C4 - moles
- number of moles = mass(g) / Ar
- OR number of moles = mass(g) / Mr
- Ar is the top number in the periodic table, if you use that number you are finding the moles for that one element
- Mr is the mass of the whole formula when you use that number you are finding the weight in grams of the whole formula
\
- how many moles are present in 2.2g of CO2
Ar C = 12 O = 16
Mr 12 + (16 x 2) = 44
no. of moles = 2.2 / 44 = 0.05 mol
Mr 14 + (1 x 3) = 14 + 3 = 17
23.9 / 17 = 1.41 mol
\ \ From videos (https://www.youtube.com/@Freesciencelessons ) summary:
- the relative atomic mass of an element is the grams needed to make 1 mole (6.02 x 10^23 atoms), carbons Ar is 12 it means it takes 12g of carbon to make 1 mole. Oxygen has the Ar of 16 which means it takes 16g to make 1 mole.
- number of moles = mass / Ar (in an element)
- number of moles = mass / Mr (in a formula, so CO2, CaO, C6H12O6)
- calculating the mass from the number of mols : first you have to rearrange the formula and you have mass = no. of mols x Mr OR Ar. Then you either find the Mr and substitute or you just substitute.
- This method is necessary for more complex equations
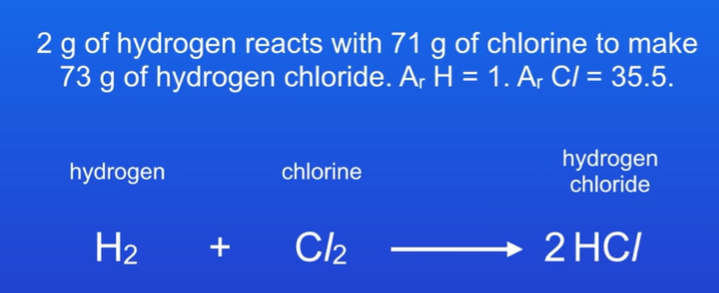
- find the number of mols for each one
H2 = mass / Ar = 2g / 2 = 1mol
Cl2 = mass / Ar = 71g/71 = 1mol
HCl = mass / Mr = 73g / 36.5 = 2mol
- present equations with mols
1H2 + 1Cl2 → 2HCl
- try simplify it as much as possible, divide by the smallest number
1H2 = 1/1 = 1
1Cl2 = 1/1 = 1
2HCl = 2/1 = 2
- remove any 1’s in front as they are not needed
\ more examples

- find mols
Al = 54g/27 = 2mols
FeO = mass/Mr = 216/ (56+16) = 3mols
Al2O3 = mass/Mr = 102/ 102 = 1mol
Fe = 168g/56 = 3mols
- 2Al + 3FeO → Al2O3 + 3Fe
\

- find mols
BaCl2 = 1248g/ (137 + (35.5x2)) = 6mols
Al2(SO4)3 = (2xAl) + (Sx3) + (Ox12) = 54+96+192 = 684g/342 = 2mols
BaSO4 = 137 + 32 + 64 = 233 = 1398/233 = 6mols
AlCl3 = 133.5 = 534/133.5 = 4mols
- rewrite
6BaCl2 + 2Al2 → 6BaSO4 + 4AlCl3
- simplify (/2)
3BaCl2 + Al2 → 3BaSO4 + 2AlCl3d
\ \ \ \ \ \ \
