Covalent Bonds
occur between 2 or more non-metals
neither will donate electrons, they must share
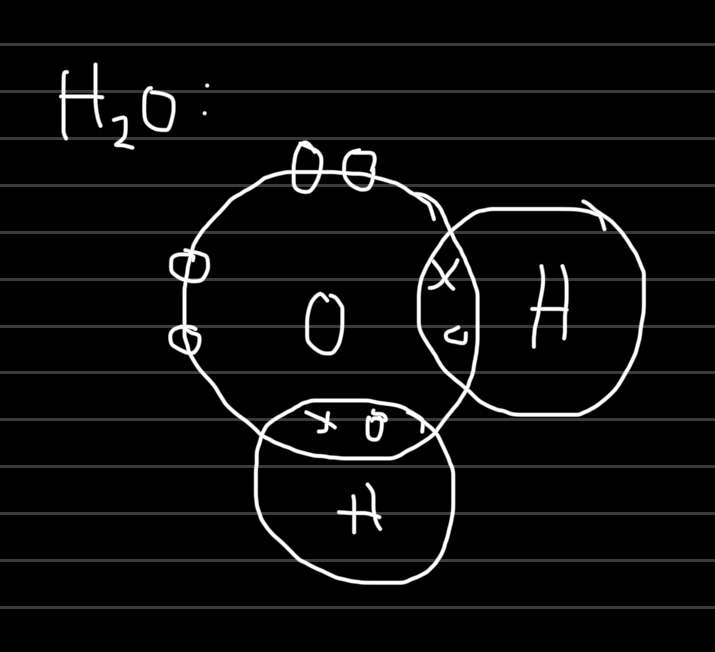
In covalent bond, the pair share the space between the 2 nuclei.
charged electrons are attracted to the positive charges of both nuclei, which overcomes the repulsion between the two shared electrons.
Covalent bond → the stong electrostatic attraction between the shared pair of electrons and the nuclei of the bonded atoms.
Single Covalent Bond:
The electrons in a covalent bond are in between the 2 atoms, therefore they are DIRECTIONAL.
How to remember all the diatomic atoms:
I → iodine
Have → Hydrogen
No → Nitrogen
Clever → Chlorine
Or → Oxygen
Bright → Bromine
Friends → Florine
Multiple Covalent Bonds:
This depends on how many single upaired electrons there are in the outershell.
Lone pairs:
An electron pair that is not used for bonding is called a LONE PAIR. When a compound has 2 sets of lone pairs, you must draw the bonds in a mickey mouse slanted shape: