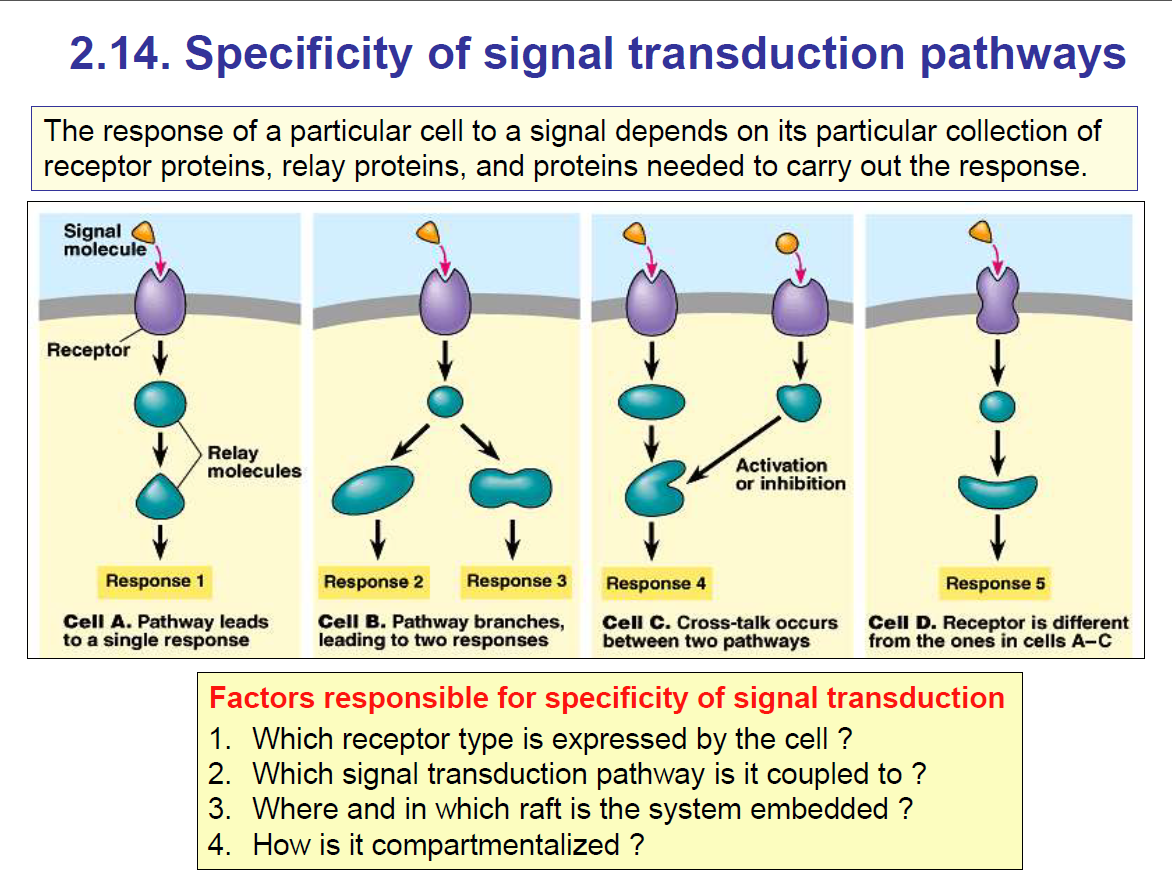
Signal Transduction
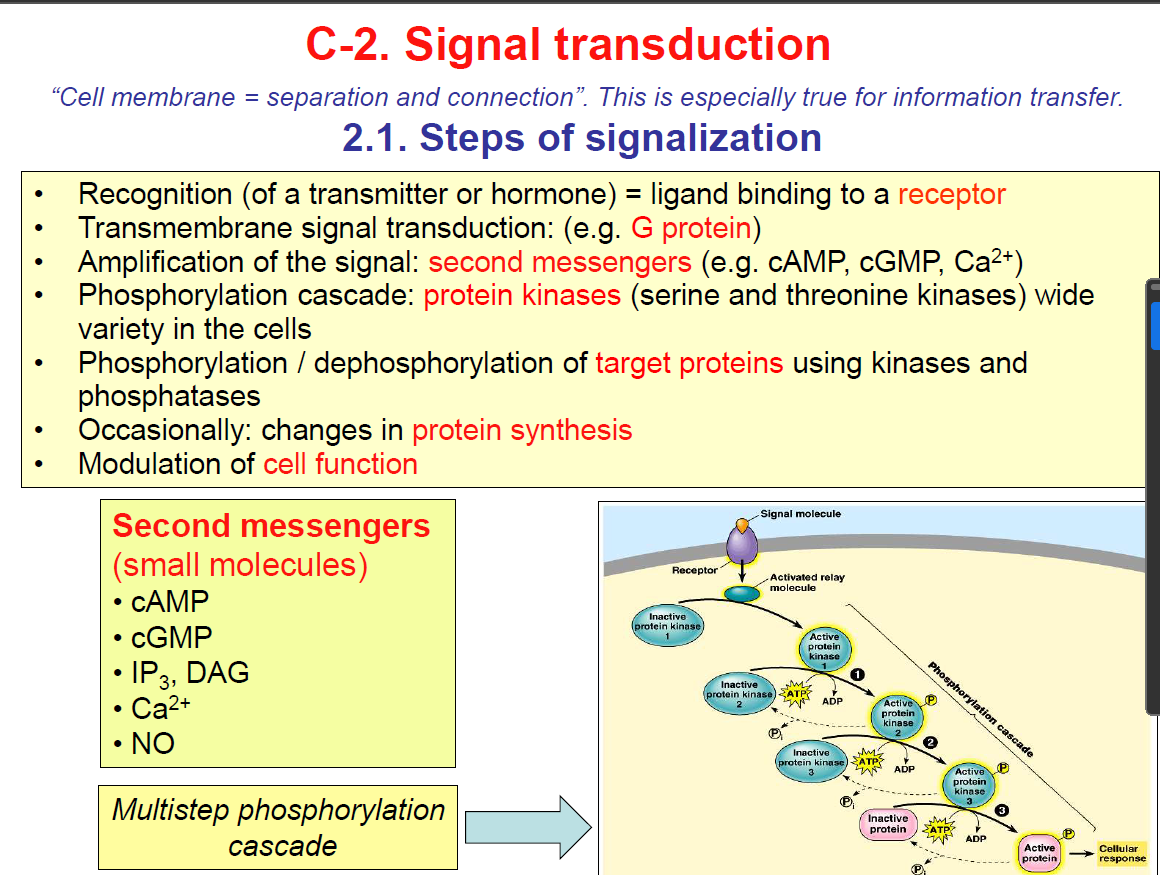
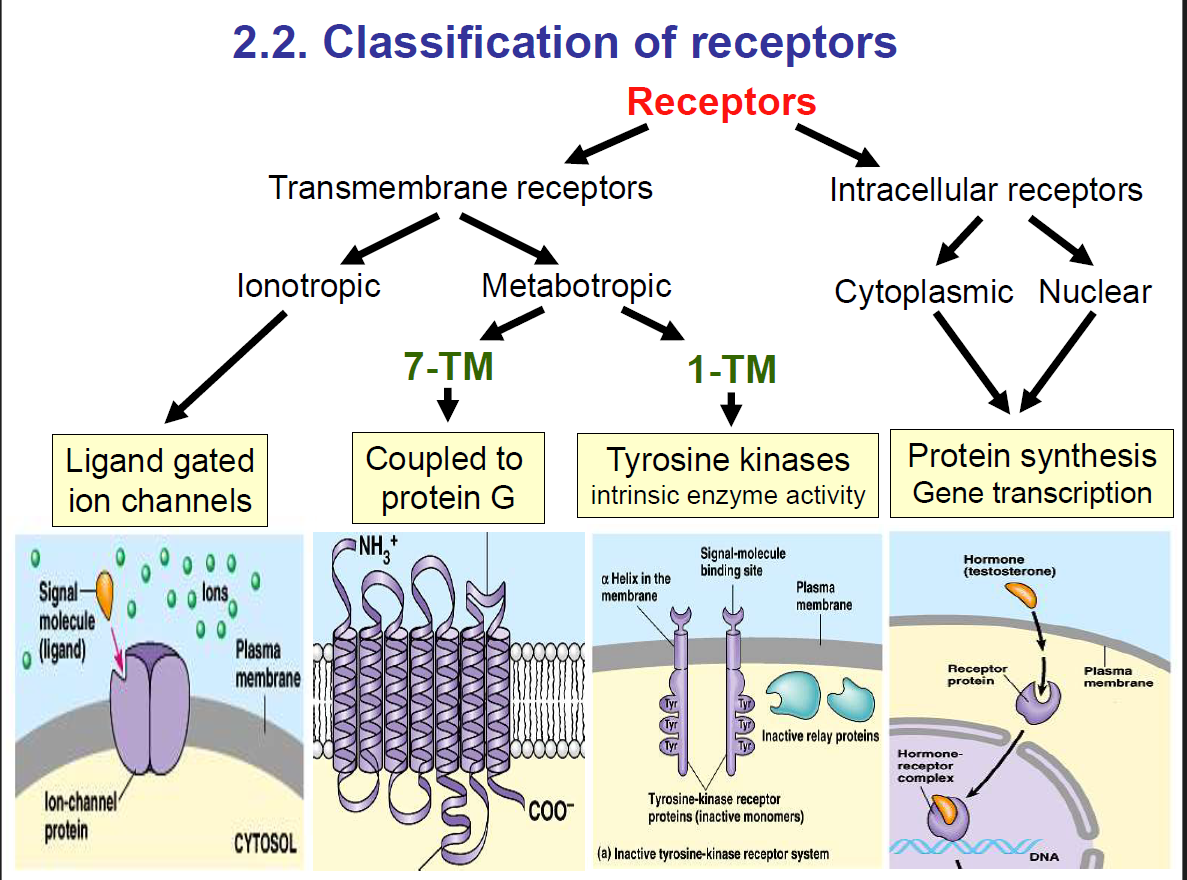
Classification of Receptors:
Receptors are further classified into two:
Transmembrane receptors
Intracellular receptors.
