Intermolecular Forces and Condensed States: Comprehensive Notes
Intermolecular Forces & Solids & Liquids
Objectives
Describe and differentiate the types of intermolecular forces.
Describe the following properties of liquid and explain the effect of intermolecular forces on these properties: surface tension, viscosity, vapor pressure, boiling point, and molar heat of vaporization.
Describe the difference in the structure of crystalline and amorphous solids.
Intermolecular Forces
Forces that hold condensed states together.
Generally attractive forces based on electrostatic attraction (positive and negative charges).
Intramolecular: "within" a molecule.

Can hold together identical particles.
Not strongly affected by physical changes.
Chemical bonds.
Determine the chemical behavior of a substance.
Intermolecular: "between" molecules.
Can hold particles together.
Strongly affected by physical changes.
Attractive forces.
Determine the state of matter.
Coulomb's Law
The potential energy (E) of two oppositely charged particles decreases (becomes more negative) with increasing magnitude of charge and with decreasing separation (r) between them.
Bonding forces result from large charges interacting at close distances.
Intermolecular forces result from smaller charges interacting at greater distances.
Types of Intermolecular Forces
Dispersion Forces (London Dispersion Forces)
Dipole-dipole Forces
Hydrogen Bonds
Ion-dipole Forces
Dispersion Forces (London Dispersion Forces)
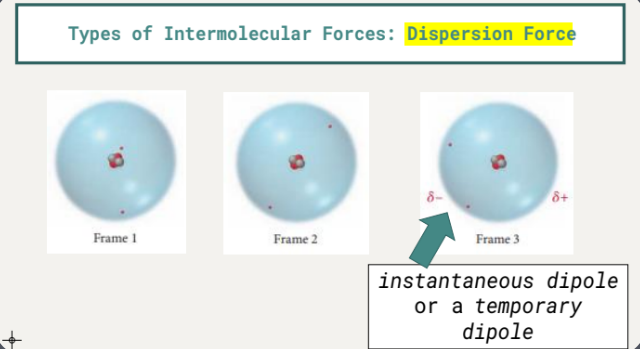

Present in all molecules and atoms.
Result from fluctuations in electron distribution within molecules or atoms.
Create instantaneous or temporary dipoles.
Magnitude depends on how easily electrons polarize, which depends on the size/volume of the electron cloud.
Dispersion Force and Molecular Shape
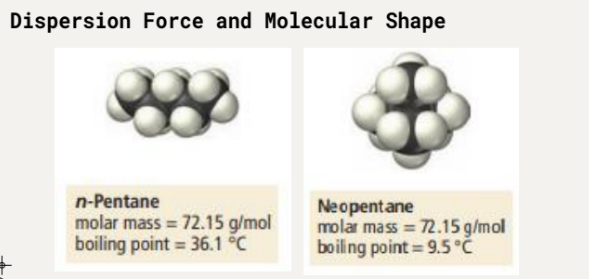
Example:
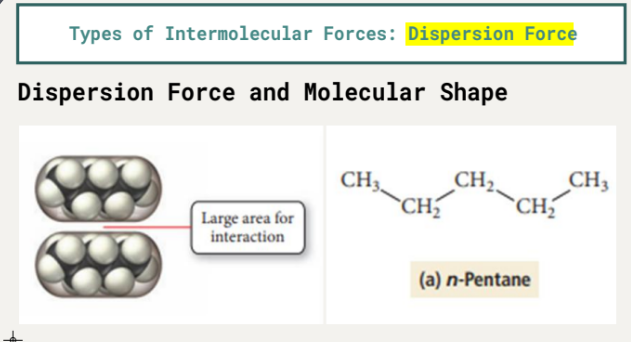
n-Pentane: molar mass = 72.15 g/mol, boiling point = 36.1 °C
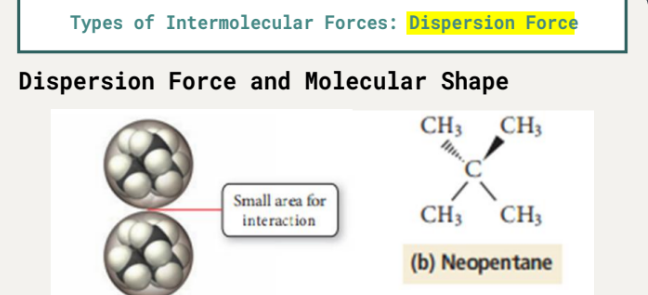
Neopentane: molar mass = 72.15 g/mol, boiling point = 9.5 °C
n-Pentane has a larger area for interaction compared to Neopentane, leading to a higher boiling point.
