Biology 1020 Final Exam #1
Evolution is the process of change that has allowed organisms to adapt to their environment.
\ Hierarchy of organization: Biosphere, ecosystem, community, population, organism, organs and organ systems, tissues, cells, organelles, molecules.
\ All living things on Earth are within what is called the biosphere: Consists of all environments on Earth that support life, Includes most areas of land, water bodies and the lower atmosphere, Within the biosphere.
\ Ecosystems: Broad groups that can be broken down all of the way to the smallest levels of molecules. Ecosystems encompass all living organisms and non-living matter existing within a particular environment. This includes all components of the environment with which the living organisms interact, air, soil, sunlight and water.
\ Communities consist of all of the different organisms that are living in a particular \n ecosystem.
\ Populations are all of the individuals of a particular species living within a particular area.
\ Organisms are the individual unit of the population. Example: A lady bug from the lady bug population, a deer from the deer population.
\ Organ Systems are groups of several organs which work together to perform specific functions, Example: The nervous system, the respiratory system, the circulatory system.
\ Organs are structures comprised of different tissues, tissues work together as a group to perform specific functions. Example: the heart is composed of muscle tissue, connective tissue etc. Tissues are made of similar cell types and each tissue has a specific function, Each organ is made of several different tissue types.
\ Cells are membrane bound structures that form the individual units of living matter. May be single celled or multi-cellular organisms.
\ Organelles are the various functional components present on the \n inside of the cell.
\ Molecules are made up of a cluster of atoms, example: DNA, protein, sugar molecules.
Lecture #2:
\ Organisms within an ecosystem interact with both the living and the \n non-living components of their environment.
\ Producers of an ecosystem provide food for the other organisms \n present. Producers include plants and other photosynthetic organisms.
\ Consumers within an ecosystem eat plants and other animals.
\ Decomposers breakdown wastes and dead organisms recycling \n nutrients so that they can be used for biosynthesis. Examples of decomposers are fungi, bacteria, and small animals present in the soil.
\ Ecosystems can be characterized in 2 ways: Recycling of chemical nutrients and Energy flow.
Recycling of Chemical nutrients: Basic chemicals needed for life (carbon, nitrogen, oxygen, etc) flow from air and soil to plants, animals and decomposers and then back to the air and soil.
Energy Flow: Energy is constantly gained and lost from an ecosystem. Energy enters an ecosystem when light from the sun is absorbed by plants and other photosynthetic organisms. Energy exits an ecosystem as heat.
\ The cell is the basic unit of life. Cells are the lowest structural level capable of performing all activities necessary for life. Cells are membrane bound. All cells have DNA as their genetic material.
\ All living things are composed of either: Prokaryotic cells (Bacteria and archaea) and Eukaryotic cells (Protists, fungi, plants and animals.)
\ DNA stands for Deoxyribonucleic acid.
\ The genome is the entire DNA content of the organism, arranged into chromosomes. Contained within the boundaries of the cell.
\ DNA arranged as chromosomes is organized into functional units called genes.
\ There are more than 1.8 million species that are known and named. Estimates of the true number of species range from 10 million to 400 million.
\ Taxonomy is the branch of biology that names and classifies these species into groups based on similarity.
\ All life can be organized into three domains: Domain Bacteria (Prokaryotes), Domain Archaea (Prokaryotes) and Domain Eukarya.
\ There are three kingdoms within the domain Eukarya, they contain multi-cellular organisms grouped according to nutritional diversity.
- Kingdom Plantae: photosynthetic plants.
- Kingdom Fungi: molds, yeasts and mushrooms.
- Kingdom Animalia: animals: vertebrates and invertebrates.
- **Protists: single celled protozoans and algae (single and multi-cellular), formerly a fourth Kingdom.
\ Deductive reasoning moves from a general statement down to a specific conclusion.
Example: \n Premise #1: all living things are composed of cells \n Premise #2: human beings are living \n Deductive reasoning leads to the conclusion human beings must then \n be composed of cells.
\ To get a true hypothesis it must be both: Testable: there must be some way of testing whether or not it is correct and Falsifiable: experimentation must be able to show that it is not true.
Lecture #3
\ Polar covalent bonds form when two atoms involved in a covalent bond do not have equal electronegativity, one of the atoms desires the electrons in the bond more than the other. Molecules that contain unequal sharing of electrons are polar molecules.
\ Hydrogen Bonds: oppositely charged atoms in different water molecules form hydrogen \n bonds with one another. The partial positive charge on the hydrogen atom is attracted to partial negative \n charge on atoms within OTHER molecules. These bonds are weaker than both covalent and ionic bonds and Hydrogen bonds create molecular networks, these networks can occur between identical molecules or different molecules.
\ Cohesion of water molecules: Hydrogen bonding keeps water molecules close to \n one another that creates a complex structure that is referred to as cohesion which allows water to move against gravity from root to shoot in plants.
\ Water is also subject to adhesion. The molecules hydrogen bond to the walls of the vessels in the plant preventing them from falling back down.
\ Kinetic energy is the energy associated with motion, the faster the atoms and molecules move the greater the kinetic energy.
\ Heat is a measure of the total kinetic energy of a body. Volume dependent. Heat always passes from a hotter object to a cooler object when brought into contact with one another, molecules in the cooler object then speed up because of the gain in kinetic energy.
\ Temperature is the average kinetic energy of molecules. Volume independent.
\ A calorie is the amount of heat needed to raise the temperature of 1 gram of water by 1C. \n • 1cal= 4.184 Joules.
\ Specific heat is the amount of heat that must be absorbed/lost in order to change the temperature of 1 gram of a substance by 1C
\ Water has a very high specific heat compared to other substances.
\ Added heat must first be used to break hydrogen bonds. After the hydrogen bonds are broken the heat can then be used to increase the temperature of the water. When water is cooled heat is released as hydrogen bonds are reformed between water molecules, this results in a decreased speed of the molecules and a decrease in temperature.
\ Water is an excellent solvent, It can dissolve many ionic compounds such as NaCl. \n
Water exists in three primary states: Solid, Liquid, and Gas (water vapor).
\ Solids are always more dense than gas where density is the number of particles (molecules) per unit area with the exception of water. Ice has a smaller density than liquid water which explains why ice cubes float in water. Due to hydrogen bonds.
\n When water freezes each water molecule forms four hydrogen bonds with adjacent water molecules resulting in the creation of a crystal lattice. These hydrogen bonds are extremely stable compared to In liquid water the hydrogen bonds are less stable and continually break and reform.
\ A solution is a liquid consisting of a uniform (homogenous) mixture of two or more substances. The solution consists of: \n • A dissolving agent called a solvent: usually water \n • A dissolved substance called a solute: examples include sugar and salt \n • **When water is the solvent the solution is referred to as an aqueous solution
\ Hydrophilic substances have an affinity for water. If these molecules are not too large they will dissolve in water, If they are too large they will form a colloid. \n \n Hydrophobic substances have zero affinity for water. Non-ionic and non-polar. They are comprised of non-polar covalent bonds, example: Oil and water will not mix.
\ Glucose: C 6H 12O6
1 mole= 6.02x1023
\ Molar Mass is the number of grams one mole of a substance weighs.
\ Molarity is the number of moles of solute/liter of solution.
\ Acids: Dissociate when placed in water, release H+ ions. The greater the amount (concentration) of H + in the solution, the lower the pH of the solution, lower pH means a greater acidity.
\ Bases: Dissociate in water also, release OH – ions. The greater the amount (concentration) of OH - in the solution, the greater the pH of the solution. A higher pH means that the solution is more basic.
\ pH Scale: Measured on a scale of 0-14. A solution with a pH of 7 is considered neutral, the amount of H+ in solution is equal to the amount of OH- in solution. Solutions with a pH of 1-6 are acidic, solutions with a pH of 8-14 are basic. Addition of H+ will cause the OH - to decrease, addition of OH- will cause the H+ to decrease.
\ Buffers are substances that allow a solution to offset large and \n potentially dangerous changes in pH from occurring.==[END OF FLASHCARDS]==
\
Lecture #4
All organic compounds contain carbon
\ Carbon has four electrons in its outer shell. To achieve a complete outer shell containing 8 electrons carbon forms four covalent bonds with other atoms.
\ Organic molecules contain carbon as a part of their framework. Exceptions include: carbon dioxide (CO2 ), carbon monoxide (CO) and carbonic acid (H2CO3 ).
\ Stanley Miller is responsible for showing that the non-living synthesis of organic compounds is possible, this is important because it supports evolution. He showed that complex organic molecules were able to arise under the conditions of early Earth. Volcanoes serve as a source of energy. \n
A flask of warm water represented the early sea, the water was heated so that some evaporated and moved into a second flask to simulate the early atmosphere.This atmosphere consisted of hydrogen, water vapor, methane and ammonia, all gases thought in the 1950s to have made up the ancient atmosphere. Electrodes discharged shocks into the flasks to simulate lightning. A condenser cooled the atmosphere, raining water and any dissolved particles back into the initial flask the miniature sea. After one week Miller found a variety of organic compounds in the solution including some amino acids.
\ Carbon has 6 electrons, 2 electrons in the inner most electron shell, and 4 electrons in the outer most electron shell.
\ Carbon can form single, double or even triple covalent bonds in order to satisfy the octet rule and achieve chemical stability.
\ Hydrogen = 1 Valence electron
Oxygen = 2 Valence electron
Nitrogen = 3 Valence electron
Carbon = 4 Valence electron
\ Hydrocarbons are organic molecules consisting of only carbon and hydrogen.
\ Isomers are compounds with the same molecular formula but different structures. Types of isomers:
- Structural isomers
- Cis/trans isomers
\ Structural isomers: Differ in the covalent arrangement of atoms, straight vs branched. Number of possible isomers increases with the number of carbon atoms.
\ Cis isomer: Both X substituents are on the same side of the double bond. \n Trans isomer: The X substituents are on opposite sides of the double bond. \n
Functional groups replace hydrogen in chemicals.
All Functional Groups:
Hydroxyl group: -OH, polar
Carbonyl group (ketones and aldehydes): C=O. Ketones if the carbonyl group is within a carbon skeleton. Aldehydes if the carbonyl group is at the end of a carbon skeleton.
Carboxyl group: O=C-OH, acts as an acid.
Amino group: H-N-H, acts as a base.
Sulfhydryl group: -SH, two sulfhydryl groups can react,forming a covalent bond.
Phosphate group: Contributes negative charge,
O
= \n O --- P--- O-
-
O-
Methyl group: CH3, non-polar, non-reactice. Addition of Methyl to DNA, or to molecules bound to DNA, affects the expression of genes. Arrangement of methyl groups in male and female sex hormones affects their shape and functions.
Lecture #5
ATP: Energy currency of the cell.
Organic molecule is adenosine. Composed of adenine and ribose sugar. Attached to three phosphate groups via high energy covalent bonds. Inorganic phosphate can be cleaved off of the ATP in order to release energy
\ There are four main groups of organic molecules: Carbohydrates, lipids, proteins and nucleic acids.
\ Macromolecules are polymers built from monomers.
\ Monomers are the individual units of the molecules.
\ Enzymes are protein polymers that increase the rate of reactions
\ \n Water is formed because one monomer contributes a hydrogen and the other monomer contributes a hydroxyl group. This reaction is repeated until the entire polymer is synthesized. \n
Polymers are broken down by adding water across a covalent bond. Hydrolysis reaction catalyzed by enzymes.
\ Carbohydrates include sugars and sugar polymers.
\ All sugars consist of a carbonyl group and \n many hydroxyl groups.
\ Sugar names generally end in ‘ose’.
\ Glucose is often depicted as a linear molecule
\ Sugars are used: As a fuel source in the cell. Also used to assemble other complex molecules for ex.amino acids. If not immediately used in one of the above reactions the sugar will be stored as disaccharides \n or polysaccharides.
\ Plant and animal cells store excess sugars for later use.
\ Starch is the plant storage form of glucose. Stored as granules in the plant cell \n inside of structures called plastids. Hydrolysis will release glucose from the starch when energy is needed. Animal cells also have the enzymes needed to hydrolyze starch.
\ Amylose: unbranched starch.
\ Amylopectin: branched starch, includes 1-6 linkages.
\ Glycogen: animal storage polymer for glucose. More branched than amylopectin. Stored in human liver and muscle cells. Hydrolyzed at an increased rate when the cell needs energy but doesn’t persist for very long in the cell unless it is replenished.
