Chapter 10: Serology Concepts
10.1: Serological Reagents
Immunogens and Antigens
- Immunogen: A foreign substance that is capable of eliciting antibody formation when introduced into a host.
- Natural immunogens are usually macromolecules such as proteins and polysaccharides.
- Epitope: Also known as determinant site; the molecular structure of an immunogen, usually a small portion recognized by an antibody.
- Multivalent: Multiple epitope in an immunogen.
- Antigen: A foreign substance that is capable of reacting with an antibody. * Hapten: An example of a substance that is antigenic but not immunogenic. They are chemical compounds that are too small to elicit antibody production when they are introduced to a host animal.
Antibodies
- Antibodies: Also known as immunoglobulins, are capable of binding specifically to antigens and are designated with an Ig prefix.
- Isotypes: The immunoglobulins that differ based on the molecular variations in the constant domains of the heavy and light chains.
- The five major classes of immunoglobulins are designated: IgG, IgA, IgM, IgD, and IgE. * IgD, IgE, and IgG are usually monomers. * IgM can be a membrane-bound monomer or a cross-linked pentamer. * IgA can be a monomer, dimer, or trimer.
- Further exposure to the immunogen can elicit a secondary response, producing IgG, IgA, IgE, and IgD immunoglobulins. * IgG is the most abundant immunoglobulin in serum.
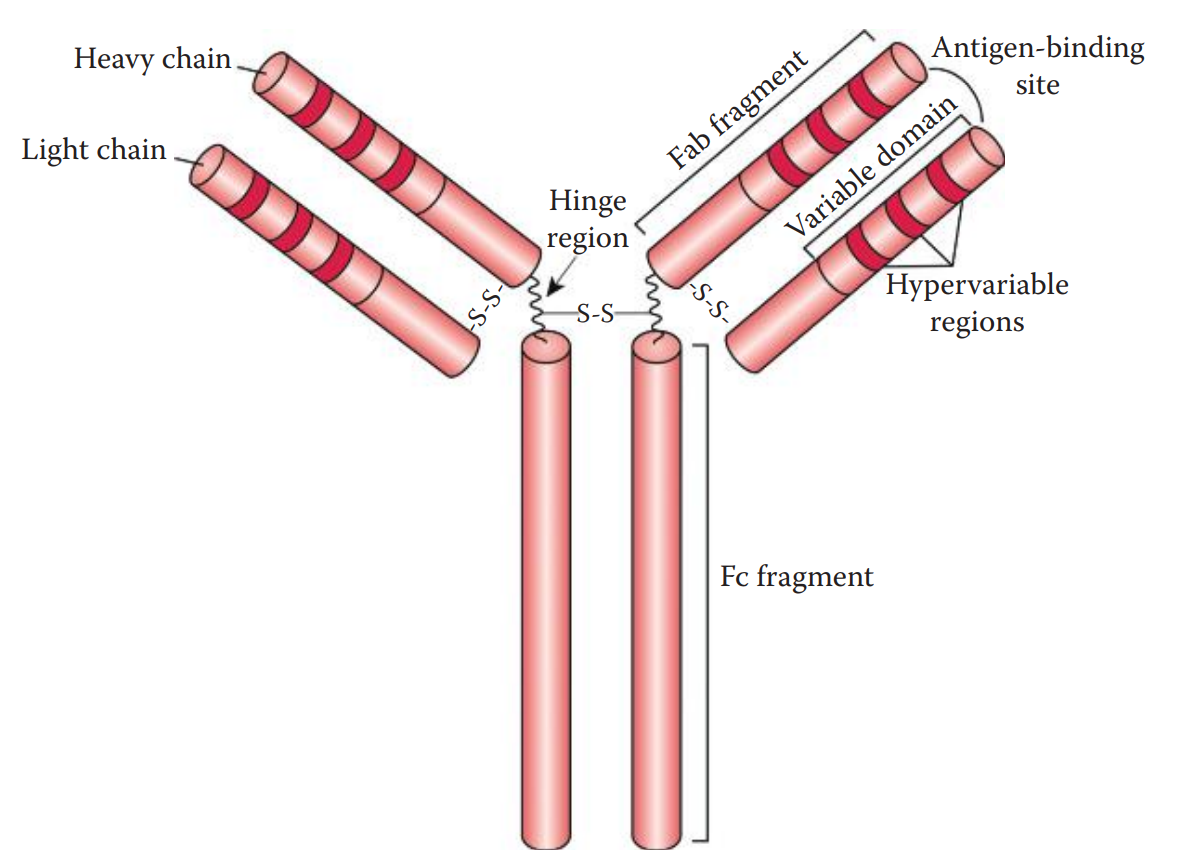
- Immunoglobulins are composed of four polypeptide chains: two heavy (H) chains and two light (L) chains. * The H chain can be divided into fragment antigen-binding (Fab) and fragment crystallizable (Fc) fragments. * The L chain consists of a Fab fragment only.