1. Transmembrane receptors:
These are receptors that span the plasma membrane.
They bind ligands that are usually hydrophilic (can’t cross the lipid bilayer).
Binding causes either direct ion flow or intracellular signalling cascades.
Two major classes of transmembrane receptors include:
Ionotropic receptors
Metabotropic receptors
1. Ionotropic Receptors (Ligand-Gated Ion Channels)
Definition: Ionotropic receptors are ligand-gated ion channels. When the ligand binds, the channel opens and ions flow directly.
Mechanism: Fast, direct, milliseconds.
Examples:
Nicotinic acetylcholine receptor (Na⁺/K⁺ channel at neuromuscular junction).
GABA_A receptor (Cl⁻ channel, inhibitory).
NMDA/AMPA receptors (glutamate, Ca²⁺/Na⁺ channels).
Function: Rapid signalling, e.g., synaptic transmission in the nervous system.
2. Metabotropic Receptors (G-Protein Coupled Receptors, GPCRs)
Definition: Receptors that, when activated by a ligand, interact with a G-protein inside the cell to trigger a second messenger cascade.
Mechanism: Slower, indirect, seconds to minutes.
Examples:
Muscarinic acetylcholine receptors (M2 in the heart).
Adrenergic receptors (α, β receptors for norepinephrine/epinephrine).
Dopamine and serotonin receptors.
Function: Modulate metabolism, gene expression, and long-lasting effects.
Metabotropic receptors are further classified into:
7 Transmembrane (7TM) Receptors
Also called: G-protein coupled receptors (GPCRs) i.e. these are receptors that are attached to G-proteins.
Structure: 7 transmembrane α-helices.
Mechanism: Ligand binding → activates G-protein (Gs, Gi, Gq etc.) → triggers second messengers (cAMP, IP₃, DAG, Ca²⁺).
Examples: Adrenergic receptors (α, β), muscarinic ACh receptors (except nicotinic), dopamine receptors.
1 Transmembrane (1TM) Receptors
These are single-pass transmembrane proteins, further divided into two types:
a) With intrinsic enzyme activity
These receptors have enzyme activity in their intracellular domain.
Example: Receptor tyrosine kinases (RTKs).
Ligand binding → dimerisation → autophosphorylation on tyrosine residues → downstream signalling.
Examples: Insulin receptor, EGF receptor.
b) Without intrinsic enzyme activity
These receptors have no enzymatic activity themselves.
Instead, they recruit cytoplasmic enzymes when ligand binds. So they are not enzymes but can activate enzymes.
Example: Cytokine receptors (e.g., growth hormone receptor, erythropoietin receptor).
They activate JAK-STAT pathway by recruiting Janus kinases (JAKs).
2. Intracellular receptors:
Located inside the cell (either in the cytoplasm or nucleus).
To activate them, the ligand must cross the plasma membrane.
Therefore, the ligands are usually lipid-soluble (hydrophobic), allowing them to diffuse through the lipid bilayer.
Examples of ligands
Steroid hormones: cortisol, aldosterone, estrogen, progesterone, testosterone
Thyroid hormones: T₃, T₄
Vitamin D
Retinoic acid (vitamin A derivative)
Intracellular receptors are further classified into:
Cytoplasmic receptors
Nuclear receptors
Mechanism:
Ligand crosses the membrane (since it’s hydrophobic).
Binds to its intracellular receptor (cytoplasmic or nuclear).
The ligand–receptor complex often acts as a transcription factor:
Moves into the nucleus (if not already there).
Binds to DNA at hormone-response elements.
Alters gene expression → changes protein synthesis.
