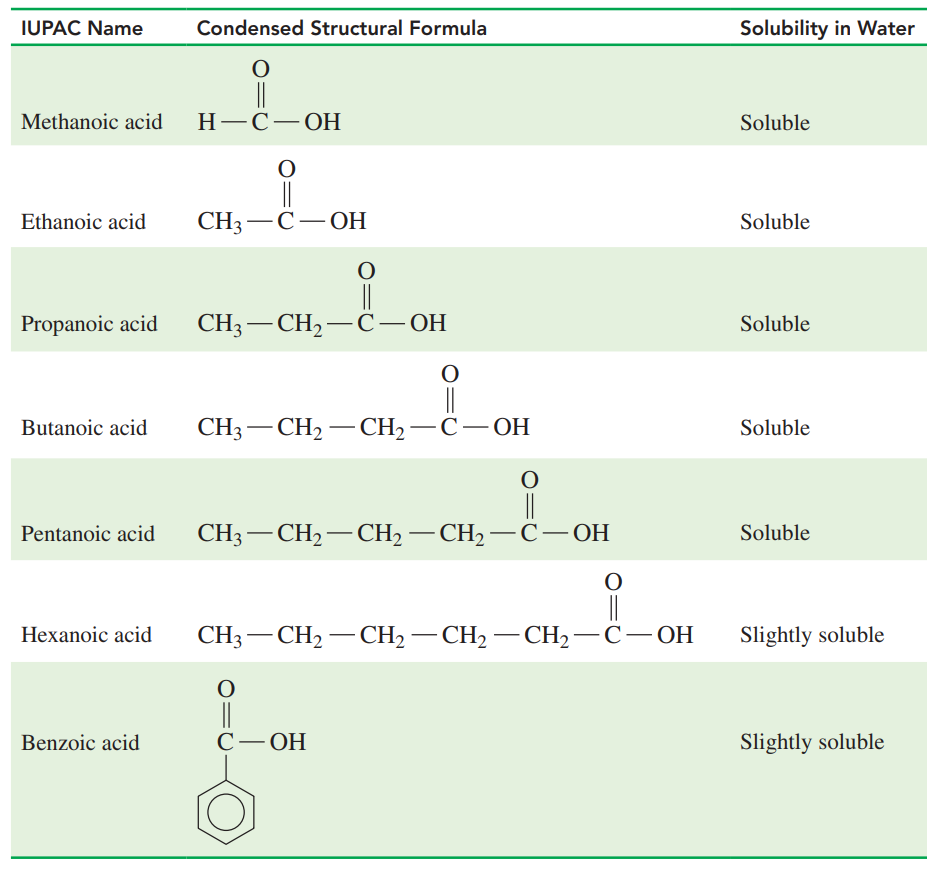
- Carboxylic acids with one to five carbons are soluble in water because the carboxyl group forms hydrogen bonds with several water molecules.
- When a carboxylic acid ionizes in water, a hydrogen ion is transferred to a water molecule to form a negatively charged carboxylate ion and a hydronium ion.
- Because carboxylic acids are weak acids, they are completely neutralized by strong bases — the products are carboxylate salt and water.
Solubility of Selected Carboxylic Acids
14.3: Esters
- A carboxylic acid reacts with an alcohol to form an ester and water.
- In an ester, the —H of the carboxylic acid is replaced by an alkyl group
- Esterification
- An ester is produced when a carboxylic acid and an alcohol react in the presence of an acid catalyst ad heat.
- The —OH group from the carboxylic acid and the —H from the alcohol are removed and combine to form water.
- An excess of the alcohol is used to shift the equilibrium in the direction of the formation of the ester product.
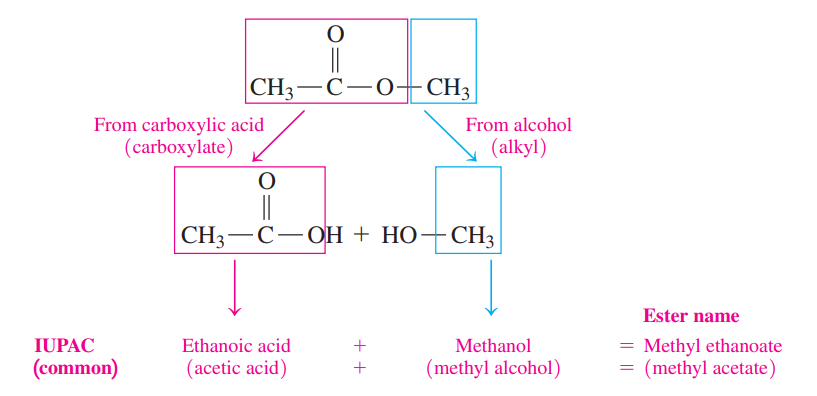
- The first word indicates the alkyl part from the alcohol. The second word is the name of the carboxylate from the carboxylic acid.
- The IUPAC names of esters use the IUPAC names of the acids, while the common names of esters use the common names of the acids.
14.4: Hydrolysis of Esters
- Acid Hydrolysis
- It occurs when esters are heated with water in the presence of a strong acid.
- Water reacts with the ester to form a carboxylic acid and an alcohol.
- It is the reverse of the esterification reaction.
- Base Hydrolysis
- It is also known as saponification, which refers to the reaction of an ester of a long-chain fatty acid with NaOH to make soap.
- When an ester undergoes hydrolysis with a strong base, the products are the carboxylate salt and the corresponding alcohol.
