Redox Reactions Notes
Identify that displacement reactions of metals, combustion, corrosion and electrochemical processes, can be modelled as redox reactions involving oxidation of one substance and reduction of another substance.
Redox Reaction: They are displacement reactions of metals, combustion, corrosion and electrochemical processes that involve the oxidation of one substance and reduction of another substance.
Determine the species oxidised and reduced, and the oxidising agent and reducing agent, in redox reactions.
Species Oxidised: Loss of electrons.
Species Reduced: Gain of electrons.
Oxidising Agents: They take electrons.
Reducing Agents: They donate electrons.
Explain that oxidation can be modelled as the loss of electrons from a chemical species, and reduction can be modelled as the gain of electrons by a chemical species; these processes can be represented using balanced half-equations and redox equations (acidic conditions only).
Oxidation: The loss of electrons form a chemical species
Reduction: The gain of electrons by a chemical species
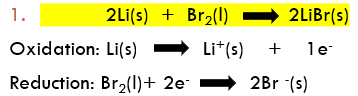
Half Equations and Redox Equations:
Determine the oxidation state (represented with the sign given before the number) of an atom in an ion or compound, e.g. +2.
Oxidation: Oxidation state increases.
Reduction: Oxidation state decreases.
Apply oxidation numbers (represented as roman numerals) to name transition metal compounds.
Naming Transition-metal Compounds: Assign the metal an oxidation number from the other ions’ charges and write it as a Roman numeral in brackets after the metal’s name.
Apply half-equations and oxidation numbers to balance redox equations (acid conditions only) and to discriminate between the species oxidised and reduced, and the oxidising agent and reducing agent.
Analyse data, including displacement reactions of metals, combustion, corrosion and electrochemical processes to determine redox reactions.