\ Cellulose: a major component of the plant cell wall
\ Glucose exists in two ring structures: alpha and beta.
\ Starch: all glucose monomers are alpha.
Cellulose: all glucose monomers are beta.
\ Animals do not have the enzyme to cut beta linkages. Passes through us as insoluble fiber. Cleans out the intestinal tract.
\ Chitin: polysaccharide component of fungal cell wall.
\ Lipids are not formed from repeating monomers, they form a diverse group including fats, phospholipids and steroids. Do not mix with water, Non-polar hydrocarbons
\ Fats are molecules assembled via dehydration reactions
\ Fatty acids tails are attached to glycerol via ester linkages, can be one, two or three fatty acids attached. May be the same fatty acids or different fatty acids.
\n Fatty acid is composed of: Carboxylic acid and a non-polar tail.
\n Triacylglycerol (triglyceride) is formed when three fatty acids are attached to the glycerol neck.
\ Fats may be saturated or unsaturated, saturated fats don’t have any double bonds, which allows for tight packing. Solid at room temperature.
\ Unsaturated fats contain one or more double bonds, Liquid at room temperature. Naturally occurring varieties are in the cis conformation, that creates a kink in the chain.
\ Omega-3 fatty acids cannot be synthesized in the body and must be supplied by diet.
\ Fats are used in the body for energy storage. One gram of fat stores twice as much energy as one gram \n of sugar. Also protects organs and provides insulation.
\ Phospholipids: Form a major component of cell membranes. Phospholipids spontaneously assemble into the membrane structure.
2 fatty acids are attached to a glycerol molecule. Fatty acids are hydrophobic and do not interact with water. The third hydroxyl group of glycerol is used to attach a phosphate group, contains a negative charge. Also has a polar head group attached to the phosphate that is hydrophilic and interacts with water.
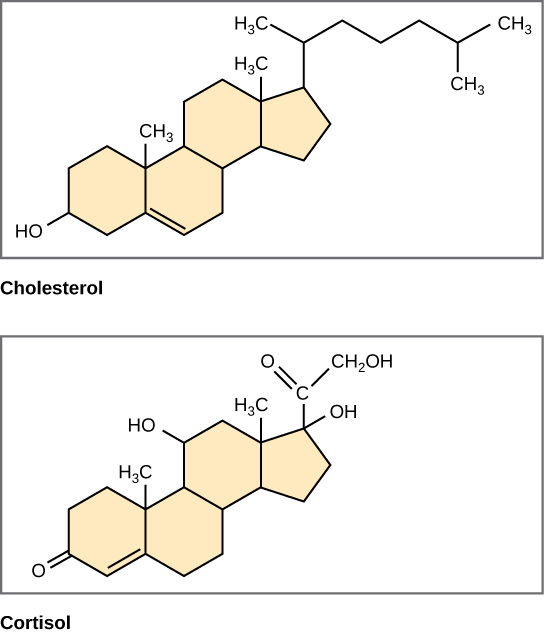
\ Steroids: Four fused rings that form a carbon skeleton. Include vertebrate sex hormones and cholesterol.
Proteins are the work horse of the cell, more than 50% of the cell’s dry weight.
Proteins function as: \n • Defense molecules \n • Enzymes \n • Storage \n • Transport \n • Cell communication \n • Structure \n • Movement
\n There are thousands of proteins that had varied 3D structures, leading to unique functions.
\ Proteins are made from one or more polypeptides associated with one another. Folded into their final functional structure.
\ Polypeptides are constructed from a unique combination of amino \n acids, there are 20 different amino acids.
The DNA sequence of the gene dictates the amino acid sequence of the protein. Amino acids are attached to one another via a dehydration reaction forming a covalent bond, peptide bond.
\ Amino acids are the monomers of polypeptide structures. Consist of a centrally located carbon atom: the alpha carbon. Consist of a carboxylic acid terminus (COO-). Consist of an amino terminus (NH 3+ )
There are four different groups of amino acids based on the type of R group present:
- Non-polar, hydrophobic
- Polar, hydrophilic
- Acidic: donate protons in solution becoming anionic
- Basic: accept protons in solution becoming cationic •
**acidic and basic amino acids are also hydrophilic, carry a charge.
\ All polypeptides will consist of an amino terminus and a carboxy \n terminus.
\ Chemical nature of the R groups determine the ‘personality’ of the protein
\ Protein has four different levels of structure:
- Primary Structure: • This is the amino acid sequence of the protein
- Secondary Structure: • Hydrogen bonds between different locations of the polypeptide sequence create secondary structure • Two types: alpha helices and beta-pleated sheets Proteins
- Tertiary Structure: • Formed from chemical interactions between R groups of individual amino acids that form the polypeptide • Disulfide bonds form between S of cysteine amino acids in the polypeptide chain
- Quaternary Structure: • Two or more polypeptides (in their tertiary structure) come together to form a functional molecule
\ Protein structure may be affected by salt concentration, temperature and pH. This is referred to as denaturation and leads to a loss of protein function.
\ Genes encode the primary amino acid sequence of a protein. Genes are made from DNA. DNA was discovered in 1953 by Watson and Crick. \n DNA: Carries the information needed to make proteins. Before protein can be assembled, DNA must be \n transcribed into another nucleic acid → RNA. Is able to self replicate allowing new cells to form.
Nucleic acids range from long to very long in length (1000s to 100s of thousands of nucleotides). \n This makes the number of potential nucleotide combinations infinite. The various combinations account for much of the genetic variation seen in the world.
\ Nucleic acid polymers are formed from nucleotide monomers. Each nucleotide has three components:
- A five carbon sugar that is either: • Deoxyribose (DNA) • Ribose (RNA)
- A phosphate group
- A nitrogenous base that is either a: i. Purine: adenine and guanine • Doubled ringed structures ii. Pyrimidine: cytosine, thymine (DNA only), uracil (RNA only) • Single ringed structures.
\ Nucleic acid polymers are joined to one another by covalent bonds via \n dehydration reactions.The phosphate group of one nucleotide monomer bonds to the sugar of the next nucleotide monomer. This results in a repeating sugar-phosphate backbone in the polymer.
\ DNA is always double stranded and is found as a double helix. Two polynucleotides are wrapped around one another. Nitrogenous bases always protrude from the sugar phosphate backbone into the center of \n the double helix. Nitrogenous bases on one strand always pair with nitrogenous bases of the second strand according to the following rule: \n • A::T \n • C:::G \n Strands are said to be complementary to one another. The amount of A=T and the amount of C=G. Adjacent strands are held together by hydrogen bonds. Most DNA molecules have thousands or even millions of base pairs. RNA is always single stranded.
Lecture #6
Microscopes were invented in 1590.Robert Hooke was the first to see and describe living cells in 1665, dead cells taken from Oak tree bark. Antony van Leeuwenhoek was the first to observe living cells in 1674, bacteria that he named animalcules.
Microscopes are critical for cytology, the study of cell structures. Artifacts: seen in the microscope image but are not present in the actual sample, which is a problem with all types of microscopy.
Two types of microscopy:
- Light microscopy: compound electron microscope
- Electron microscopy: Scanning electron microscope, Transmission electron microscope.
\ \n Light microscope: Any microscope that uses visible light to observe specimens. \n 1. Compound Light Microscope: Uses two lenses to observe specimens. \n i. Objective Lens: \n • This lens is located closest to the \n specimen \n • Magnifies the specimen \n • 10x-100x \n ii. Ocular Lens: \n • This lens is located within the eye \n piece of the microscope \n • Magnifies the specimen 10x
\ Calculating Magnification for a compound microscope: Total magnification= objective lens magnification X ocular lens magnification. Example: ocular lens= 10x, objective lens = 100x, total magnification= 10 x 100 = 1000.
\ If a microscope has a 6nm resolving power than it can distinguish two points if they are at least 6nm apart.
\ In order for two objects to be seen as distinct from one another light must pass in between them. The light that passes in between the objects must be of a short enough wavelength otherwise they will be seen as one object.
\ The General Principle of Resolution: The shorter the wavelength of light the better the resolution will be.
\ Electron microscopes: These microscopes use beams of electrons instead of light. Like the light of light microscopes, electrons also travel in waves but the waves are much shorter than light waves. Since electrons travel in smaller waves electron microscopes achieve a greater resolution.
Electron microscopes also achieve a much greater magnification (as high as 500,000x). The added ability of these microscopes allow us to view internal cell structures and viruses.
\ The smallest object visible with the human eye is 0.10mm. A compound microscope can be used to view an object as small as 0.20μm. An electron microscope can be used to view objects as small as 0.20nm. \n Using the same magnification an electron microscope will also provide an image with a better resolution than a light microscope.
\ Scanning Electron Microscopy (SEM): Can only be used to view the surface of an object. The specimen must be coated with a thin film of heavy metal, Ex) gold. Allows a wide range of magnifications to be achieved, (15x-100,000x).
\ Transmission Electron Microscopy (TEM): Used to examine internal cell structure, electron beams do not penetrate the thick cell wall. Requires the cell to be cut in order to be viewed, this process is called thin sectioning. These thin sections must then be stained before they can be viewed under the TEM, Uranium is an example of a stain that is used. Stains function to improve the contrast in between different cell structures.
\ Cell fractionation separates intracellular materials so that they may be studied in depth. Requires disruption of the plasma membrane in order to isolate cytoplasmic material. The sample is subjected to increasing g \n forces (accomplished by increasing the rpm setting) in a centrifuge. Lower speed centrifugation will sediment \n large cellular material at the bottom of the tube. The collection of material at the bottom of the tube is called the pellet.
\
Lecture #7
\ All living cells fall within one of two groups:
- Prokaryotes: • Pro=before karyon=nucleus • DNA is not bound by a membrane • Single, circular chromosome • Located in a region of the cell called the nucleoid • Do not have membrane bound nuclei • Can have additional external structures that promote attachment • Includes Bacteria and Archaea
- Eukaryotes: • Eu=true karyon=nucleus • DNA is located inside of a nucleus, bound by a double membrane • Multiple, linear chromosomes • Additional DNA is located in other areas of the cell • Ex) inside of the mitochondria • Also have membrane bound organelles • Perform various cellular functions • Includes protists, fungi, plants and animals
\ Common Features of All Cells
- Plasma Membrane: • Provides the boundaries of the cell
- Cytosol: • Viscous fluid that fills the boundaries of the cell • Internal cellular components are suspended within the cytosol • Together the cytosol and the suspended materials are called the cytoplasm
- Chromosomes: • Composed of DNA • Genetic material • Forms the ‘recipe’ for protein synthesis
- Ribosomes: • Serve to synthesize proteins according to information that originates within the DNA sequence • They are different sizes in prokaryotes and eukaryotes → excellent target for antibiotics → selective toxicity.
\ Size: Eukaryotes are much larger than prokaryotes. \n • Cells can only be so small before they loose the ability to perform key tasks \n • Size correlates very closely with function \n • Smallest cell: 0.10-1.0μm in diameter \n • Mycoplasma species \n • Small disease causing bacterium \n • Typical prokaryotic cell: 1-5μm diameter \n • Eukaryotes are 10-100μm in diameter
\ The plasma membrane forms the boundary of every cell. \n • A selective barrier \n • Allows vital nutrients and O2 to enter \n • Allows wastes such as CO2 to exit \n • The surface area to volume ratio of the cell is very important \n • This is especially true for cells that function in absorption/material exchange \n • An increase in cell size will not correspond to an increase in membrane surface \n area \n • The smaller the cell the greater the surface area: volume ratio will be \n • Bigger organisms have a greater number of cells \n • Cells are not larger
\ Nucleus: \n • The nucleus contains the bulk of all DNA inside of the eukaryotic cell \n • Some in the mitochondria and chloroplasts as well \n • ~5μm in diameter \n • Enclosed by a nuclear envelope: \n • Double membrane \n • Each is a bilayer structure \n • Contains pores \n • Regulate the entry/exit of protein \n and mRNA \n • Shape is supported by a protein framework attached to the inside face of the nuclear membrane \n • Nuclear lamina \n • **There may also be a nuclear matrix \n inside
\ Nuclear DNA is organized into units called chromosomes. \n • Chromosomes are long segments of DNA complexed together with protein \n • Protein organizes the DNA creating a more compact structure \n • DNA and protein together is called chromatin \n • When the cell is not dividing chromosomes are uncoiled and active \n • During periods of cell division the chromosomes are condensed and easily visible as individual units \n • The number of chromosomes in the nucleus of a cell is dependent on the species and the class of cell \n • Humans have 46 chromosomes in somatic cells: diploid \n • We have 23 chromosomes in sex cells: haploid \n • Dogs have 78 chromosomes in somatic cells: diploid \n • They have 39 chromosomes in sex cells: haploid
\ The Nucleolus: \n • Prominent in the nucleus when the cell is not dividing \n • During cell division the nucleolus disappears \n • Protein imported from the cytoplasm is complexed together with ribosomal RNA (rRNA) made from the DNA template. \n • Forms large and small ribosomal subunits \n • Exported to the cytoplasm and assembled into ribosomes \n • Cells may have more than one nucleolus \n • mRNA made in the nucleus from the DNA template is exported through the nuclear pores to the cytoplasm when it will be used by the ribosome to synthesize protein.