- A typical antibody has two identical antigen-binding sites and is thus considered bivalent.
- At the amino acid sequence level, both H and L chains have variable and constant domains.
- The variable domains are located at the N-terminal ends of the immunoglobulins.
- Additionally, three small hypervariable regions are located within the variable domain of each chain.
\
\
Polyclonal Antibodies
- To produce an antibody, an immunogen is usually introduced into a host animal.
- A multivalent immunogen is capable of eliciting a mixture of antibodies with diverse specificities for the immunogen.
- As a result, a polyclonal antibody is produced by different B lymphocyte clones in response to the different epitopes of the immunogen.
- Humoral Antibodies: Circulating immunoglobulins.
- The blood from an immune host is drawn and allowed to clot, resulting in the formation of a solid consisting largely of blood cells and a liquid portion known as serum containing antibodies.
- Polyclonal Antiserum: Preparation of humoral antibodies.
\
Monoclonal Antibodies
- To produce a monoclonal antibody, spleen cells are harvested from a host animal, such as a mouse, inoculated with an immunogen.
- Next, the plasma cells of the spleen, which produce antibodies, are fused with myeloma cells.
- The fused cells, called hybridoma cells, are immortal in cell cultures.
- Pools of hybridoma cells are diluted into single clones and are allowed to proliferate. The clones are then screened for the specific antibody of interest.
- Monoclonal antibodies have been utilized in many serological assays. However, it react with only a single epitope of a multivalent antigen and, therefore, cannot form cross-linked networks in precipitation assays.
