Chemistry of Life
- Matter v. Mass v. Weight
- Elements, Molecules, and Compounds
Questions
• True/False: All compounds are molecules, but not all molecules are compounds.
TRUE
• Classify the following as either an element, molecule, compound, or both molecule and compound
• O2 - molecule
• H2O - both molecule and compound
• Na - element
• CO2 - both molecule and compound
• H2 - molecule
- Atoms
- Atomic number vs mass number
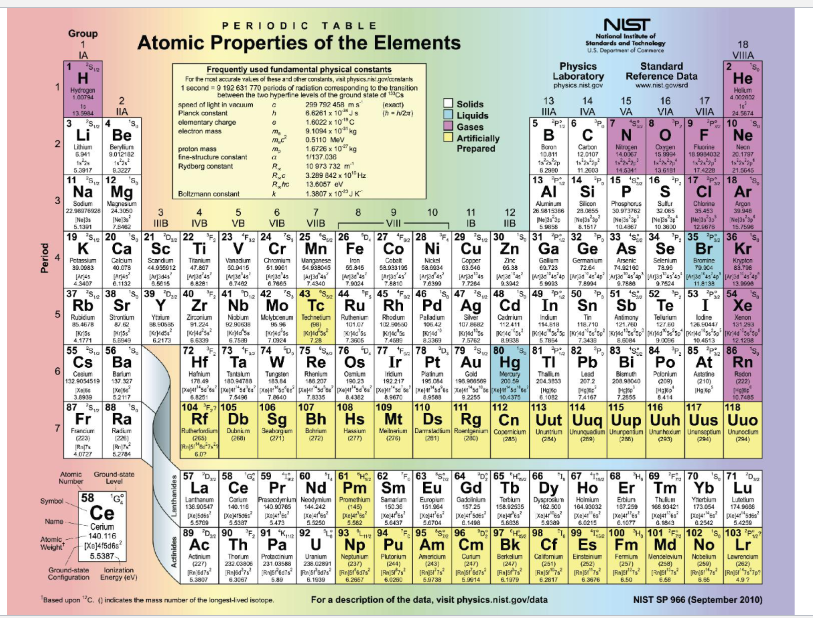
Practice Using the Periodic Table
• How many protons are in an oxygen atom? - 8
• What is the atomic mass of calcium? - 40.078
• What element has 18 protons? - Argon
• What is the atomic number of silicon? - 14
- Isotopes
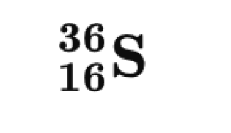
• How many protons? - 16
• How many neutrons? - 20
• How many electrons? - 16
• What is the atomic number? - 16
• What is the mass number? - 36
- Electrons
- the arrangement of the periodic table
- ions
- chemical bonds (3 types)
- ionic bonds
- covalent bonds
- Polar Covalent Bonds
- Hydrogen bonds (between oxygen and hydrogen)
What is a negative ion called? - Anion
What type of bond involves the sharing of electrons? covalent bonds
What type of bond is strongest? covalent bond
What type of bond is weakest? Hydrogen bond
When an atom loses an electron, it become a(n) - cation
- what component of solution (solution, solute, solvents)
- why does NaCl dissolve in water? (interact with ions)(like dissolve like)
- hydrophobic and hydrophilic
- amphipathic
: Ex: If you spill salad dressing on your hands and try to wash it off using only water, why don’t your hands feel completely clean? (this is the question in essay “Know that substances dissolve best in solvents with similar chemical properties (polar vs. nonpolar).
The salad dressing is nonpolar, and the water is polar. Like dissolves like, so salad dressing doesn’t dissolve in water.
Why does soap need to be used, in addition to water, to effectively clean oil off surfaces?
Since soap is amphipathic, it has a hydrophobic end that can dissolve the salad dressing (which contains oil), and a hydrophilic end that binds to water.
- Macromolecules
- Monomer vs Dimer vs Polymer
- Carbohydrates
- carbohydrates: monosaccharides
- hexose sugars (CH2OH)
- Pentose sugars (HOCH2)
- carbohydrates: disaccharides
- carbohydrate: polysaccharides
- lipids
Do you think lipids dissolve in water? - NO
- lipids: glycerides
- saturated vs unsaturated fats
Which is more likely to be solid at room temperature? Liquid?
Saturated is solid at room temperature
Unsaturated is liquid at room temperture
- lipids: phospholipids
-lipids: steroids
-proteins
-protein: structure of an amino acid
-proteins: a formulation of peptide bonds
-proteins: form = function
-nucleic acids