\ Ribosomes:
• Made of rRNA and protein
• If a cell synthesizes large quantities of protein it will contain many ribosomes
• Nucleoli will also be more prominent.
• Ribosomes exist in two cellular locations:
- Free in the cytoplasm: • Make proteins destined to stay in the cell
- Bound ribosomes: • Attached to the endoplasmic reticulum and the nuclear membrane • Make protein destined to: • Be inserted into membranes • Be packaged into organelles • Be secreted from the cell • A cell that secretes large quantities of protein will have more bound ribosomes
\ The endomembrane system: \n • Consists of a number of membranes \n • Some are physically connected \n • Some are vesicular \n • Vesicles are sacs composed of membrane \n • Includes the: \n • Nuclear membrane \n • Endoplasmic reticulum \n • Golgi apparatus \n • Lysosomes \n • Vacuoles \n • Plasma membrane \n • The nuclear membrane is connected to the endoplasmic reticulum \n • The bulk of these organelles work together to synthesize, store and export molecules.
\ The endoplasmic reticulum can be smooth or rough: \n • Smooth ER does not have any bound ribosomes \n • Rough ER has ribosomes attached giving it a rough appearance \n 1. The Smooth Endoplasmic Reticulum (smooth ER) functions in: \n i. Lipid production: oils, phospholipids and steroids \n • Ex) ovarian cells and testicular cells synthesize sex hormones which are steroids, therefore these cells are rich in smooth endoplasmic reticulum. \n ii. Detoxification of drugs \n • As a person consumes a greater amount of drugs, the amount of smooth ER and associated detox enzymes increaseà increased drug tolerance. \n • Higher doses of the drug are necessary to achieve the same effect \n • The smooth ER does not respond with specificity to a drug so tolerance to other drugs will increase as well. \n iii. Stores calcium ions.
\ The Rough ER functions to: \n • Produce more membrane phospholipids which are then inserted into the membrane \n • Bound ribosomes produce proteins that will be: \n • Inserted into the membrane of the rough ER \n • Transported to other organelles \n • Secreted by the cell \n • Ex) insulin is a protein produced by bound ribosomes and secreted from the cells in the pancreas into the blood.
\ Synthesis, modification and packaging of a secretory protein:
- As the polypeptide is synthesized by a bound ribosome it is inserted into the cavity of the rough ER via a protein pore • Once inside the ER the protein folds into its final 3D shape
- Short sugar chains are often linked to the polypeptide, producing a glycoprotein
- Once ready for export the molecule is packaged into a transport vesicle
- The transport vesicle now buds from the ER membrane and the protein travels to the golgi apparatus where it will be further processed.
\ The Golgi Apparatus \n • Transport vesicles exit the ER and travel to the golgi apparatus \n • The golgi apparatus consists of flattened sacs stacked atop one another \n • The flattened stacks are called cisternae and are not interconnected \n • The number of golgi stacks correlates with the quantity of protein that the particular cell secretes. \n • The golgi apparatus functions to receive and modify/store proteins that have arrived from the ER.
\ One side of the golgi apparatus serves as the receiving end and the other side serves as the shipping end • The receiving end serves to receive transport vesicles from the ER (called the cis face) • The shipping end gives rise to vesicles which bud off and travel to other sites (called the trans face) • Protein is modified as it travels from the receiving end to the shipping end • Carbohydrate groups on glycoproteins may be modified/substituted in the golgi, phosphate groups may also be added to proteins serving as identification tags later on in protein transport • Membrane proteins may also be modified • Products of the golgi may be:
- Secreted from the cell
- Added to the plasma membrane or to organelle membranes (ex) lysosome membranes
\ The golgi manufactures sugars destined for secretion from the cell \n • Secreted from the trans face of the golgi \n • Eventually fuse with the plasma membrane for export \n • Different cisternae have unique enzyme packages \n • Products are refined in stages \n • May also have external molecules on vesicle structure for docking to their target.
\ Lysosomes are membrane bound sacs containing digestive enzymes \n • Lysosomal enzymes and lysosomal membranes are made by the rough ER and modified by the golgi apparatus. \n • Digestive enzymes of the lysosome must be kept within a membrane because they are acidic and would damage the other cell components \n • Lysosomes may function by fusing with vacuoles containing food that has been engulfed by the cell \n • Lysosomal enzymes break down the food and release the nutrients back into the cell where they can be metabolized to generate energy.
\ Lysosomes can also fuse with: \n • Vacuoles containing engulfed bacteriaà Kills the organism \n • Damaged organellesà called autophagy \n • Breaks them down to release their components for cell use \n • Allows the cell to continually renew itself \n • Unique 3D shape of protein protects lysosome from self-destruction \n • Tae-Sachs Disease is a rare disorder that results when a lipid digesting enzyme is missing from lysosomes \n • Lysosomes become engorged with fatà accumulates in the brain, impairing brain function \n • A child with Tae-Sachs disease will die within a few years
\ Vacuoles are multi-functional membrane bound sacs \n • Derived from the ER and the golgi \n • Can store food, transport solutes and remove excess water from the cell \n • The central vacuole of a plant cell functions to: \n • Store sap \n • Breakdown ingested material with its enclosed enzymes \n • Store water and enlarge \n • Store necessary nutrients, other chemicals and waste products \n • Stores excess ions \n • Some vacuoles in flowering plants also contain pigments which work to attract pollinators \n • They can also contain poisons that protect the plant from predation
\ Mitochondria: \n • Hundreds in eukaryotic cells \n • Dependent on quantity of metabolism occurring \n • Mitochondria function to carry out cellular respiration in all eukaryotic cells \n • Cellular respiration is the conversion of chemical energy from foods and sugars into ATP using oxygen. \n • Mitochondria are enclosed by a double membrane: \n • Both are phospholipid membranes that contain embedded proteins that are unique to the mitochondria.
\ There are two regions of space in the mitochondria:
- Inter-membranous space: narrow region between the inner and the outer membrane
- Mitochondrial matrix: enclosed by the inner mitochondrial membrane • Houses DNA, ribosomes and many of the enzymes needed for cellular respiration • The inner membrane has many folds called cristae and contains protein molecules that function to synthesize ATP • Increased surface area allows for greater ability to synthesize energy
\ Photosynthesis is the conversion of light energy from the sun into chemical energy of sugar molecules \n • Eukaryotes that are capable of photosynthesis (plants and algae) use an organelle called the chloroplast \n • The chloroplast contains many membranes which partition the organelle into compartments \n • The chloroplast contains an inner and outer membrane separated by a thin inter-membranous space \n • The area bound by the inner membrane contains fluid called the stroma \n • Also contains enzymes, DNA and ribosomes.
\ Thylakoids are the network of interconnected sacs located inside of the chloroplast \n • The space inside of the thylakoid is called the thylakoid space \n • Thylakoids contain chlorophyll pigments \n • When the thylakoids are stacked atop one another they are called granum (grana=pleural) \n • The grana function to absorb the sun’s energy.
\ Mitochondria and chloroplasts evolved by an endosymbiosis. \n • Both mitochondria and chloroplasts are similar to prokaryotes because they: \n • Have a single circular molecule of DNA \n • Have ribosomes smaller than eukaryotic ribosomes \n • Reproduce by splitting in half \n • Are enclosed by a double membrane and the inner membrane is very similar to the plasma membrane of prokaryotes. \n • The endosymbiotic theory proposes that mitochondria and chloroplasts were both originally prokaryotes that began living inside of larger cells \n • Endosymbiont refers to a small cell living within another cell (host cell).
\ Peroxisomes are bound by a single membrane. \n • Enzymes inside oxidize/reduce cell components forming H 2 O2 \n • H 2 O2 is toxic \n • An enzyme is also present to convert H2 O2 to water \n • Used in fatty acid breakdown and detoxification \n • Enzymes and contents of peroxisome are toxic and therefore sequestered
\ The cytoskeleton is a network of protein fibers that extend throughout the cell cytoplasm and: \n • Provide structural support \n • Assist in the movement of organelles \n • Anchor organelles within the cell \n • Assist in movement of the cell \n • Requires interaction with motor proteins \n • Maintain cell shape \n • Can be rapidly assembled/taken down \n • The cytoskeleton is composed of three different proteins: \n • Microfilaments \n • Microtubules \n • Intermediate filaments
\ 1. Microfilaments: \n • Solid rods composed of globular protein called actin \n • Arranged in a twisted double chain \n • Made to withstand force \n • Forms a three-dimensional network that helps support the cell shape \n • Creates a more viscous cytoplasm near to the cytoplasmic surface of the plasma membrane \n • Microfilaments are also very important in muscle contraction \n • Thin filaments
\ 2. Intermediate Filaments: \n • Composed of various fibrous proteins \n • Have a ropelike structure \n • Used to reinforce cell shape and to anchor organelles in place \n • Usually permanent cell fixtures \n • Made to withstand force
\ 3. Microtubules: \n • Straight, hollow tubes \n • Composed of protein called tubulin \n • Each unit is a dimer composed of two slightly different \n tubulin proteins \n • Length of the microtubule is increased by adding dimer \n units to the end of the structure \n • Centrioles are composed of microtubules which grow \n out of an area of the cell called the centrosome \n • Important in cell division \n • The microtubules also shape and support the cell \n • Organelles can also be moved along microtubules \n • Eukaryotic cilia and flagella are made from microtubules \n • Extend from the surface of some cells \n • Important for cellular motility
\ The plant cell wall is an extracellular structure \n • Unique to plants \n • Protects against osmotic rupture \n • Maintains cell shape \n • Greater thickness than the plasma membrane \n • Varied composition \n • Cellulose is most common \n • Adjacent cells are connected to one another via plasmodesmata \n • Middle lamella is a thin sticky layer that glues adjacent cells together \n • Some plant cells have a secondary cell wall \n • Thickened for protection and support
\ The extracellular matrix is composed of glycoproteins \n • Mainly collagen in animals \n • Embedded in proteoglycans \n • Sugar/protein complex \n • Fibronectin attaches proteins to the extra-cellular matrix \n • Binds to cell surface receptors \n • Ex) integrins \n • Plant cells are joined together by plasmodesmata \n • Membrane bound channels filled with cytoplasm \n • Animal cells are connected by tight junctions, desmosomes and gap junctions.
\
Lecture #8
Plasma Membrane \n • Selectively permeable \n • Allows hydrophilic molecules to pass through the membrane with ease \n • Allows nutrient uptake and waste removal \n • Phospholipid membranes are described as fluid mosaic \n • The mosaic portion of the membrane refers to the proteins floating within \n • These proteins have varied function \n • There are more than 50 different proteins in the plasma membrane of a red blood cell alone \n • Different cell types have different proteins and in a single cell the plasma membrane and the organelle membranes contain different proteins \n • The fluid portion refers to the proteins being able to move about in the membrane.
\ Membranes \n • Fatty acids are amphipathic and their double bonds prevent them from packing closely together \n • Keeps the membrane fluid \n • Fatty acids and thus the membrane is held together by hydrophobic interactions \n • Phospholipids can move sideways within the membrane \n • Can also move from one leaflet of the membrane to another \n • Membranes with different functions will have very different structure and chemical composition
\ Membrane Fluidity \n • Membrane fluidity is maintained with decreasing temperature \n • Membrane fluidity is needed for function \n • Eventually as temperature continues to decrease the phospholipids will solidify into position \n • Phospholipid composition dictates freezing temperature \n • Saturated vs unsaturated fatty acids \n • Long chain vs short chain fatty acids \n • Organisms evolved to have phospholipid composition that keeps the membrane semi-fluid at environmental temperatures \n • Cholesterol is found in animal plasma membranes \n • At high temperatures the membrane is kept from melting because cholesterol hinders phospholipid movement \n • At low temperatures cholesterol interferes with close packing of fatty acid tails and interferes with freezing \n • Cholesterol behaves as a temperature buffer.