\
Antiglobulins
- If a purified foreign immunoglobulin or a fragment of a foreign immunoglobulin is introduced into a host, the antibodies produced are known as antiglobulins.
- Antiglobulins that are specific to a particular isotype can be produced in laboratories.
- It is possible to produce nonspecific antiglobulins, which recognize an epitope that is common to all isotypes of an immunoglobulin class.
- They are important reagents in many serological tests.
10.2: Strength of Antigen-Antibody Binding
- The binding of an antigen to its specific antibody is mediated by the interaction between the epitope of an antigen and the binding site of its antibody.
- Noncovalent bonds can be formed during antigen–antibody binding.
- Affinity: The energy of the interaction of a single epitope on an antigen and a single binding site on a corresponding antibody. * Cross-reaction: Antibodies bind with a lower strength to antigens that are structurally similar to the immunogen.
- Avidity: The overall strength of the binding of an antibody and an antigen. * It reflects the combined synergistic strength of the binding of all the binding sites of antigens and antibodies rather than the sum of individual affinities. * It also reflects the overall stability of an antigen–antibody complex that is essential for many serological assays.
\
10.3: Antigen-Antibody Binding Reactions
Primary Reactions
- It is the binding of a single epitope of an antigen (Ag) and a single binding site of an antibody (Ab) to form an antigen–antibody complex.
Secondary Reactions
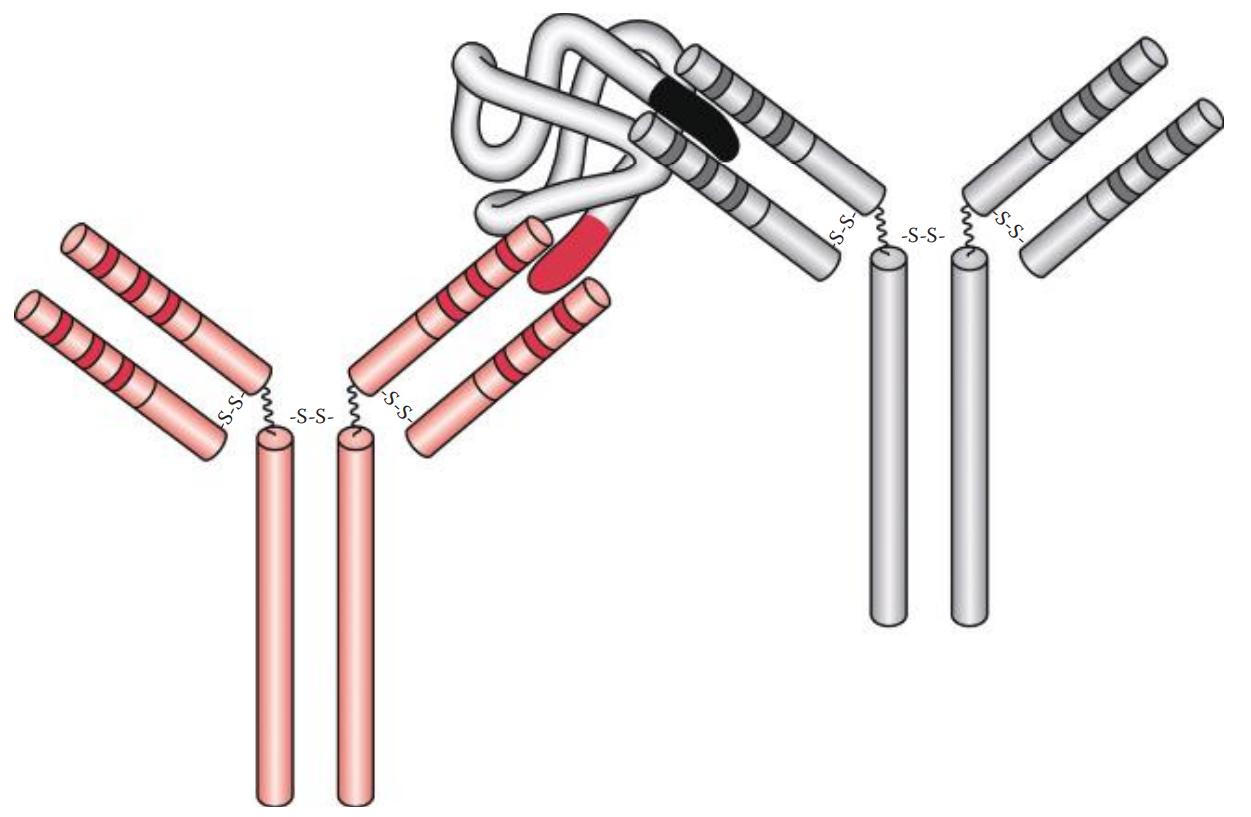
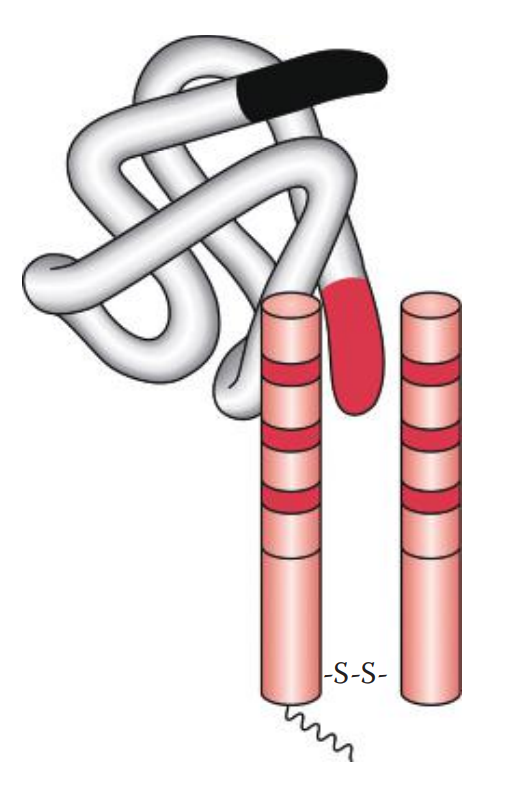
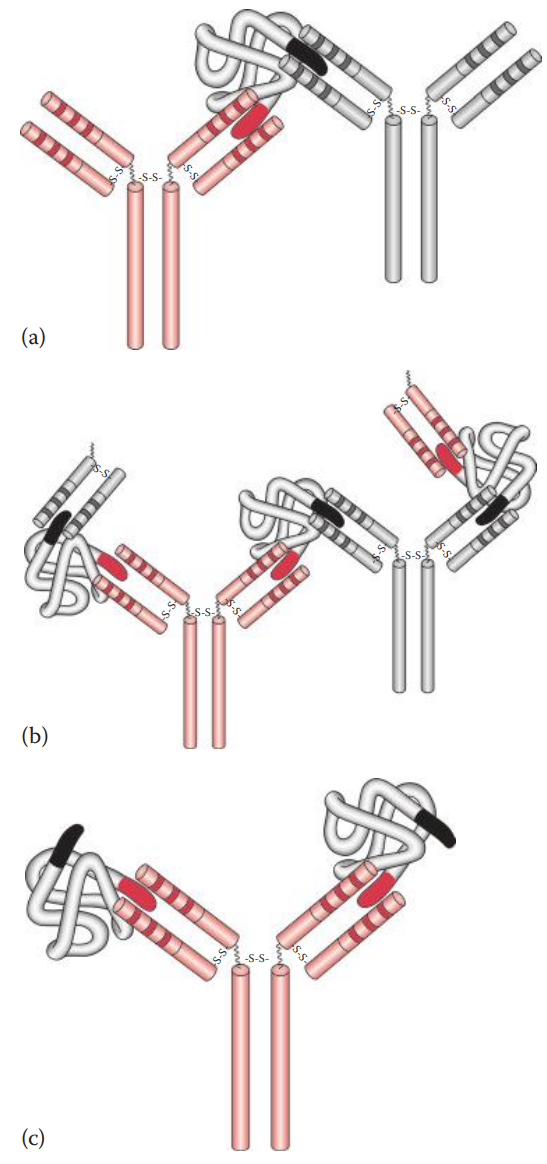