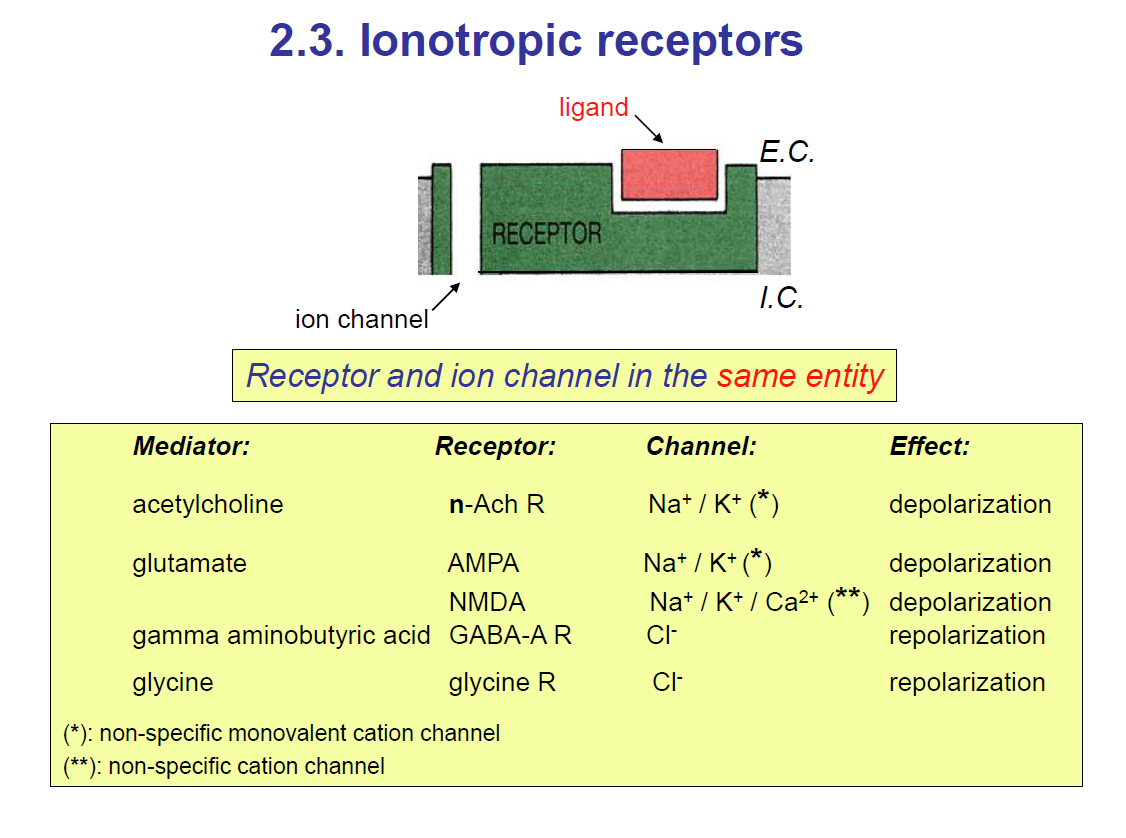
Ionotropic receptors are ligand-gated ion channels
For this type of receptor, when the ligand binds to the receptor, it activates a G-protein which then stimulates a signalling pathway.
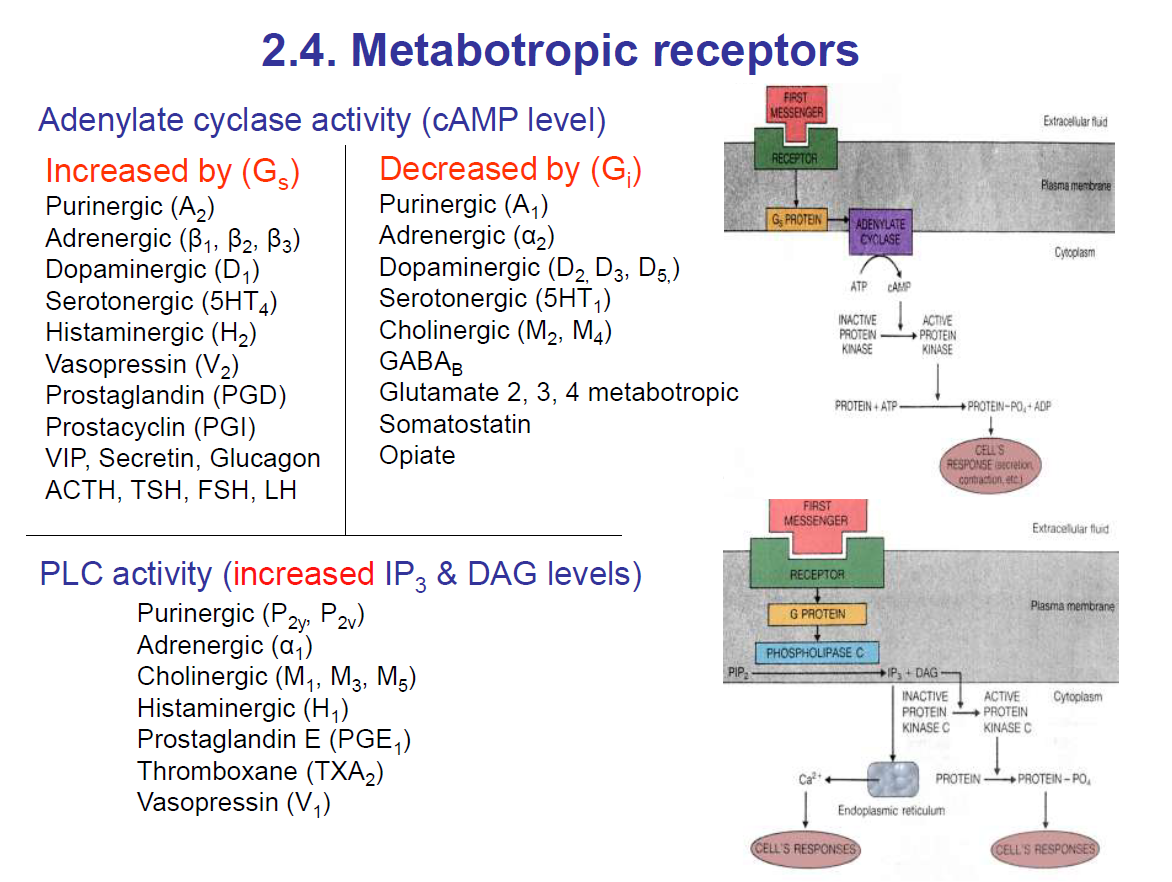
Adenylate Cyclase activity
Adenylate Cyclase activity is increased by Gs-
Gs activates adenylate cyclase which converts ATP to cAMP which activates protein kinase A.