Boiling Points of n-Alkanes
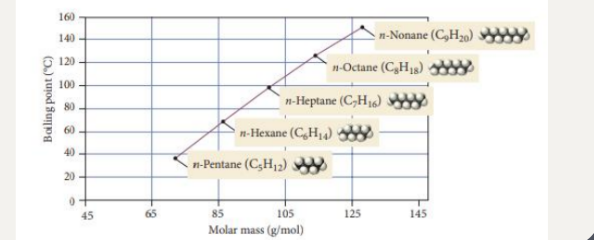
Boiling point increases with molar mass due to increased dispersion forces.
Example: n-Nonane (C9H20) has a higher boiling point than n-Pentane (C5H12).
Quick Exercise
Which halogen has the highest boiling point?
Answer: Iodine (I2)
Dipole-Dipole Forces
Exist in all polar molecules.
Polar molecules have electron-rich (partial negative charge) and electron-deficient regions (partial positive charge).
The positive end of one polar molecule is attracted to the negative end of another.
How to determine if a molecule has dipole-dipole forces:
Determine if the molecule contains polar bonds.
Determine if the polar bonds add together to form a net dipole moment.
Bond Polarity:
Depends on the electronegativity difference between the bonding atoms.
The greater the difference, the more polar the bond.
Electronegativity values (Pauling scale) are provided in a table.
Bond Polarity
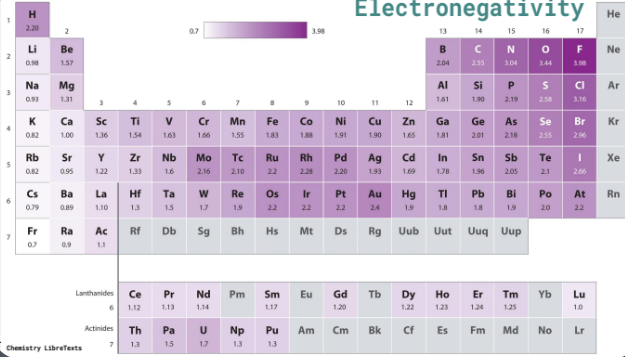
Small (0 - 0.4): Non-polar covalent bond.
Intermediate (0.4 - 2.0): Polar covalent bond.
Two atoms with identical electronegativities form a nonpolar covalent bond (e.g., Cl-Cl).
Large electronegativity difference leads to electron transfer, forming an ionic bond.
Continuum of Bond Types:
Pure covalent: Electrons shared equally.
Polar covalent: Electrons shared unequally.
Ionic: Electrons transferred.
Molecular Shape & Polarity
Geometric shapes influence polarity.
Examples:
Linear: Can be nonpolar if symmetrical.
Trigonal planar: Can be nonpolar if symmetrical.
Bent: Usually polar.
Tetrahedral: Can be nonpolar if symmetrical.
Trigonal pyramidal: Usually polar.
Net dipole moment is the vector sum of individual bond dipoles.
Polar molecules have higher melting and boiling points than nonpolar molecules of similar molar mass.
Dipole Moment and Boiling Point
Boiling points increase with increasing dipole moment.
Polar and Nonpolar Compounds
Miscibility:
Polar + Polar: Miscible (forms a solution).
Nonpolar + Nonpolar: Miscible.
Polar + Nonpolar: Non-miscible.
Water (polar) and pentane (nonpolar) do not mix.
Hydrogen Bonding
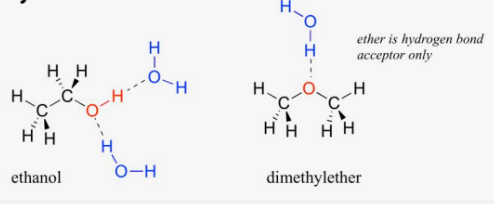
Occurs in polar molecules containing hydrogen atoms bonded directly to small, electronegative atoms (F, O, N).