\ Membrane Protein Function • ‘mosaic’ of the fluid mosaic model • Very diverse • Functional portion of the membrane • Two major classes:
- Integral • Penetrate membrane interior • Most are trans-membrane proteins • Consist of hydrophobic amino acid stretches • Some penetrate only part of the membrane interior
- Peripheral membrane proteins: • Not embedded in the membrane • Loosely bound to the surface of the cell • Attachment is often to exposed integral proteins • Attachment on the cytoplasmic side is to the cytoskeleton • Attachment on the extracellular side is to the extracellular matrix
\ Membrane Proteins
Six primary functions of membrane protein include:
- Providing a stronger membrane framework: integrins for example • Integrins bound to the cytoskeleton on the inside of the cell and to the extracellular matrix on the outside of the cell
- For cell to cell recognition: • Ex) Glycoproteins • Outside surface of the membrane has carbohydrates bound to either membrane proteins or membrane lipids • Carbohydrates vary among different species, different individuals and different cell types • Allow immune cells to recognize and reject foreign cells such as bacteria and transplanted tissues
- In order to form tight membrane junctions between different cells
- Membrane proteins function as enzymes • Carrying out sequential steps in a pathway
- Some membrane proteins function as receptors • Bind to chemical messengers that have been secreted from other cells • Receptor is very specific to the protein messenger Ex) a hormone • Binding of the messenger triggers a series of events that turn on or turn off processes occurring within the cell • Referred to as signal transduction
- Membrane proteins are also important in transport • Membranes are selectively permeable • Only non-polar molecules are able to directly pass through the membrane • Polar molecules and ions are not able to move through the hydrophobic interior • Require transport proteins to enter and exit the cell
\ Membrane Carbohydrates \n • Membrane carbohydrates are involved in cellular recognition \n • Allows removal of foreign cells from the body \n • Carbohydrates are short branched chains \n • Less than 15 sugar unitsà called oligosaccharides \n • Covalently attached to lipidsà glycolipids \n • Can also be covalently attached to membrane proteinsà glycoproteins \n • Attached sugars vary: \n • From species to species \n • From different individuals of the same species \n • From cell to cell
\ Membrane Transport \n • There is continuous bidirectional movement across the plasma membrane \n • Nutrients enter and wastes exit \n • Oxygen moves into the cell and carbon dioxide exits the cell \n • Important for ion regulation \n • Maintains the selective permeability of the cell \n • Diffusion rate through the plasma membrane varies according to the size, shape and chemical nature of the material \n • Non-polar compounds such as O 2 , CO2 and hydrocarbon molecules are able to cross the plasma membrane easily from areas of [high] to areas of [low] \n • No assistance required \n • Polar and charged molecules may not pass directly through the membraneà Diffusion occurs very slowly \n • **transport proteins are used to assist and regulate the movement of solutes \n • Allow membranes to be permeable to polar and charged materials
\ Two types of transport proteins:
- Channel proteins: form a hydrophilic channel through the membrane • Ex) aquaporins allow the movement of 3 billion water molecules per second
- Carrier Proteins: • Physically grab and escort material through the membrane • These proteins undergo conformational changes • Show a high degree of substrate specificity • Glucose transporters show a 50,000 fold increase in rate of transport
\ Passive Transport \n • Diffusion is the ability of particles to spread out through any region of \n space \n • Movement is from regions of high concentration to regions of lower concentration \n • Movement of particles will continue until concentrations are equal on either side of the \n membraneà Ex) perfume \n • The movement of molecules is possible because of the heat available \n • Higher temperature yields faster moving molecules \n • A passive process and requires no workà occurs spontaneously \n • Non-polar material like O 2 and CO2 move naturally \n across membranes from [high] to [low] \n • Ex) O2 moves from the lungs into the blood \n • Polar molecules and ions can still diffuse \n across the membrane → require transport proteins
\ Osmosis \n • Osmosis is the movement of water across a semi-permeable membrane \n • Always a passive process \n • Membrane is permeable to water but not to a solute \n • Ex) Membrane is separating two solutions with different concentrations of glucose \n • Water will move across the membrane until the solute concentrations are equal on both sides \n • Membrane is impermeable to the solute so solute cannot move \n • Water will ALWAYS move from regions of LOW solute concentration to regions of HIGH solute \n concentration \n • Always occurs regardless of the identity of the solute
\ Water Balance Without a Cell Wall
• Tonicity describes the ability of solution to cause a cell to gainor loose water
• Tonicity is mainly dependent on the concentrations of solute that are unable to cross a membrane
- Animal cell is immersed in solution that has equal solute concentration to the cell →isotonic solution relative to the cell • No net water movement
- Animal cell is immersed in solution that has lower solute concentration than the cell → hypotonic solution relative to the cell • Net movement of water into the cell causing the cell to expand and eventually burst →called osmolysis
- Animal cell is immersed in solution that has a higher solute concentration than the cellà hypertonic solution relative to the cell • Net movement of water out of the cell causing the cell to dehydrate and shrivel
\ Water Balance Without a Cell
Plant and animals cells must regulate water gain/loss in order to \n survive → called osmoregulation \n • Organisms that lack a cell wall may live in an isotonic environment to compensate \n • Organisms that live in hypotonic environments need mechanisms to prevent cell lysis \n • Altered membrane permeability to water will slow movement of water into the cell \n • Contractile vacuoles: have pumps to force water out of the cell
\ Water Balance and The Cell Wall \n • Plants, bacteria, fungi and some protists all have a cell wall \n • Allows structural maintenance when the organism is placed in a hypotonic environment \n • Ex) a plant immersed in rain water \n • Cell wall will only swell so much before the wall exerts a counter force on the cell called turgor pressure \n • The cell will be turgor (firm) once this force is applied \n • Allows mechanical support for small plants \n • The cell will be flaccid (limp) in isotonic environments \n • In hypertonic environments the cell wall does not offer assistance \n • The cell membrane pulls away from the cell wall → called plasmolysis \n • The cell shrinks which can lead to wilting and death.
\ Facilitated Diffusion
Transport proteins facilitate the movement of polar molecules and ions across membranes \n • If this allows polar molecules to diffuse down their concentration gradient it is referred to as facilitated diffusion. \n • Facilitated diffusion is a passive processà does not require energy \n • It is driven by the concentration gradient \n • Transport proteins substantially increase the speed that polar molecules are able to cross the membrane \n • The transport protein is usually very specific to certain materials.
\ Facilitated Diffusion
- Channel proteins: • Provide a corridor for the movement of hydrophilic/ionic materials • Can be gated: • Only open in response to certain stimuli: usually electrical, chemical, mechanical • Ex) K + channels in the neuron membrane open in response to voltage changes
- Carrier Proteins: • Ex) glucose transporter.
• Interior of the transporter changes shape to allow movement of materials across the cell membrane
• Binding of the molecule is triggers the change in shape
• Move materials down their concentration gradient→ passive process
\ <<END OF AUTOMATED NOTES. 95 TOTAL.<<
\ Facilitated Diffusion
Transport proteins are always specific to the molecules that they transport \n • The more transport proteins present in a membrane for a particular solute, the greater the rate of diffusion will be across the membrane \n • Many sugars, amino acids and sometimes even water all use facilitated diffusion to cross the membrane \n • Water is small but polar \n • Requires protein channels to diffuse across the plasma membrane at a reasonable rate →aquaporins \n • Specific for water transport \n • Very are important in the kidney
\ Active Transport
Active transport involves the expenditure of energy to move a solute against its concentration gradient \n • From low concentration to high concentration \n • The energy needed to power this transport is provided: \n • Directly by ATP \n • By another energy source available in the cell \n • All proteins involved in active transport are carrier proteins \n • Allows the maintenance of concentration gradients across the plasma membrane \n • Ex) Movement of 3Na+ out of the cell and 2K+ into the cell coupled to the use of 1 ATP molecule \n • Occurs in animal cells.
\ There are four main stages of active transport:
- Solute that is to be transported binds to a specific site on the transport protein found on the cytoplasmic side of the plasma membrane
- ATP molecule containing three phosphate groups, transfers one of the phosphates to the transport protein
- Binding of the phosphate group causes the transport protein to change its shape so that the solute can be released on the other side of the membrane
- Phosphate group detaches and the transport protein returns to its original shape • Makes the transport protein available to transport a new molecule
\ Ion Pumps and Membrane Potential • Voltage differences exist across the membrane of the cell • The membrane separates opposing charge • The cytoplasm is more negatively charged • The extracellular environment is more positively charged • **this creates a membrane potential • The negative cytoplasm will function to pull cations into the cell • Called an electrochemical gradient • Two forces drive the movement of the solute
- The concentration difference • Chemical force
- The charge difference • Electrical force
\ Electrogenic Pumps \n • Electrogenic pumps create charge separation \n • Gradient can be used to perform work and synthesize ATP \n • Energy is stored as a voltage difference
\ Co-transport \n • Substances that are actively transported across the membrane can be used to do work when they are left to flow down their concentration gradient \n • Co-transporters can couple the movement of one material down its concentration gradient (energy releasing) to the movement of another material up its concentration gradient (energy requiring) \n • Ex) sucrose transporter \n • Moves sucrose into the cell against the concentration gradient \n • H+ moves into the cell down its concentration gradient \n • The proton pump is responsible for maintaining the proton gradient
\ Bulk Transport • Larger molecules require bulk transport in order to be able to gain entry into the cell → occurs using vesicles
• Diffusion and osmosis are not sufficient for large molecules
• Includes:
- Exocytosis • Exports materials from the cell
- Endocytosis • Imports materials into the cell
• Three types: a. Pinocytosis b. Phagocytosis c. Receptor mediated endocytosis
Exocytosis:
Exocytosis is used to export large molecules such as sugars and proteins from the cell \n • A transport vesicle full of the molecule to be exported buds from the golgi apparatus and moves toward the plasma membrane \n • The vesicle then fuses with the plasma membrane, expelling the vesicular contents to the outside of the cell \n • The vesicular membrane is now a part of the plasma membrane \n • Ex) insulin is released from pancreatic cells via exocytosis \n • Neurotransmitters are also released via exocytosis
\ Endocytosis: \n • Endocytosis is essentially the reverse of exocytosis \n • The cell takes in substances with this process \n • A portion of the plasma membrane pinches inward collecting material from the outside of the cell and moving it inside of the cell \n • Material is now internal and within a membrane bound vesicle \n 1. Phagocytosis: \n • Cellular eating \n • Cell engulfs a particle by wrapping it with extensions called pseudopods \n • Particle is then packaged in a vacuole \n • Vacuole fuses with a lysosome \n • Digestive enzymes breakdown the contents of the vacuole
- Pinocytosis: • Referred to as cellular drinking • Droplets of fluid are taken up into tiny vesicles • Very non-specific • Any solutes that are dissolved in the extra-cellular fluid are also taken up
- Receptor-mediated endocytosis: • A highly specific process • Receptor proteins specific for a certain molecule are embedded within the plasma membrane • Receptor proteins pick up the specific molecule and the plasma membrane then pinches inward forming a coated pit Endocytosis
\
Lecture #9
Metabolism
• Metabolism manages all material and energy resources of the cell \n • Catabolic pathways break molecules/polymers into smaller fragments \n • Releases energy as ATP \n • Anabolic reactions build molecules and polymers from smaller starter materials \n • Requires energy \n • **all metabolic processes rely on enzymes in order to make them \n happen efficiently.
