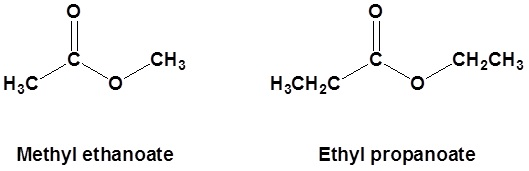
Esters
Formed through a reaction, known as “esterification”, between alcohols and carboxylic acids
They are often sweet smelling compounds (strong, pleasant). These smells may occur naturally (e.g. fruit flavours/scents) or can be synthetic (e.g. polyesters, terylene).
Esters can be used for food flavouring and perfumes
Their functional group is -COO, located in the middle of the chain
Smell of esters
Ester | Smell/flavour |
|---|---|
ethyl 2-methylbutanoate | apple |
3-methylbutyl ethanoate | pear |
1-methylbutyl ethanoate | banana |
butyl butanoate | pineapple |
octyl ethanoate | orange |
methylpropyl methanoate | raspberry |
pentyl butanoate | strawberry |
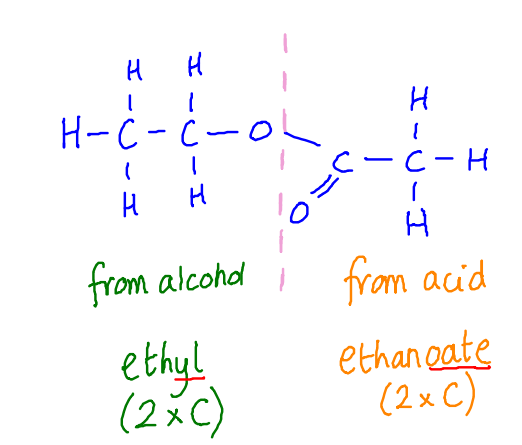
Naming an ester: alcohol name + carboxylic acid name
E.g. methanoic acid + propanol → propyl methanoate
The alcohol side will be attached to an O by a single bond, whereas the acid side will be attached to an O by a double bond.
Making esters
Generally, when we make esters, we use a concentrated sulphuric acid (H2SO4) catalyst. s
Carboxylic acid + alcohol → ester + water
ethanoic acid + ethanol → ethyl ethanoate + water
CH3COOH + C2H5OH → CH3COOC2H5 + H2O
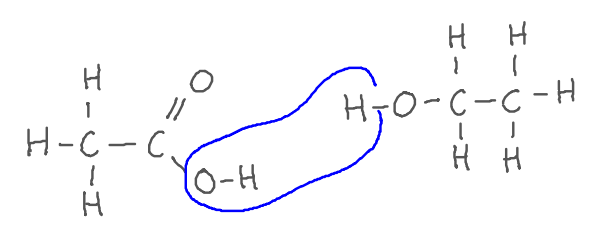
It is a condensation reaction as a small molecule of water is eliminated