A "super" dipole-dipole force.
Example: Hydrogen bonding in HF.
Ethanol (C2H5OH) has a higher boiling point than dimethyl ether (CH3OCH3) due to hydrogen bonding.
*Hydrogen bonding between antiparallel double stranded DNA is a vital function to its stability
Hydrogen Bonding in Water
Anomalously high boiling point compared to other hydrogen-containing compounds due to hydrogen bonding.
Quick Exercise:
One of the compounds shown here is a liquid at room temperature. 30.03g/mol, 34.03g/mol, 34.02g/mol. Which one and why?
Ion-Dipole Force
Occurs when an ionic compound is mixed with a polar compound.
Important in aqueous solutions of ionic compounds.
Responsible for the ability of ionic substances to form solutions with water.
Summary of Intermolecular Forces
Type | Present In | Strength (kJ/mol) | Molecular Perspective |
|---|---|---|---|
Dispersion | All molecules and atoms | 0.05-20+ | |
Dipole-dipole | Polar molecules | 3-20+ | |
Hydrogen | Molecules with H bonded to F, O, N | 10-40 | |
Ion-dipole | Mixtures of ionic and polar compounds | 30-100+ |
Intermolecular Forces in Action
Surface Tension
Viscosity
Capillary Action
Surface Tension
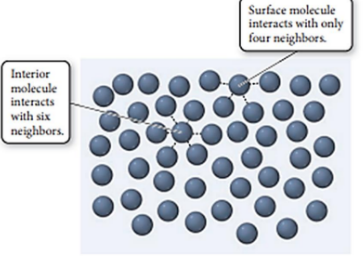
The energy required to increase the surface area of a liquid by a unit amount.
Viscosity
The resistance of a liquid to flow.
Viscosity of Several Hydrocarbons at 20 °C
Hydrocarbon | Formula | Molar Mass (g/mol) | Viscosity (cP) |
|---|---|---|---|
n-Pentane | CH3CH2CH2CH2CH3 | 72.15 | 0.240 |
n-Hexane | CH3CH2CH2CH2CH2CH3 | 86.17 | 0.326 |
n-Heptane | CH3CH2CH2CH2CH2CH2CH3 | 100.2 | 0.409 |
n-Octane | CH3CH2CH2CH2CH2CH2CH2CH3 | 114.2 | 0.542 |
n-Nonane | CH3CH2CH2CH2CH2CH2CH2CH2CH3 | 128.3 | 0.711 |
Viscosity decreases as temperature increases.
Capillary Action
The ability of a liquid to flow against gravity up a narrow tube.
Results from:
Cohesive forces: Between molecules in a liquid.
Adhesive forces: Between liquid molecules and the surface of the tube.
Meniscuses: curvature of water and mercury.
Vaporization and Vapor Pressure
Vaporization: The process by which thermal energy overcomes intermolecular forces, causing a state change from liquid to gas.
The higher the temperature, the greater the average energy of the molecules.
Factors affecting the rate of vaporization:
Increasing temperature increases the rate.
Increasing surface area increases the rate.
Decreasing strength of intermolecular forces increases the rate.
Types of Solids
Amorphous solids:
Ex: rubber, cotton candy, plastics.
Crystalline solids:
Ex: ice, table salt, metals.
Crystalline Solids: Unit Cells & Basic Structures
The crystalline lattice is represented by a small collection of atoms, ions, or molecules called the unit cell.
Fundamental Types of Crystalline Solids
Molecular: Composite units are molecules, held together by intermolecular forces, low to moderately low melting points.
Ionic: Composite units are ions, held together by coulombic interactions, high melting points.
Atomic: Composite units are individual atoms.
Nonbonding atomic solids: Noble gases in solid form, held together by weak dispersion forces.
Metallic atomic solids: Held together by metallic bonds (interaction of metal cations with a "sea" of electrons).
Network covalent atomic solids: Crystal structures restricted by geometrical constraints of covalent bonds.