Protein kinase A then phosphorylates the serine/threonine residues on target proteins thus activating or inactivating these target protein.
Adenylate Cyclase activity is decreased by Gi
Gi inhibits adenylate cyclase activity.
PLC activity
Gq activates Phospholipase C (PLC) which breaks down PIP2 to IP3 and DAG.
IP3 goes to the IP3 receptors to cause Ca2+ ion release.
DAG activates protein kinase C.
Structure of 7-TM metabotropic receptors
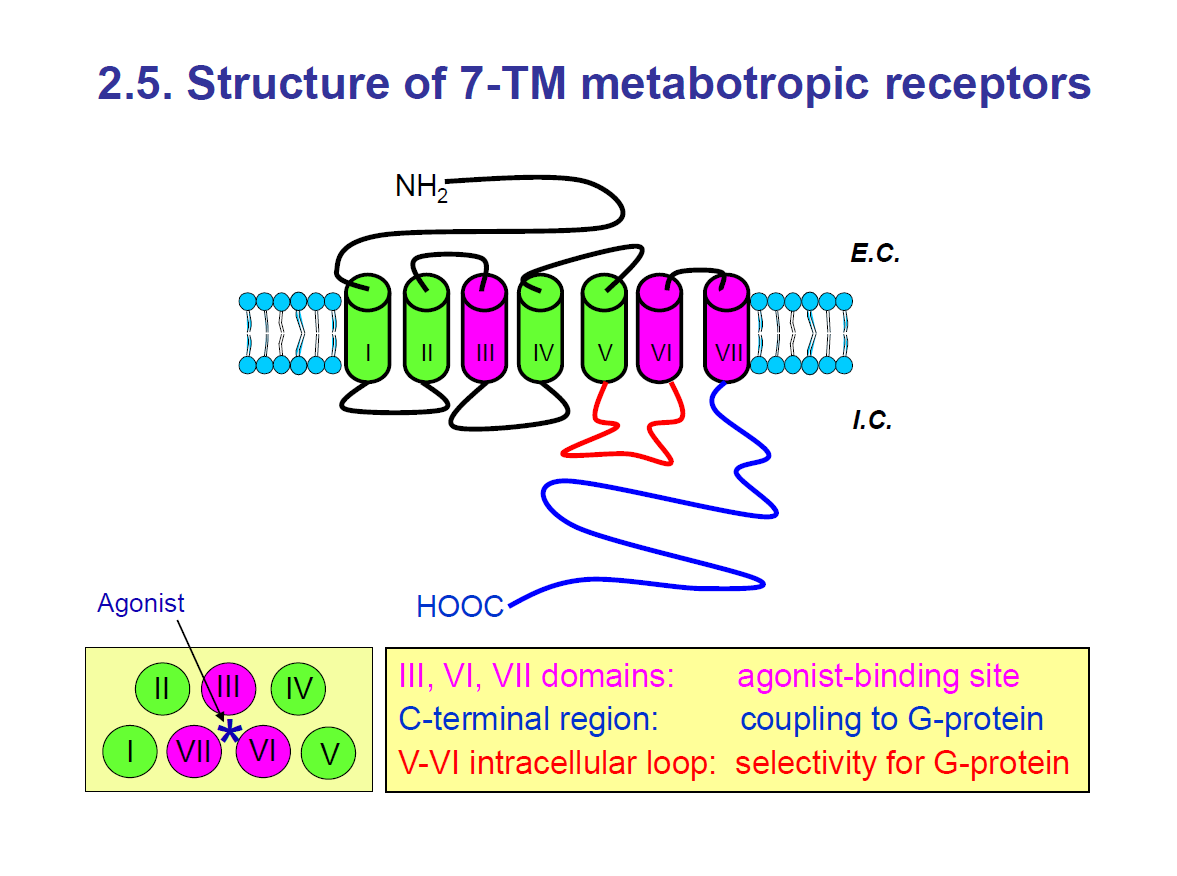
The 7-TM metabotropic receptors have 7 transmembrane proteins.