\ Bioenergetics
• Bioenergetics is the study of how energy flows
• Energy can be used to do work (rearrange matter)
• Chemical energy is energy stored within the bonds of molecules and can be released during chemical reactions
• Two laws govern thermodynamics:
- Energy can be transferred and transformed but it may not be created or destroyed
- Energy conversions are not 100% efficient and some energy is lost as heat each time • This heat is used to increase the entropy of the system • Entropy is the disorder of the system
Spontaneous processes occur without adding energy to the system \n • Non-spontaneous processes require an input of energy in order to proceed \n • The entropy of the universe is always increasing \n • Catabolic reactions release energy (increase entropy) \n • Anabolic reactions require energy (decrease entropy) \n • DG= DH – TDS \n • DG is the free energy available to do work \n • T is the temperature in Kelvins \n • DS is the change in entropy \n • DH is the change in enthalpy \n • DG
\ Enzymes \n • Enzymes are protein molecules that act to increase the rate of a chemical reaction \n • They are not consumed during a chemical reaction \n • They work to lower the activation energy of the reaction \n • They are specific to a particular chemical reaction \n • All chemical reactions have a particular activation energy that must be overcome in order for the reaction to proceed \n • Enzymes decrease this activation energy \n • Heat could be used to increase the rate of a reaction \n • Non-specific \n • All reactions would see an increase in rate
• Activation energy is needed to destabilize the bonds of the reactants so that they are readily able to break \n • Heat absorption provides the energy needed to reach this point \n • Once enough energy is absorbed the transition state is reached \n • The bonds will break and energy will be released \n • Activation energy provides the barrier to the forward movement of the reaction
\ Enzyme Specificity \n • Enzymes are proteins with unique three dimensional shape \n • Enzymes act on specific reactants called substrates \n • The substrate fits into a particular region on the enzyme called the active site which is a groove on the surface of the enzyme \n • The remainder of the protein maintains the active site shape \n • Enzymes act specifically on a particular target because the shape of the active site caters to the shape of the specific substrate
• Enzymes usually have the ending ‘ase’ and are named after their substrate
• Ex: the enzyme maltase catalyzes the breakdown of the disaccharide maltose
- The enzyme begins with an empty active site
- Substrate enters into the active site, attaching with weak bonds
• The induced fit hypothesis refers to the fact that the active site changes shape slightly in order to fit the substrate more closely
• Also functions to strain substrate bonds making them easier to break and places amino acids of the active site in proper position to catalyze the reaction
- The strained substrate bond reacts with water converting reactants into products
- The enzyme releases the newly formed products and is released unchanged from the reaction ready to catalyze a new reaction • A single enzyme may act on thousands or even millions of substrate molecules per one second
\ Optimal Enzyme Conditions
• The shape of an enzyme is critical to its function
• Enzyme environment may act to alter the enzyme’s shape and thus its function
• Certain parameters of an enzyme environment affect its function if they are not optimal:
• Ex) temperature and pH
- Temperature: • Optimal temperature for a particular enzyme maximizes contact between the enzymes active site and substrate molecules • Temperatures that are higher than optimal function to denature the enzyme rendering it non-functional • Ex) most human enzymes function best at 37oC
- pH: • Most human enzymes function best near neutral pH • At pH values higher and lower than 7, enzyme function is impaired
\ Enzymes and Cofactors \n • In order to function, most enzymes require non-protein molecules called cofactors \n • Cofactors can be either organic or inorganic \n • Inorganic cofactors are usually ions of zinc, copper or iron \n • Organic cofactors are called coenzymes \n • Vitamins that are essential dietary components often function as coenzymes
\ Enzyme Inhibitors
• A chemical that interferes with enzyme’s activity is called an inhibitor \n • Inhibitors bound to the enzyme by tight covalent bonds are irreversible \n • Inhibitors that are only weakly associated with the enzyme are reversible \n • Some inhibitors resemble the substrate of the enzyme: \n • These inhibitors compete with substrate for the enzymes active site and are called competitive inhibitors \n • They block substrate from entering the enzyme’s active site \n • Can be overcome by increasing the amount of substrate present so that it outcompetes the inhibitor \n • Other inhibitors do not resemble the substrate: \n • Non-competitive inhibitors bind to the enzyme in a different spot than the active site \n • Cause a change in the enzyme’s active site so that it no longer fits the substrate
\ Feedback Inhibition
• When a cell produces more product than it needs, the product can act to inhibit an enzyme that works to produce one of the substrates early on in the pathway \n • Feedback inhibition \n • Important metabolic regulator
\ Adenosine Tri-Phosphate • ATP is the energy currency of the cell • Used to perform three types of cellular work
- Chemical work: • Endergonic reactionsà Anabolism
- Transport Work: • Movement of materials against the concentration gradient
- Mechanical Work: • Muscle contraction • Flagellar rotation • Energy coupling pairs endergonic reactions with exergonic reactions • ATP hydrolysis is exergonic and can be used to do workà Releases 30.5kJ/mol of energyà -DG • Measured under standard conditions • Cell conditions are not standard and much more energy is available from the hydrolysis as a result
• Phosphate groups all carry negative charge \n • Repulsion of like charge causes energy release when the bonds are cleaved \n • Hydrolysis of ATP in artificial systems will be used to heat the surroundings \n • In the cell energy released from ATP hydrolysis is used to perform work \n • Small amounts of energy will be lost as heat \n • Ex) shivering \n • Enzymes work to couple exergonic reactions with reactions that require energy \n • Creates a net exergonic reaction \n • Often occurs by the transfer of a phosphate group to an intermediate \n • Raises the energy of the intermediate
• ATP is produced using energy that arises from catabolic reactions in the cell \n • ADP + Pià ATP \n • Endergonic \n • 10 million molecules of ATP are consumed and regenerated per second \n • Anabolic reactions require ATP \n • Catabolic reactions produce ATP
\ Equilibrium and Metabolism
• When a reaction reaches equilibrium it may no longer be used to perform work \n • DG=0 → Dead cell \n • Constant inward and outward movement of materials keeps the cell from ever reaching equilibrium \n • Catabolic reactions always occur via a series of reactions \n • Product pull forces secondary reactions \n • Consumes products of the first reaction preventing accumulation \n • Cellular catabolism: \n • Begins with: Glucose + O2 à high energy \n • Finishes with CO2 + H2 Oà low energy
\ Enzyme Regulation • Metabolic processes must be tightly regulated • Regulation may:
- Turn the production of an enzyme offà Stop transcription of the gene
- Regulate the activity of the enzyme post-production • Allosteric Regulation: • The function at one site of an enzyme is affected by the binding of a regulatory molecule at a different site • Enzymes often consist of multiple subunits which assemble into a functional unit • Regulatory molecule may be a(n): • Activator • Inhibitor
\ Allosteric Regulation \n • Allosteric enzymes have more than one subunit \n • Each subunit has its own active site \n • Each active site may be active or inactive \n • Allosteric regulators bind to an alternative site \n • Activators stabilize the active conformation \n • Inhibitors stabilize the inactive conformation \n • This change of shape is transmitted to all other subunits \n • Allows one regulatory molecule to affect all subunits \n • ATP is able to act as an allosteric inhibitor \n • ATP serves as a high energy signal \n • ATP binds to allosteric enzymes and turns off catabolism \n • AMP and Pi indicate that energy is being consumed \n • Bind to allosteric enzymes and turn on catabolism \n • Replenishes ATP supply
• Cooperativity \n • Amplifies the response of the enzyme to the substrate \n • Substrate binds to the active site of a multi-subunit enzyme \n • Triggers a change in shape of all subunits \n • Increases the catalytic activity of the enzyme \n • Hemoglobin is an allosteric protein that does not work as an enzyme \n • Exhibits cooperative behavior \n • Works to transport O2 from the lungs à tissuesà inside of the red blood cells \n • In the lung the binding of O2 to one hemoglobin subunit triggers a conformational change in the protein \n • Increases the affinity for O2 at all other binding sites \n • In the tissues O2 is low \n • O2 is released from one site and the affinity for O2 at all other sites is decreased \n • Causes O2 to unload
\n Feedback Inhibition \n • Accumulation of metabolic end products serve to inhibit enzyme activity early in \n the pathway \n • **behavior is observed with allosteric inhibitors \n • Ex: The amino acid isoleucine can slow its own production by behaving this way
\ Compartmentalization of the Cell \n • Cellular compartmentalization creates order in metabolic pathways \n • Enzyme teams may form complexes \n • Product of the first reaction will be the reactant for the next reaction
\
Lecture #11
\ Cellular Respiration \n • Organic molecules serve as sources of fuel for the cell \n • The origin of all chemical energy is the sun \n • Glucose can be directly catabolized to release energy \n • Polysaccharides, proteins, amino acids and lipids will first require some processing before they can be catabolized \n • Energy is converted between various forms \n • Some energy is always lost as heat \n • Chemicals are recycled \n • Oxidation of organic molecules \n • High energy molecules are broken into low energy molecules \n • Negative DG \n • Require enzymes because of high activation energy \n • Photosynthesis uses H 2O, CO 2 and sunlight to create O 2 and organic compounds
\ Aerobic Cellular Respiration vs Fermentation
• Aerobic Cellular Respiration: • C 6 H 12 O6 + 6O2 à 6CO2 + 6H 2 O + ATP + heat
• Requires O2
• Extremely efficient
• One mole of glucose release 2870kJ of energy
• Consists of glycolysis, the intermediate step, the TCA cycle and the electron transport chain
• Occurs as a mode of energy production in the absence of oxygen
• Ancient pathway
• Partial oxidation of organic carbon sources
• Used by some prokaryotes as their primary means of energy production
• Consists of glycolysis followed by either:
- Alcohol Fermentation: • Produces ethanol as an end product
- Lactic Acid Fermentation: • Produces lactic acid as an end product
\ Redox Reactions \n • Energy stored in organic molecules is released via electron transfers from one reactant to another \n • Loss of electrons from one substrate is oxidation \n • Gain of electrons from another substrate is reduction \n • Electron donor is the reducing agent \n • Electron acceptor is the oxidizing agent \n • **oxidation and reduction are always coupled \n • Called redox reactions
• Not all redox reactions will involve the complete transfer of electrons from one molecule to another \n • C-H covalent bonds are non-polar \n • Oxidation of methane (CH 4 ) forms CO2 \n • C-O covalent bonds are polar \n • The electrons are held more closely to the oxygen and the carbon has less of a share of the electron than when previously covalently bound to hydrogen \n • Carbon has therefore lost some of the electron and undergone an oxidation \n • Oxygen is a potent oxidizing agent \n • High positive reduction potential \n • Very electronegative \n • Great desire for electrons \n • Energy is required to strip an electron from a molecule \n • Transferring an electron to oxygen will release energy \n • This energy may be used to perform work in the cell
• Heavily reduced organic molecules have large potential energies \n • Lots of energy released from oxidation of these molecules during catabolism \n • Transfer of these electrons during catabolic reactions forms a lower energy source of electrons \n • Organic molecules don’t spontaneously decompose because of the high activation energy barrier \n • Enzymes are needed to reduce this barrier and allow the reaction to occur \n • Catabolism of an organic molecule in a single step would release too much energy too quickly → energy would not be harvested \n • Instead catabolism occurs in a series of steps each with its own enzyme \n • Energy is slowly and efficiently harvested this way \n • Electrons always travel together with a proton (H+ ) \n • Travel as a hydrogen atom
<<TEST<<
NAD+
• Nicotinamide Adenine Dinucleotide
• An electron carrier that temporarily holds electrons that are removed from organic molecules during catabolism
• Dehydrogenase enzymes strip electrons from glucose
• Glucose + NAD+ à 6CO2 + NADH
• Holds 2 electrons
• Undergoes a reduction
• Recyclable
• Transfers the electrons to O2 oxidizing NADH to NAD+
\ NAD +
• NADH produced during glucose oxidation can be used to perform cellular work
• NADHà O2 + NAD + + H2O
• Energy is released and harvested in the electron transport chain
• Protein and associated molecules built into the inner mitochondrial membrane of eukaryotes and the plasma membrane of bacteria
• Capable of aerobic respiration
• Electrons move from the high energy end of the chain to the low energy end of the chain