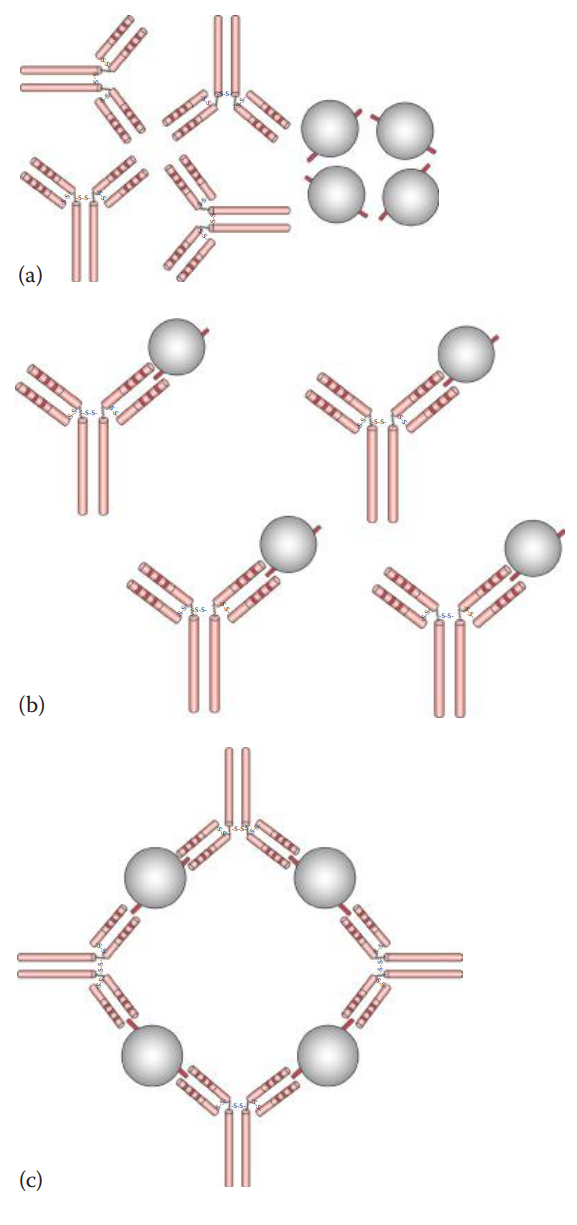
- Precipitation * If a soluble antigen is mixed and incubated with its antibody, the antigen–antibody complexes can form cross-linked complexes at the optimal ratio of antigen-to-antibody concentration. * Precipitins: Antibodies that produce such precipitation. * If an increasing amount of soluble antigen is mixed with a constant amount of antibody, the amount of precipitate formed can be plotted. * Precipitin Curve: Illustrates the results observed when antigens and antibodies are mixed in various concentration ratios. It has three zones: * Prozone: Here the ratio of antigen–antibody concentration is low. * The Zone of Equivalence: As the concentrations of antigen increase, the amount of precipitate increases until it reaches a maximum. * Post zone: Here the ratio of antigen–antibody concentrations is high or in excess.
\
- Agglutination * If the antigens are located on the surfaces of cells or carriers antibodies can bind to the surface antigens and can form cross-links among cells or carriers, causing them to aggregate. * If the antigen is located on an erythrocyte, the agglutination reaction is designated hemagglutination. * Initial Binding: The first step of the reaction involves antigen–antibody binding at a single epitope on the cell surface. * Lattice Formation: The second step involves the formation of a cross-linked network resulting in visible aggregates that constitute a lattice. * Agglutinins: Antibodies that produce agglutination. * Complete Antibody: Capable of carrying out both primary and secondary interactions that result in agglutination. * Incomplete Antibody: An antibody that can carry out initial binding but fails to form agglutination.
\