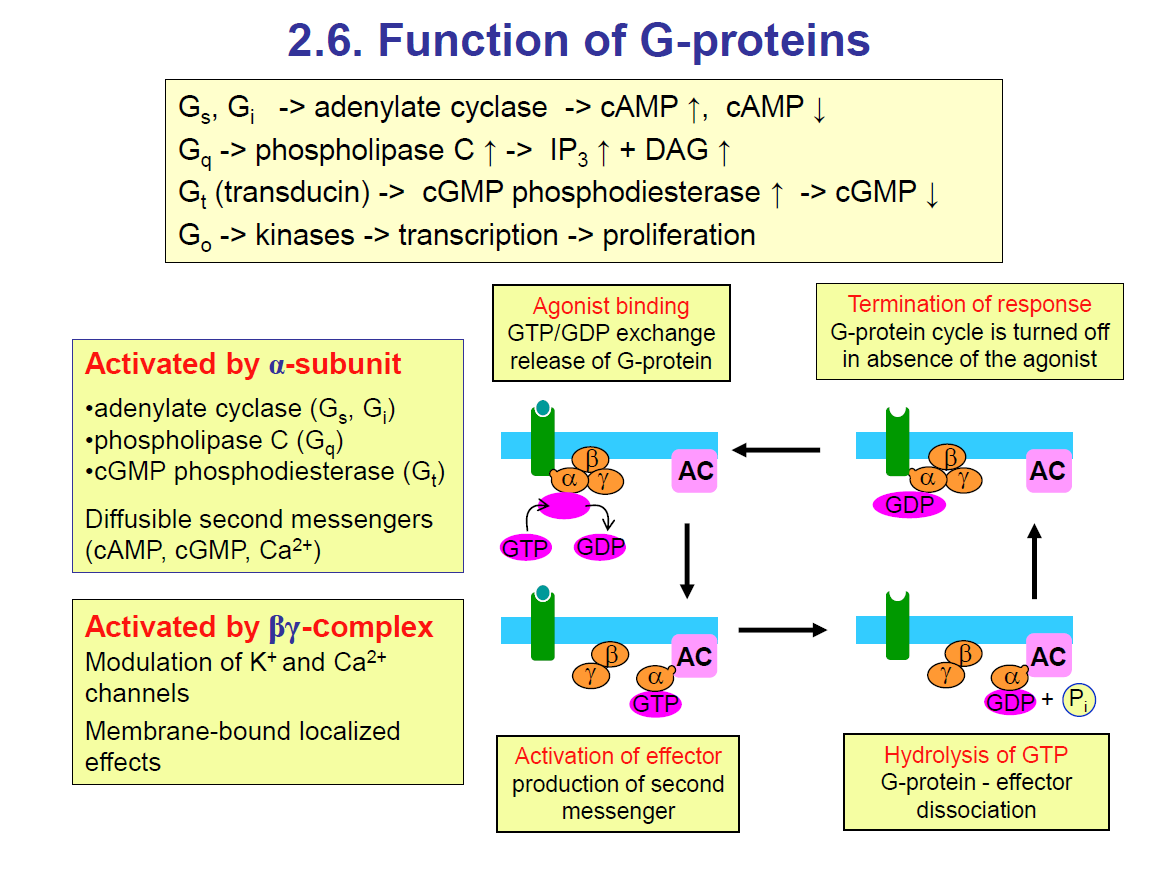
Function of G-proteins
G-proteins are molecular switches inside cells that couple receptors to effectors (enzymes or ion channels). They are made of 3 subunits: α, β, and γ.
Top Yellow Box: Different G-proteins & their effectors
Gs, Gi → act on adenylate cyclase
Gs → stimulates → ↑ cAMP
Gi → inhibits → ↓ cAMP
Gq → activates phospholipase C (PLC) → produces IP₃ + DAG → ↑ Ca²⁺ and activates PKC.
Gt (transducin) → activates cGMP phosphodiesterase (PDE)→ ↓ cGMP (important in vision, retina)
Go → activates kinases → transcription/proliferation
Left Yellow Box: What gets activated
Activated by α-subunit (depends on the G-protein type):
Adenylate cyclase (Gs, Gi)
Phospholipase C (Gq)
cGMP phosphodiesterase (Gt)
Produces second messengers: cAMP, cGMP, Ca²⁺
Activated by βγ complex:
Modulates K⁺ and Ca²⁺ channels
Has localised effects at the membrane
👉 So both α and βγ can signal, not just α.
Right diagram: The G-protein cycle
Agonist binding (top left box)
A ligand binds to the receptor (GPCR).