• Each of the electron transfers is exergonic
• Small steps prevent an inefficient release all at once
• A series of redox reactions
• Each electron carrier is more electronegative than the former
• Draws electrons along the chain • Glucoseà (NAD + ) à NADH à (O2 ) à H 2 O
ATP Production
- Substrate Level Phosphorylation: • Pi from a substrate molecule is removed and attached to ADP forming ATP • Produces small quantities of ATP in glycolysis and the TCA cycle
- Oxidative Phosphorylation: • Phosphate group originates as Pi in the cytoplasm • Transfer of electrons to O2 provides energy to drive phosphorylation of ADP • Occurs in the electron transport chain • Electrons originate on NADH and FADH 2 • Transferred to O2 • Produces large quantities of ATP • ~90% of energy release during cell respiration • ~32 ATP are available in the glucose structure
Glycolysis • Glycolysis occurs in the cytosol of both prokaryotes and eukaryotes • Begins the process of glucose oxidation (catabolism/breakdown) • Glucoseà 2 x pyruvate (3 carbon molecules) • Consists of:
- Energy requiring phase • Requires ATP (2 in total)
- Energy payoff phase • Produces ATP (4 in total)
Glycolysis • Net production in glycolysis: • 2 ATP produced by substrate level phosphorylation • 2 NADH • **25% of the available energy in glucose is released during glycolysis • No CO2 is released • Occurs in ten different steps • Each step requires a unique enzyme
\ The Intermediate Step
• 2 pyruvate molecules produced during glycolysis must be groomed for entry into the citric acid cycle
• Pyruvate is first translocated from the cytoplasm into the mitochondrial matrix
• **in prokaryotes the intermediate step takes place in the cytoplasm
• In the mitochondrial matrix each high energy pyruvate molecule is converted into acetyl-coA
• A multi-subunit enzyme called the pyruvate dehydrogenase complex will:
• Remove COO- from pyruvate as CO2
• CH3 COO- is produced from the remaining 2 carbon structure
• Produces one NADH
• Coenzyme A is added to the CH3 COO- via a
sulfur bond
• Forms acetyl coA
• Starting material for the TCA cycle
• Of higher potential energy
The TCA Cycle • Occurs in the mitochondrial matrix • Cytoplasm of prokaryotes • Also called the citric acid cycle and the Kreb’s cycle • The citric acid cycle serves to oxidize acetyl coA completely • Forms 2 CO2 from the remaining 2 carbon of glucose • Occurs via 8 steps each with its own enzyme • In the process of oxidizing 1 acetyl coA: • 1 GTP is formed via substrate level phosphorylation • 3 NADH are formed • 1 FADH2 is formed • ** all FADH2 and NADH produced will be starting material for the electron transport chain
The TCA Cycle \n • Acetyl coA entering into the citric acid cycle combines with a four \n carbon structure called oxaloacetate (OAA) \n • Forms a six carbon structure called citrate \n • 7 additional steps oxidize citrate \n back to the four carbon OAA \n • A ‘cycle’ \n • Six carbonà four carbon \n • 2 carbon are lost as 2 CO 2
\ Electron Transport Chain \n • Only 4 ATP equivalents have been produced so far \n • Substrate level phosphorylation \n • NADH and FADH 2 hold most of the energy that originated in glucose \n • Electron shuttles that need to be oxidized and recycled \n • The inner mitochondrial membrane is home to the electron transport chain \n • There are 1000s of copies of this chain in the membrane \n • Cristae folds increase the surface area making the high copy number possible \n • Each ETC has 4 complexes and an ATP synthase enzyme \n • I/II/III/IV \n • Consist mainly of protein \n • Multi-subunit structures \n • Each complex has tightly bound prosthetic groups \n • Needed for function
\ Electron Transport Chain \n • As electrons move down the chain there is a \n decrease in free energy \n • Carriers alternate between oxidized and reduced \n forms \n • NADH is first oxidized to NAD+ transferring its \n electrons to complex I \n • FMN is a flavoprotein that accepts these \n electrons \n • The electrons are then moved from the \n flavoprotein to Fe-S \n • Fe-S transfers the electrons to ubiquinone \n • A non-protein molecule that is hydrophobic \n and mobile through the membrane interior \n • Ubiquinone is also called coenzyme Q \n • Ubiquinone carries electrons to complex II
\ Electron Transport Chain \n • Other electron carriers in the chain are cytochromes \n • Many different varieties \n • Iron is included in the structure \n • Undergoes oxidation and reduction \n • Final carrier is cytochrome a3 \n • Transfers electrons to O2 \n • O2 also picks up two H+ from the mitochondrial matrix forming water \n • FADH 2 enters the chain at complex II \n • Contributes 2 electrons like NADH but enters the chain later \n • Worth less ATP as a result
\ Chemiosmosis • Chemiosmosis: energy coupling • ATP synthase is a fifth protein complex at the end of the ETC • Forms ATP from ADP and Pi • An H + pump that can work to consume ATP and form an H + gradient • Can also produce ATP by dissipating an H + gradient • H + gradient is a power source that is coupled to ATP synthesis • ATP synthase is a multi-subunit structure • Four different components • Each subunit consists of many polypeptide chains • H + enters binding sites inside the rotor portion of the structure causing the structure to spin • Release of energy is used to form a high energy covalent bond between ADP and Pi • The H+ gradient is established by the electron transport chain • As electrons are passed from carrier to carrier the release of energy is used to push H + into the inter-membranous space
Chemiosmosis • H+ is pumped into the inter-membranous space at complexes I, III and IV • Complex II is not a transmembrane protein and cannot pump H + across • H + gradient provides a proton motive force that can be used to produce ATP • Energy stored in the gradient can be used to do cellular work • ATP synthesis • Each NADH is worth 2.5 ATP – 10H + are pumped due to movement of 2 electrons from NADH • 4H += 1ATP • Each FADH2 is worth 1.5 ATP • Enters the chain at complex II • Does not result in proton movements across complex I • 4H+ less than NADH
Efficiency • Under maximum efficiency and without any competing reactions 32 ATP could be produced from one glucose • 4 ATP equivalents from substrate level phosphorylation • 28 ATP from oxidative phosphorylation • **under normal circumstances energy from the proton gradient is used to drive cellular transport • Movement of pyruvate from the cytoplasm into the mitochondria • Movement of ATP into the cytosol following production in the ETC • Efficiency: • 32 ATP x 30.5kJ/mol = 976kJ/mol • 2870 kJ/mol available in glucose • (976/2870)*100 = 34% harvested • Remainder of the energy is lost as heat
Fermentation & Anaerobic Respiration • Oxidation of organic fuel in order to produce energy in the absence of oxygen • Two different processes:
- Anaerobic respiration: • Uses the ETC • Alternative electron acceptors are usedà no O2 • Also generates a proton motive force • Some prokaryotes living in anaerobic environments will do this • Less energy is available • Nothing compares to oxygen in terms of electron affinity • Ex) SO42- is an electron acceptor (replaces O2 ) • H2 S is the reduced product (replaces H2 O)
- Fermentation: • No ETC • Pyruvate is converted to an end product • Occurs because NAD+ needs to be regenerated in order to keep glycolysis going • 2 ATP from glycolysis = only energy produced
Alcohol Fermentation
- Alcohol Fermentation: • Bacteria and yeast do this • Bubbles are seen in the reaction mixture from CO2 i. Begins with Glycolysis • Glucose is split into pyruvate • 2 ATP and 2 NADH are produced • Occurs in the cytoplasm of prokaryotes and eukaryotes ii. Pyruvate is then converted into acetaldehyde and CO2 iii. Acetaldehyde is then converted into ethanol • NADH is also oxidized back to NAD+
Lactic Acid Fermentation
- Lactic Acid Fermentation:
• Used by fungi and bacteria in the production of cheese and yogurt
• Human skeletal muscle also switches to lactic acid fermentation when O2 is limited
• Lactic acid is sent to the liver where it is converted to pyruvate
• Enters into the intermediate step for normal catabolic processing i. Begins with glycolysis ii. Pyruvate is then converted into lactic acid
• NADH is also oxidized back to NAD+
Fermentation • Obligate anaerobes are incapable of tolerating O2 • Killed in the presence of O 2 • May only use fermentation or anaerobic respiration for energy production • Facultative anaerobes are capable of using respiration or fermentation • If O2 is present aerobic respiration will be used • Much greater ATP production • If O2 is not present anaerobic metabolism will be used in order to keep the cell going
Versatility of Catabolism • Free glucose is not common • Disaccharides, proteins and lipids are more commonly consumed as energy • These structures must be processed but may enter into glycolysis or the TCA cycle as intermediates • Used to produce ATP • Glycogen may be catabolized in order to release free glucose for cell respiration • Proteins may be cleaved into free amino acids • Amino acids can be used for anabolic reactions • Protein synthesis • Amino acids may also be used for ATP production via catabolism • Must first undergo deamination to remove the amino group from the carbon skeleton • Amino group is used to produce urea and other waste products such as ammonia
Versatility of Catabolism • Fats can be cleaved into fatty acids and glycerol • Glycerol is converted into a three carbon glycolysis intermediate • Fatty acids are consumed in a process called beta- oxidation • Converts the fatty acid into many acetyl coA molecules • These are worth large quantities of NADH/FADH2 /GTP in the TCA cycle • Fats store large quantities of energy • 1 gram of fat stores twice as much energy as one gram of sugar • Also why it is more difficult to loose weight
Biosynthesis • Not all food that enters the body is completely catabolized • Some must be used in order to build molecules required by the body • These catabolites from food serve as carbon skeletons to build larger organic molecules in the cell • Glycolysis and TCA cycle intermediates can be used for building reactions • Glucose can be synthesized beginning with pyruvate • Fatty acids can be synthesized from acetyl coA • All anabolic reactions consume ATP • This ATP can later be released when the molecule is catabolized
\
Lecture #12
Photosynthesis • Solar power is the origin of all energy • Chloroplasts are responsible for the capture of light energy • Photosynthesis • Organic material is needed for cell activities • Carbohydrates, proteins, lipids and nucleic acid • Carbon may originate from:
- Carbon dioxide • Autotrophic nutrition • Producers • Plants • Need water and minerals from soil and carbon dioxide from the atmosphere
- Organic sources: • Heterotrophic nutritionà Use preformed organic carbon • Live on compounds produced by other organisms • Cannot make their own carbon compounds • Consumers of the biosphere • Obtain carbon through decomposition (fungi, bacteria) or direct consumption • Photoautotrophs: use light energy to synthesize organic compounds • Algae, protists, cyanobacteria
\
Photosynthesis • Photosynthesis is a series of redox reactions • Movement of electrons from water to carbon dioxide forming sugar • The potential energy of the electrons are increased along the way • Endergonic reaction • Required energy comes from the sun • Two stages of photosynthesis:
- Light Reactions
- Dark Reactions • The Calvin Cycle • Photosynthesis began in photosynthetic bacteria • Highly folded plasma membrane • Same structure of the chloroplast membrane • **endosymbiotic theory • Chloroplasts were photosynthetic bacteria
Chloroplasts • Chloroplasts are the site of photosynthesis • 30-40 chloroplasts/cell • All green plants have chloroplasts concentrated in the leaves • 1mm3 =500,000 chloroplasts • Accumulate in the mesophyll • Middle tissue of the leaf • CO2 enters and O2 exits via pores called stomata • H 2 O needed is absorbed by the roots • Moves to the leaf via veins called xylem • Veins export sugar to the roots and other non- photosynthetic structures
Chloroplasts • Chloroplasts have a double membrane • The interior of the chloroplast is filled with thick fluid called stroma • Thylakoids are stacked in the chloroplast interiorà stacks of thylakoid are grana • Membrane rich, fluid filled structures • Chlorophyll pigment is concentrated in the thylakoid membranes • Provides characteristic green color and absorbs light energy • **analogous to the internal membranes of photosynthetic prokaryotes • Light absorption by chlorophyll drives photosynthetic reactions • 6CO2 + 12H 2 O + light energy à C 6 H12 O6 + 6O2 + 6H 2 O • Glucose is assembled from 2 three carbon intermediates • Actual photosynthetic products
\ Oxygen Gas is Produced in Plants by the Splitting of Sugar
• The bubbles in the adjacent picture are O2 gas generated during photosynthesis
• Photosynthesis uses water as a reactant but also generates water as a product
• Initially thought that the O2 generated from photosynthesis comes from CO2
• Experimentation using labeled oxygen revealed that the O2 produced actually originates in water molecules • Two water molecules are used to produce every one molecule of O2 • 2H2O → O2
\ Photosynthesis • Two stages:
- Light reactions: • Photo stage • Solar energy is converted into ATP (chemical energy) • Takes place in the thylakoid membranes • Water is split into 2 protons and 2 electrons, releasing oxygen • Light absorbed by chlorophyll is used to power the transfer of electrons from water to NADP+ • ATP is also produced by chemiosmosis • Photophosphorylation • No sugar is produced
- Dark Reactions (The Calvin Cycle): • Carbon fixation step • CO2 + ATP + NADPH à C6 H12 O6 • Light and dark reactions both take place in daylight • Dark reactions do not require light**