14.5: Amines
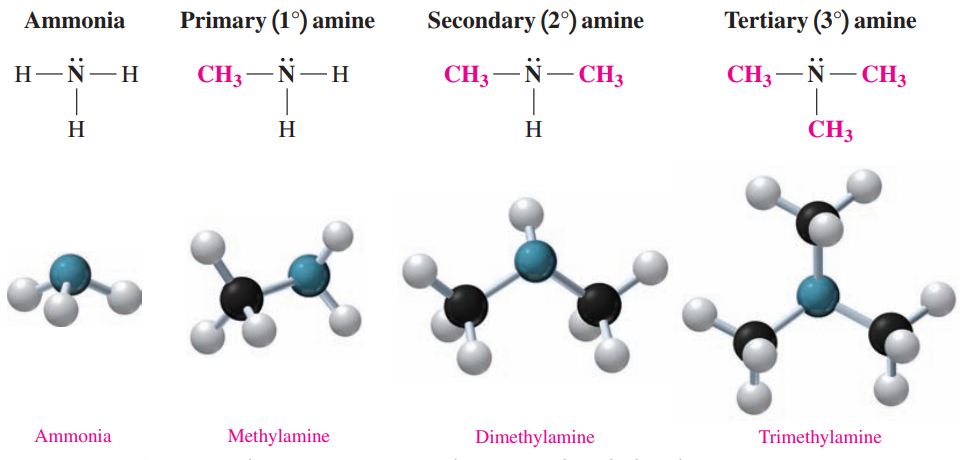
- Amines: These are derivatives of ammonia in which one or more hydrogen atoms are replaced with alkyl or aromatic groups.
- In methylamine, a methyl group replaces one hydrogen atom in ammonia.
- The bonding of two methyl groups gives dimethylamine.
- The bonding of three methyl groups in trimethylamine replaces all the hydrogen atoms in ammonia.
- Amines are classified by counting the number of carbon atoms directly bonded to the nitrogen atom.
- In a primary (1°) amine, the nitrogen atom is bonded to one alkyl group.
- In a secondary (2°) amine, the nitrogen atom is bonded to two alkyl groups.
- In a tertiary (3°) amine, the nitrogen atom is bonded to three alkyl groups.
- The aromatic amines use the name aniline, which is approved by IUPAC.
- Alkyl groups attached to the nitrogen of aniline are named with the prefix N- followed by the alkyl name.
- Examples: Aniline; 4-Bromoaniline; N-Methylaniline
- Because amines contain a polar N—H bond, they form hydrogen bonds with water.
- In a primary (1°) amine, —NH2 can form more hydrogen bonds than the secondary (2°) amine.
- In a tertiary (3°) amine, which has no hydrogen on the nitrogen atom, can form only hydrogen bonds with water from the N atom in the amine to the H of a water molecule.
- Ammonia acts as a Brønsted–Lowry base because it accepts H+ from water to produce an ammonium ion and a hydroxide ion.
- In a neutralization reaction, an amine acts as a base and reacts with an acid to form an ammonium salt.
- Ammonium salts (NH4Cl)
- These are ionic compounds with strong attractions between the positively charged ammonium ion and an anion, usually chloride.
- These salts are solids at room temperature, odorless and soluble in water and body fluids.
- Amines used as drugs are usually converted to ammonium salts.
- Heterocyclic amine
- A cyclic compound that contains one or more nitrogen atoms in the ring.
- The rings typically consist of five or six atoms and one or more nitrogen atoms.
- Pyrrolidine: A ring of four carbon atoms and a nitrogen atom, all with single bonds.
- Piperidine: A six-atom heterocyclic ring with a nitrogen atom.
14.6: Amides
- Amides (CO-NH): These are derivatives of carboxylic acids in which a nitrogen group replaces the hydroxyl group.
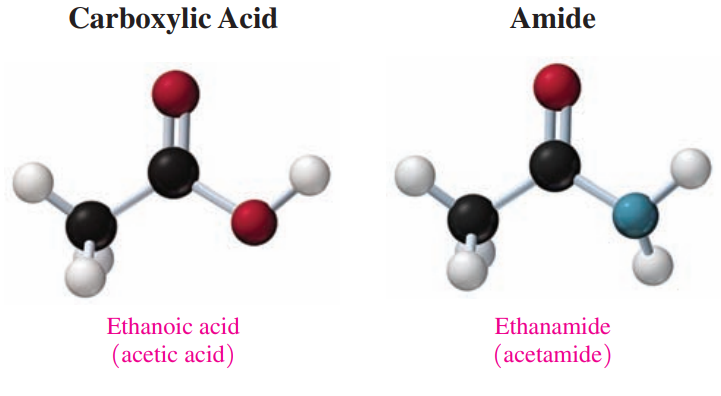
- Amidation: A reaction where an amide is produced, in which a carboxylic acid reacts with ammonia or a primary or secondary amine.
- In both the IUPAC and common names, amides are named by dropping the –oic acid or —ic acid from the carboxylic acid name and adding the suffix amide.
- Example: N, N-Dimethylbutanamide (IUPAC); N, N-Dimethylbutyramide (Common Name)
- The amides with one to five carbon atoms are soluble in water because they can hydrogen bond with water molecules.
- In amides with more than five carbon atoms, the effect of hydrogen bonding is diminished as the longer carbon chain decreases the solubility of an amide in water.
- Amides undergo hydrolysis when water is added to the amide bond to split the molecule.
- When an acid is used, the hydrolysis products of an amide are the carboxylic acid and the ammonium salt.
- In base hydrolysis, the amide produces the carboxylate salt and ammonia or an amine.