This causes GDP → GTP exchange on the α-subunit.
G-protein becomes active and splits into α-GTP and βγ.
Activation of effector (bottom middle box)
α-GTP activates the target enzyme (e.g., adenylate cyclase, phospholipase C, PDE).
Effector produces second messenger (cAMP, IP₃, DAG, cGMP, etc.).
Hydrolysis of GTP (bottom right box)
The α-subunit has GTPase activity.
GTP → GDP + Pi.
α reunites with βγ, turning the signal off.
Termination of response (top right box)
When the ligand (agonist) is gone, the cycle resets and signalling stops.
✅ In summary:
G-proteins couple receptors to enzymes.
Different α-subunits = different signalling pathways.
Cycle = agonist binding → GTP exchange → effector activation → GTP hydrolysis → reset.
Events induced by activation of 7-TM receptors
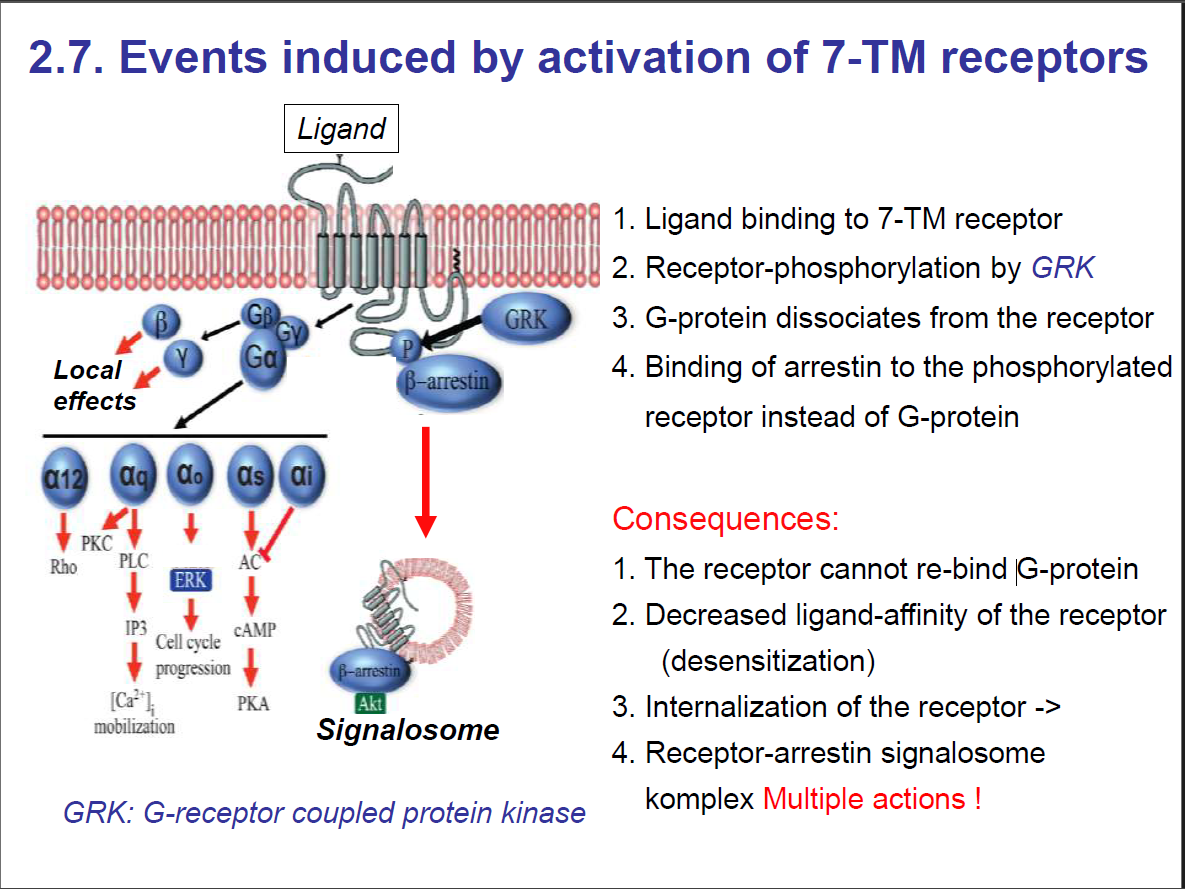
Role of Arrestin in GPCR Regulation
GPCR overstimulation risk:
If a ligand (like adrenaline) keeps binding, the receptor could signal too much. The cell needs a way to turn it off.Step 1: Receptor phosphorylation
After prolonged activation, GPCR kinases (GRKs) phosphorylate the cytoplasmic tail of the receptor.
Step 2: Arrestin binding
Arrestin binds to the phosphorylated receptor.
This prevents the receptor from coupling with G-proteins again → signal is blocked ("arrested").
Step 3: Receptor internalisation
Arrestin links the receptor to clathrin-coated pits, leading to endocytosis.