The Light Reactions • Sunlight is electromagnetic radiation • Travels in waves that have electrical and magnetic properties • Wavelength is the distance between crests • Visible light is from 380nm to 750nm in the electromagnetic spectrum • Seen as colors by the human eye • Behaves as though it consists of discrete particles of light called photons • Each has a fixed quantity of energy • Wavelength is inversely related to energy • Shorter wavelength, higher energy • Visible light reaches the Earth and drives photosynthesis
Photosynthetic Pigments • When light interacts with matter it can be: • Reflected, absorbed or transmitted • Pigments serve to absorb light energy • The color that we observe is of a wavelength that the pigment cannot absorb • We can create an absorption spectrum which is characteristic of the pigment • **light must be absorbed if it can be used to perform work • 3 pigments in chloroplasts:
- Chlorophyll a: participates directly in the light reactions
- Chlorophyll b: an accessory pigment
- Carotenoids: group of accessory pigments • **green light is the least effective color because it is reflected • Cannot be absorbed/used to perform work
Photosynthetic Pigments • Accessory pigments allow absorption of an increased number of wavelengths • In addition to the capabilities of chlorophyll a • Prevents waste • Small structural differences between pigments allow for increased absorption • Chlorophyll a: blue green • Chlorophyll b: olive green • Carotenoids: yellow/orange • Serve as photo-protectants • Absorb and dissipate energy • Energy would have otherwise damaged other pigments or reacted with oxygen • Anti-oxidant properties
\ Photosynthetic Pigments \n • Pigments absorb light energy exciting an electron to another orbital \n • Increased potential energy \n • Origin=ground state \n • Absorption of a photon boosts the electron to an excited higher energy state \n • The absorbed photon must be of an energy value exactly equal to the difference in \n energy between the origin and the excited energy orbital \n • Every pigment has a unique absorption spectrum \n • Excited state is very unstable \n • The electron drops back down to the \n ground state very quickly \n • Excess energy is released as heat \n • Energy may also be released as light \n • Fluorescence
\ Photosystems \n • Chlorophyll behaves much differently in chloroplast membranes \n • Arranged into a photosystem \n • Photosystem is a reaction center complex \n • Consists of a pair of chlorophyll a molecules and light harvesting complexes \n • Light harvesting complexes have pigments \n a, b, and carotenoids bound to different proteins \n • Increased quantity and variety allows light energy \n harvesting to occur over a greater surface area and \n an increased spectrum of absorption \n • As compared to a single pigment \n • Light harvesting complex is an antenna \n for the reaction center complex \n • Photon absorbed by a pigment is \n transferred from pigment to pigment \n until the reaction center complex is reached \n • Reaction center complex contains a \n molecule capable of undergoing a reduction
\ Photosystems \n • Chlorphyll a absorbs light energy \n • Boosts an electron to a higher energy state \n • The electron is transferred to a primary electron acceptor \n • Redox reaction \n • Converts light energy to chemical energy \n • Thylakoids have photosystem II and \n photosystem I \n • PSII functions first: called P680 \n • PSI functions second: called P700 \n • **P680 and P780 are essentially identical but \n associate with different proteins altering their \n electron distribution \n • Changes the wavelength that each is \n capable of absorbing
\ Electron Flow and ATP/NADPH Synthesis \n 1. Light energy and pigments in photosystem II \n • Electrons move from the ground state to a high energy state \n • As the electron falls back down the energy release stimulates excitation of a \n nearby electron \n • This continues until the chlorophyll a pair of P680 is reached \n • P680 is excited (P680à P680*) and transfers electrons to the primary electron \n acceptor \n • H2 O is split into 2H +, 2e- and O \n • Occurs with the assistance \n of an enzyme \n • O combines with \n another to form O2 \n • Electrons are used to \n replace the donated pair \n from P680
\ Electron Flow and ATP/NADPH Synthesis
• The electrons that were excited from P680 to P680* are then \n transferred via an electron transport chain \n • Uses plastoquinone, cytochrome complex and plastocyanin \n • Fall of electrons is exergonic \n • H+ are pumped into the thylakoid lumen \n • ATP is synthesized \n
Electron Flow and ATP/NADPH Synthesis \n • The proton gradient that is created is used for chemiosmosis \n • Light harvesting complex also transfers more light to photosystem I \n exciting the electrons of the chlorophyll a pair of P700 forming P700* \n • P700* passes the electrons to an electron acceptor \n • The electrons of P700 are replaced with the electrons moving from PSII down \n the electron transport chain \n • Electrons from P700* move down a second electron transport chain \n • Uses ferrodoxin \n • Transfers electrons to NADP+ forming NADPH \n • Reducing power for the dark reactions \n • NADPH is more energized than H2 O \n • Electron origin \n • More energy is released
\ Cyclic Electron Flow and ATP Synthesis • Cyclic electron flow only uses PSI • Electrons move from ferrodoxin to cytochrome complex to P700 in PSI • No NADPH released • No O2 released • ATP is produced
The Calvin Cycle • The dark reactions • NADPH + ATP + CO2 à Glucose • Anabolic cycleà TCA=catabolic • Requires energy in the form of ATP • Requires a source of reducing powerà NADPH • Doesn’t directly produce glucose • Produces the three carbon glyceraldehyde 3 phosphate (G3P) • For every three turns of the cycle 3 CO2 are fixed and one G3P is made
The Calvin Cycle • 3 phases:
- Phase I: carbon fixation • Ribulose bis-phosphate (RuBP) + CO2 • Catalyzed by an enzyme called RUBISCO (most abundant protein on Earth) forms a six carbon intermediate • Very unstableà splits into 2 x 3-phosphoglyerate
- Phase II: reduction • 3 phosphoglycerate accepts a phosphate from ATP forming 1,3 bis-phosphoglycerate • 1,3 bis-phosphoglycerate accepts 2 electrons from NADPH and releases one phosphate group • Forms glyceraldehyde 3-phosphate • Has more potential energy • Also formed in glycolysis • **for every three turns of the Calvin Cycle: • 6 G3P will be made • Only 1/6 is a net gain • 15 carbon from RUBISCO have been consumed in these three cycles • Must be recycled The Calvin Cycle • 3 phases:
- Phase III: ribulose bis-phosphate regeneration: • 5x G3P are rearranged into 3x RuBP • Requires 3 ATP • Net synthesis of one G3P • Requires 9 ATP and 6 NADPH • ATP and NADPH are products of the light reactions
Photorespiration • Photorespiration • On hot days the stomata are closedà prevents water loss • Simultaneously decreases CO2 intake and therefore photosynthetic yield • Increased O2 release from the light reactions • Photorespirationà C3 plants make G3P first • Examples: rice, wheat • RUBISCO can bind to O2 as well as CO2 • A 2 carbon compound is produced • Leaves the chloroplast • Perioxisomes and mitochondria rearrange the compound and CO2 is released • Consumes O2 and releases CO2 • Does not generate ATPà consumes ATP • Does not produce sugar • Decreases photosynthesis by using carbon fixation material • Releases CO2 • Drains fixed carbon and decreases plant production
Alternatives to Carbon Fixation • C4 plants • Produce a 4 carbon intermediate first • Used in many plants • **unique leaf anatomy is needed • Two cells:
- Bundle Sheath Cells: • Arranged around the veins of the leaf
- Mesophyll Cells: • Loosely arranged between the bundle sheath cells and the leaf surface
Alternatives to Carbon Fixation • CO2 accumulates in the mesophyll cell • An enzyme called PEP carboxylase combines PEP with CO2 forming a 4 carbon intermediate called oxaloacetate • PEP carboxylase has a higher CO2 affinity than Rubisco • Does not have O2 affinity allowing for efficient CO2 fixation in hot, dry climates • Mesophyll exports the 4 carbon product to the bundle sheath cells via plasmodesmata • In the bundle sheath cell the 4 carbon intermediate looses CO2 à used as a substrate for Rubisco • The remaining 3 carbon molecule pyruvate is converted back to PEP at a cost of one ATP molecule • The price required to increased the CO2 concentration in the bundle sheath cell • High CO2 concentration keeps Rubisco from binding to O2 minimizing photorespiration
Alternatives to Carbon Fixation • CAM plants: • Photosynthesis adapts to hot climates • Ex: cacti • Stomata are open at night and closed during the day • Opposite of standard behavior • **helps to conserve water • Prevents CO2 from entering • CO2 is taken into the leaf at night and incorporated into organic acids • Mesophyll cells store acids that are made at night in vacuoles until morning • In the daylight solar energy is used to produce ATP and NADPH • Organic acids release CO2 • Used to produce sugar in the chloroplast
Fate of Photosynthetic Products • Photosynthesis produces chemical energy and carbon skeletons • Used to make all major organic molecules of plant cells • 50% is consumed as fuel for cellular respiration • Some is lost to photorespiration • Leaves are autotrophic • The remaining plant structures receive these organic carbon structures via the veins • Usually as sucrose • Delivered to other cells for cell respiration and anabolic reactions • Glucose is stored as starchà main ingredient of the plant cell wall • Excess sugar is stored in the roots, seeds and fruits • Energy/carbon supply for heterotrophs • One metric ton=1000kg • Plants produce 160 billion metric tons of carbohydrates per year • O2 production
\
Lecture #13
Cell Division \n • Most cell division serves to divide one parent cell into two identical \n daughter cells \n • Mitosis in eukaryotes \n • Binary fission in prokaryotes \n • **exception: meiosis \n • Parent cell divides into 4 daughter cells \n • 2 copies of 2 genetically non-identical cells \n • Regardless of the type of cell division this is always a highly accurate, \n highly complex process
\ Cell Division • Genome: total cellular DNA content • Prokaryotes have one single circular DNA molecule • Eukaryotes have many linear DNA molecules • Humans have 2 meters of genomic DNA organized onto 46 chromosomes • 250,000x cell diameter • **must be able to replicate this DNA but also must be able to separate into two equal daughter cells • Chromosomes are DNA and associated proteins in a complex • Proteins allow the maintenance of cell structure and also assist with DNA function • Allow the DNA structure to be kept highly ordered • Consist of 100s-1000s of genes • Specify an individual’s traits • DNA + protein = chromatin • Exist in varied degrees of condensation
Cell Types • All human cells fit into one of two categories:
- Somatic cells: • All body cells except sperm and egg (sex cells) • Have 46 chromosomes in the nucleus • 23 pairs • One version from mom and one version from dad
- Gametes: • Sex cells used for reproduction • Sperm and egg • Contain half the number of chromosomes • 23 • Allow restoration of the diploid number (46) following fertilization • *Number of chromosomes vary depending on the species • Humans: 46 • Dogs: 78
\ Chromosomes
• Chromosomes are long and thin when not actively undergoing divisionà chromatin
• Chromatin condenses in preparation for cell division
• Form a densely coiled, folded structure that’s shorter and easily visible with a microscope
• Duplicated in order to provide enough DNA for two cells following division
• Each chromosome is a duplicate
• Photocopy of the original joined together with the original
• Sister chromatids attached to one another by cohesins
• Each chromatid also has a centromere used for attachment
• Sister chromatids will separate from one another during cell division
• No longer called sister chromatids once they separateà individual chromosomes
• Each cell that results from the division will be identical to the parent cell
\ Mitosis vs Meiosis • Mitosis is used for growth and repair • Produce new cells and replace damaged cells • Consist of two phases:
- Division of genetic material
- Division of cytoplasmic contents • Meiosis is a variation of mitotic division • Used to produce gametesà Sperm and egg • Produces non-identical cells • Contributes to genetic variation • Produces cells with ½ the diploid number (2n=46) of chromosomes • Haploid (n=23) • Only one set of chromosomes • Occurs in the gonadsà testes and ovaries • Allows fertilization to restore the diploid number • Sperm (n=23) + egg (n=23) à 2n=46
Mitosis • The cell cycle consists of:
- Interphase • Does not involve cell division • Accounts for 90% of the cell cycle • Duplication of chromosomes and increase in cell size and number of organelles • Three phases: i. G1 : 4-6 hours (varies in length) ii. S: chromosomal duplication (10-12 hours) iii. G2 : 4-6 hours • **all three phases involve protein and organelle synthesis
- Mitosis • Actual division of chromosomes • Accounts for 10% of the cell cycle • Less than one hour to completion
Interphase
- Interphase: • A period of intense metabolic activity • Proteins and new organelles are produced, the cell increases in size • Chromosomes are also duplicated during interphase • A cell will spend approximately 90% of the cell cycle in interphase • Interphase can be divided into three sub-phases all of which include cell growth • G1 , S and G2 phase • The cell grows, increasing in size during G 1 of interphase • S phase stands for DNA Synthesis as it is during this phase that the DNA is duplicated via DNA replication, the cell also continues to grow during S phase • At the start of S phase each chromosome is single, at the end of S phase each chromosome consists of 2 sister chromatids • The cell continues to grow and prepares for cell division during G 2 phase
