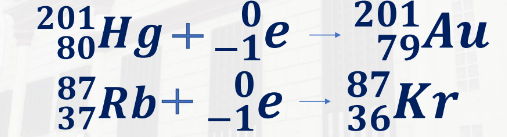Nuclear Reactions
- Atoms are building blocks of matter.
- Nucleosynthesis is the process of creating new atomic nuclei from preexisting nucleons (protons and neutrons).
- A nuclear reaction is a process wherein a nucleus is either undergoing nuclear fusion or nuclear fission.
- nuclear fusion is when two atoms join together to form a heavier atom
- nuclear fission is when an atom splits into smaller pieces
A nuclear reaction also involves the emission of energetic particles from an atom, a phenomenon known as radioactivity.
- The following radioactive particles are: elements, electrons, protons, neutrons
Element Keme
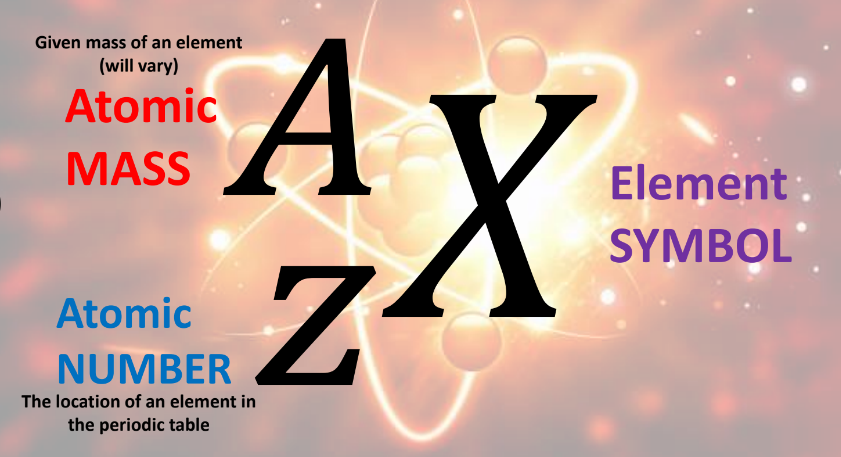
The atomic mass is, from what the name suggests, the given mass of an element. It will vary.
The atomic number is the location of an element in the periodic table.
The element symbol is then denoted as X.
Radioactive Particles
- electron - negatively charged particle
- proton - positively charged particle
- neutron - no charge at all
- alpha particle - high-speed particle consisting of 2 protons and 2 neutrons
- beta particle - high-speed electron
- gamma ray - high energy stream of photons.
- positron - positive-charged electron
Types of Nuclear Reactions PT.1 - Formulas

- alpha decay = 4,2α
- beta decay = 0,-1β
- gamma radiation = 0,0Υ
- positron emission = 0,+1e
- electron capture = 0,-1e
Types of Nuclear Reactions PT.2 - Problems
Alpha decay is the loss of alpha particles. It is denoted by 4,2α.
Alpha decay occurs when a nucleus is unstable because it has too many protons.
Examples:
- Polonium-210 = 210,84Po - 206,82Pb + 4,2α
- Platinum-180 = 180,78Pt - 176,76Os + 4,2α
- Cadmium-320 = 320,48Cd - 316,46Pd + 4,2α
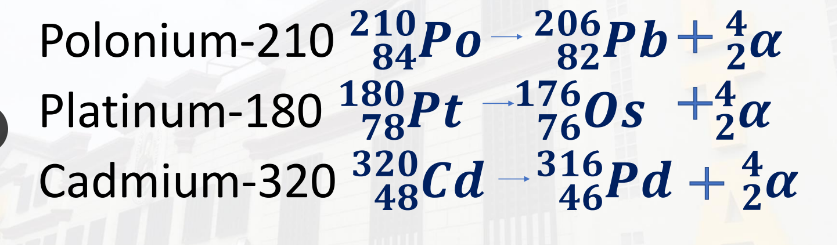
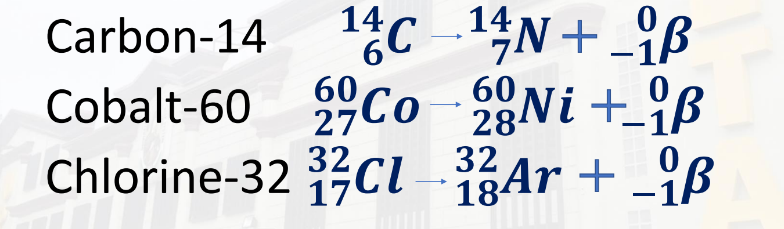
Beta decay is the loss of beta particles. It is denoted by 0,-1β.
Beta decay occurs when an atom has either too many protons or too many neutrons in its nucleus.
Examples:
Carbon-14 = 14,6C - 14,7N + 0,-1β
Cobalt-60 = 60,27 - 60,28Ni = 0,-1β
Chlorine-32 = 32,17Cl - 32,18Ar = 0,-1β
Gamma radiation is the emission of gamma rays. It is denoted by 0,0Y.
Gamma rays are produced during the disintegration of radioactive atomic nuclei and the decay of certain subatomic particles.
Example:
Gamma radiation in alpha decay of Uranium-238
alpha decay: 238,92U - 234,90Th + 4,2α
gamma radiation: 234,90Th + 0,0Y

Positron emission is the conversion of a proton in a nucleus into a neutron, along with the release of a positron 0,+1e
Positron emissions are produced if there are too many protons and not enough neutrons____.
Example:
Positron emission of Oxygen-15 and Chlorine-33
Oxygen-15 = 15,8O - 15,7N + 0,+1e
Chlorine-33 = 33,17 - 33,16S = 0,+1e
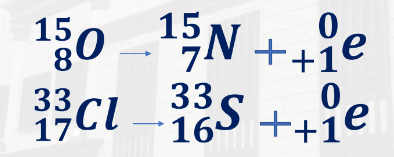
Electron capture is the drawing of an electron (0,-1e) into an atom’s nucleus
When a nucleus captures an electron, a proton changes into a neutron, and an electron-type neutrino is emitted. The atomic number goes down by one unit, accounting for the loss of a proton, and the total number of protons plus neutrons stays unchanged, accounting for the gain of a neutron.
Example:
Electron capture of Mercury-201 and Rubidium-87
201,80Hg + 0,-1e - 201,79Au
87,37Rb + 0,-1e - 87,36Kr