\
- The Mitotic Phase: • Period of the cell cycle where cell division actually takes place • The cell spends only 10% of the cell cycle in this phase • There are two sub-phases of mitosis: i. Mitosis ii. Cytokinesis: begins before the termination of mitosis • During mitosis the nucleus and all of its contents, including duplicated chromosomes divideà forms two daughter nuclei • During cytokinesis the cytoplasm and all of its contents, nuclei and organelles, divide into two • End result of mitosis and cytokinesis: two identical daughter cells each with a nucleus, cytoplasm and a plasma membrane • Each daughter cell now enters G1 of interphase and begins cell division • Mitosis is extremely organized and accurate • Errors occur approximately once per 100,000 divisions
\
Mitosis
• A human cell is always within a phase of division
• After the completion of mitosis daughter cells enter into G1
• Continue to take part in normal cellular activities
• Cells that are not destined to divide again enter into G 0 à Dead end
• Ex: neurons
• Mitosis consists of five phases:
- Prophase
- Prometaphase
- Metaphase
- Anaphase
- Telophase • Cytokinesis overlaps with telophase • Symbolizes the completion of mitosis
\ Mitosis \n • The mitotic spindle \n • Forms in the cytoplasm during prophase \n • Consist of fibers made of microtubules and associated proteins \n • The cytoskeleton partially dismembers in order to provide material for the \n spindle to form \n • Microtubules polymerize by increasing tubulin in order to elongate \n • Microtubules depolymerize by removing tubulin in order to shorten \n • The Centrosome is the site of spindle \n assembly \n • Consists of a pair of centrioles \n • Plant cells do not have centrioles \n • Non-essential for the cell division \n process
\ Prophase \n • During prophase chromatin becomes coiled forming visible \n chromosomes \n • The nuclear membrane and the nucleolus disappear \n • Animal cells have centrosomes duplicate during interphase \n • They begin side by side near the nucleus \n • Move to opposite poles of the cell during prophase \n • Spindle microtubules will grow out from them \n • Symbolizes the end of pro-metaphase when the two centrosomes \n are at opposite poles of the cell \n • The aster is an array of short microtubules \n that project out from each centrosome \n • The spindle includes: \n i. Asters \n ii. Spindle microtubules \n iii. Centrosomes
\ Prophase \n • Each sister chromatid contains a kinetochore \n • Network of proteins associated with the DNA of the centromere \n • Site of attachment of spindle microtubules \n • Called kinetochore microtubules \n • # varies according to species \n • Attachment of kinetochore to the microtubule \n causes the pull of sister chromatids to each \n pole of the cell
\ Metaphase \n • During metaphase the spindle apparatus is fully formed and located at \n opposite poles of the cell \n • Duplicated chromosomes align down the center of the cell at what is called \n the metaphase plate \n • Centromeres are located midway between the spindle’s two poles \n • Asters have elongated and contacted the plasma membrane \n • Non-kinetochore microtubules have elongated and \n contacted non-kinetochore microtubules originating \n at the other pole of the cell
\ Anaphase \n • During anaphase centromeres separate from one another pulling sister \n chromatids apart \n • Separase cleaves cohesins that hold sister chromatids together \n • Chromatids move to opposite poles of the cell \n • Region of overlap between non-kinetochore microtubules is reduced \n • Motor proteins walk microtubules away from one anotherà consumes ATP \n • Elongates the cell by pushing spindle poles apart from one another \n • Microtubules continue to lengthen \n simultaneously by the addition of tubulin \n at overlapping ends \n • Anaphase is complete when chromosome \n duplicates have reached opposite poles of \n the cell
\ Telophase & Cytokinesis \n • Telophase is essentially the reverse of prophase \n • The nuclear membrane and the nucleolus reform \n • Chromosomes unravel forming chromatin once again \n • Microtubules depolymerize \n • Disassembly of the spindle apparatus \n • Mitosis is now complete as the identical daughter nuclei have now divided \n • Cytokinesis is the division of the cytoplasm and its contents \n • Occurs at the same time as telophase \n • A cleavage furrow forms pinching the cell into two
\ Cytokinesis in Animal Cells \n • Cytokinesis in animal cells occurs via cleavage furrow formation \n • A shallow groove in the surface of the plasma membrane \n • On the cytoplasmic side of the membrane actin forms a contractile ring \n • Interaction of actin and myosin cause the ring to contract \n • The cleavage furrow deepens until the cell pinches off \n into two complete daughter cells
\ Cytokinesis in Plant Cells \n • Cell wall inhibits the ability of cleavage \n furrow formation \n • Vesicles from the golgi carry cell wall \n material \n • Move along microtubules to the middle of \n the cell during telophase \n • Vesicles fuse here forming a cell plate \n • The surrounding membrane eventually fuses \n with the plasma membrane \n • Forms two complete daughter cells
\ Binary Fission \n • Used as a mode of cell division in prokaryotes \n • Cell grows to double the original size \n • Divides into two genetically identical daughter \n cells that are the same size as the parent cell \n • Smaller chromosome but still 500x the length of \n the bacterial cell \n • Must be highly folded and coiled as a consequence \n • Chromosome replication begins at the origin of \n replication (ori) \n • 2 origins as division proceeds outward \n • As the newly formed chromosome moves toward one \n end of the cell the cell simultaneously increases in length \n • Plasma membrane pinches inward and two identical \n daughter cells are created \n • Protein assistance is required to move chromosomes to opposite \n poles of the cell
\ Evolution of Mitosis \n • Mitosis followed a simpler prokaryotic mode of reproduction \n • Proteins used in bacterial binary fission are related to proteins used in \n mitosis \n • Single celled eukaryotes use mitosis \n • Ex: Dinoflagellates \n • Nuclear membrane remains intact \n • **intermediate between binary fission and mitosis
\
Lecture #14
Homologous Pairs \n • Chromosomes are present in homologous pairs \n • There are 46 chromosomes in a human somatic cell \n • Each chromosome in the somatic cell has a twin chromosome that is identical \n in length and centromere position \n • If you stain the chromosomes in a cell with dye, the matching chromosomes \n of a homologous pair display identical patterns that consist of stripes \n • In total a cell has 23 chromosome pairs \n • The two chromosomes of a pair are called a homologous pair because they \n both contain genes that code for the same trait such as hair color or eye color \n • The location on a chromosome that a particular gene \n is found is called the locus \n • Homologous pairs will contain a gene encoding the \n same trait at the same locus \n • The homologous pairs can encode a different \n version of the same gene
\ Karyotypes • A karyotype is a visual display of homologous chromosomal pairs • A display of an individual’s magnified chromosomes beginning with the longest and arranged in homologous pairs • The chromosomes viewed in a karyotype are condensed and doubled, and are thus located in metaphase of mitosis • White blood cells, specifically lymphocytes are used to prepare a karyotype • These lymphocytes are chemically treated such that they begin mitosis • Following days of growth a second chemical is added to arrest the cells in metaphase of mitosis
Homologous Pairs • An exception to the homologous pair rule in the human cell are the sex chromosomes • Human females will have a homologous pair of sex chromosomes present as XX • Human males will not have a homologous pair of sex chromosomes but instead have XY • Most genes carried on the X chromosome do not have a counterpart on the Y chromosome • The Y chromosome is also much smaller than the X chromosome and will carry a few genes of its own that cannot be found on the X chromosome • One chromosome from each homologous pair and one sex chromosome is inherited from our mother and the other sex chromosome and the remaining member of the homologous pair is inherited from our father • Somatic cells have 23 chromosome pairs • 1-22 are called autosomes • 23 is the sex chromosome pair
\ Gametes
• In order for our lifecycle to continue as we know it, a sexually reproducing organism must have two chromosome sets
• One inherited from each parent
• A cell with two sets of chromosomes is called a diploid cell
• The total number of chromosomes in a cell is called the diploid number abbreviated 2n
• The human diploid number is 46 therefore 2n= 46
• Humans are referred to as diploid organisms because the only cells in the human body that are not diploid are the gametes (sperm or egg)
• Each gamete has only a single chromosome set
• A cell with only one set of chromosomes is referred to as haploid and the haploid number is n= 23 in humans
\ Gametes Have One Set of Chromosomes
• Fertilization is the fusion of two haploid gametes
• One sperm and one egg
• Results in a zygote that now has two chromosome sets → diploid
• If gametes were not haploid, fertilization would produce a tetraploid organism
• The zygote now has 46 chromosomes and divides by mitosis, eventually producing a mature organism that will produce its own haploid gametes • The human life cycle is thus an alteration of diploid and haploid stages • The haploid gametes are produced by a process called meiosis • Occurs in the ovaries and the testes of adults • Meiosis reduces the chromosome number by half • 2nà n • 46à23 • Mitosis produces two daughter cells with the same chromosome number as the parent cell Gametes Have One Set of Chromosomes
\ Diploid vs Haploid
• Diploid number= 6
• 2n=6
• Haploid number =3
• n=3
• Following completion of S phase of interphase the cell has duplicated chromosomes
• 12 in total
• Sister chromatids attached at the centromere Different Sexual Life Cycles
\ Different sexual life cycles
\ • Three different sexual life cycles exist:
- Human/animal life cycle: • Meiosis takes place in germ cells • Produces gametes • Fertilization produces a diploid zygote • Divides by mitosis to produce a multi-cellular organism
- Plant/algae life cycle: • Contain haploid and diploid multi-cellular stages • Multicellular, 2n sporophyte produces n spores by meiosis • n spore divides by mitosis to produce a multicellular, n phase called a gametophyte • The gametophyte phase produces n gametes by mitosis • Gametes fuse producing a 2n zygote • Zygote divides by mitosis to produce a multicellular sporophyte phase
\ 3. Fungal life cycle:
• Gametes fuse producing a 2n zygote
• Zygote divides by meiosis producing n cells
• n cells divide by mitosis to produce the n multicellular adult
• Cells of the adult phase divide by mitosis in order to produce gametes
• **single celled zygote is the only 2n phase
\ Meiosis
• Meiosis produces haploid cells
• Many stages of meiosis are very similar to appearance to corresponding stages in mitosis
• Both Mitosis and Meiosis are preceded by the doubling of each chromosome in the cell
• Mitosis has one cell division
• Results in two daughter cells
• Meiosis has two cell divisions
• Results in four daughter cells
• Meiosis produces daughter cells with only half the number of chromosomes as the parent cell
• Meiosis consists of meiosis I and meiosis II
\ Interphase Preceding Meiosis \n • Before a cell enters meiosis I it goes through interphase the exact same way that a pre-mitotic cell does \n • Interphase duplicates the chromosomes and the organelles present inside of the cell \n • During interphase the cell doubles in size \n • Interphase consists of G1 , S and G 2 phases \n • Meiosis I consists of: \n • Prophase I \n • Metaphase I \n • Anaphase I \n • Telophase I (may be incomplete) \n • Cytokinesis I
\ Prophase I of Meosis I
• The most complex phase of the entire meiotic process \n • The chromatin coils, becoming visible as chromosomes \n under the microscope \n • The nuclear membrane and the nucleolus disappear \n • The spindle apparatus forms \n • A process called synapsis occurs \n • Homologous chromosome pairs line up beside one another \n ‘single file’ \n • Each of the homologues are present in duplicate containing \n two sister chromatids \n • Structure that results from synapsis is called a tetrad \n • Consists of four chromatids
• During prophase I chromatids of homologous pairs exchange pieces of \n chromosomes \n • Called crossing over \n • Responsible for producing genetic variation \n • This variation is produced because the \n homologues can carry alternate versions of a gene \n • Ex: one homologue encodes brown hair and the \n other encodes blonde hair \n • Crossing over occurs and the homologue encoding \n brown hair may now encode blonde hair and vice versa \n • The homologues contain genes other than hair color \n so crossing over has created a different combination \n of gene types on the same chromatid
\ Metaphase I of Meiosis I \n • Chromosome tetrads are aligned at the \n metaphase plate down the center of the cell \n • The sister chromatids remain attached to one \n another by their centromeres \n • Spindle microtubules are attached to the \n centromeres in preparation for cell division \n • Spindle microtubules from one pole of the cell \n are attached to one homologue \n • Spindle microtubules from the other pole are \n attached to the other homologue \n • Sets the stage for movement of chromosomes to \n opposite poles of the cell \n • Homologous pairs are held together at this point by \n the sites of crossing over
\ \ \