The receptor is pulled inside the cell.
Step 4: Fate of receptor
Inside the cell, the receptor can be:
Recycled back to the surface (resensitization), or
Degraded in lysosomes (downregulation).
Summary: How does the body prevent overstimulation of a receptor?
Arrestin
If a ligand (like adrenaline) keeps binding, the receptor could signal too much. The cell needs a way to turn it off. HOW?
Arrestin stops GPCR signalling by:
Blocking G-protein interaction- Arrestin binds to the receptor to prevent the receptor from coupling with G-proteins again —> signal is blocked (arrested).
Bringing receptors inside the cell (endocytosis).
Amplification
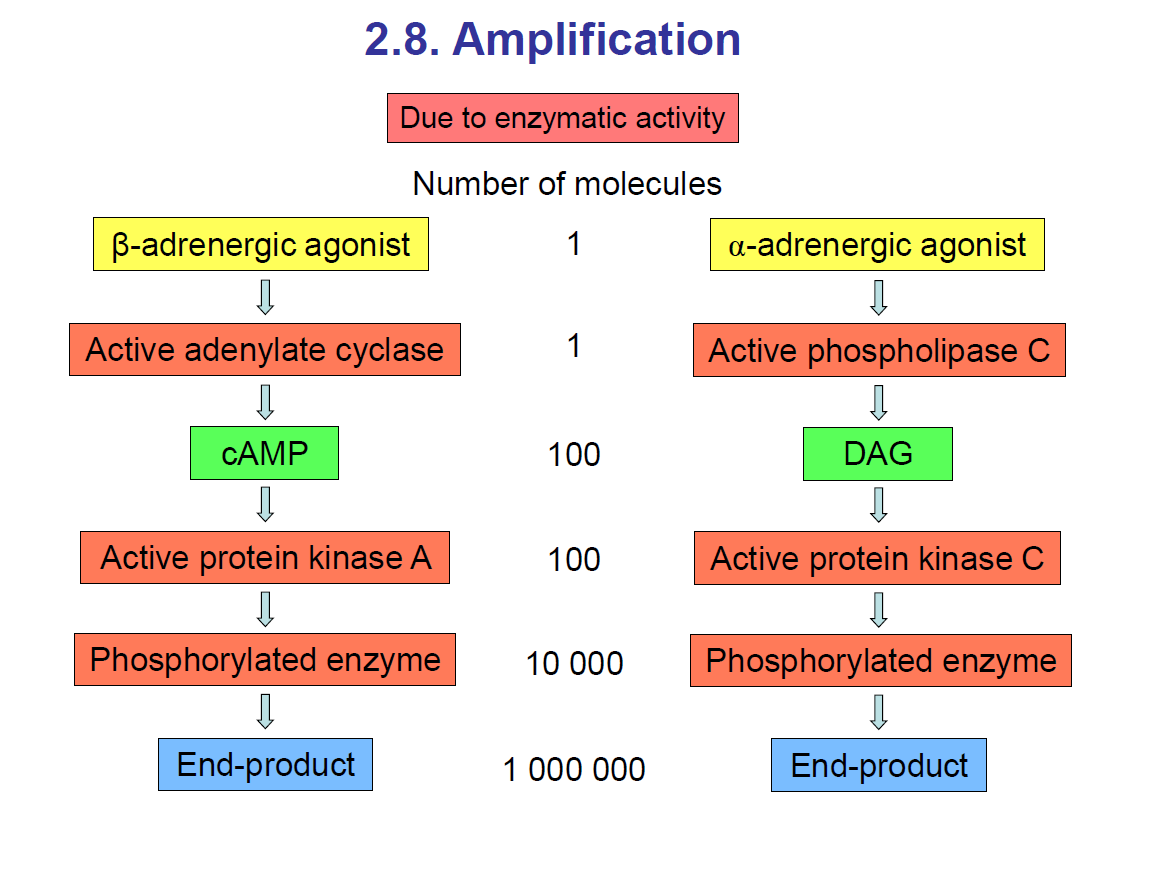
Take note: An agonist is a ligand which binds to and activates a receptor.
For example, the beta-adrenergic agonist is a ligand that binds to the beta-adrenergic receptor. It would activate the G-protein, which would activate adenylate cyclase, which would break down ATP to cAMP and inorganic phosphate. Then cAMP would activate protein kinase A. Then protein kinase A would phosphorylate the target enzyme, activating or inactivating this target protein.
Another example: An alpha-adrenergic agonist, which is a ligand, would bind to the alpha-adrenergic receptor. This would activate a G-protein, which would activate phospholipase C. Phospholipase C would break down PIP2 —> IP3 + DAG. Then DAG will activate protein kinase C. This would phosphorylate the target protein.
The second messengers, such as DAG or cAMP, help amplify the signal, allowing the cell to respond with sufficient stimulus.
Intracellular receptors
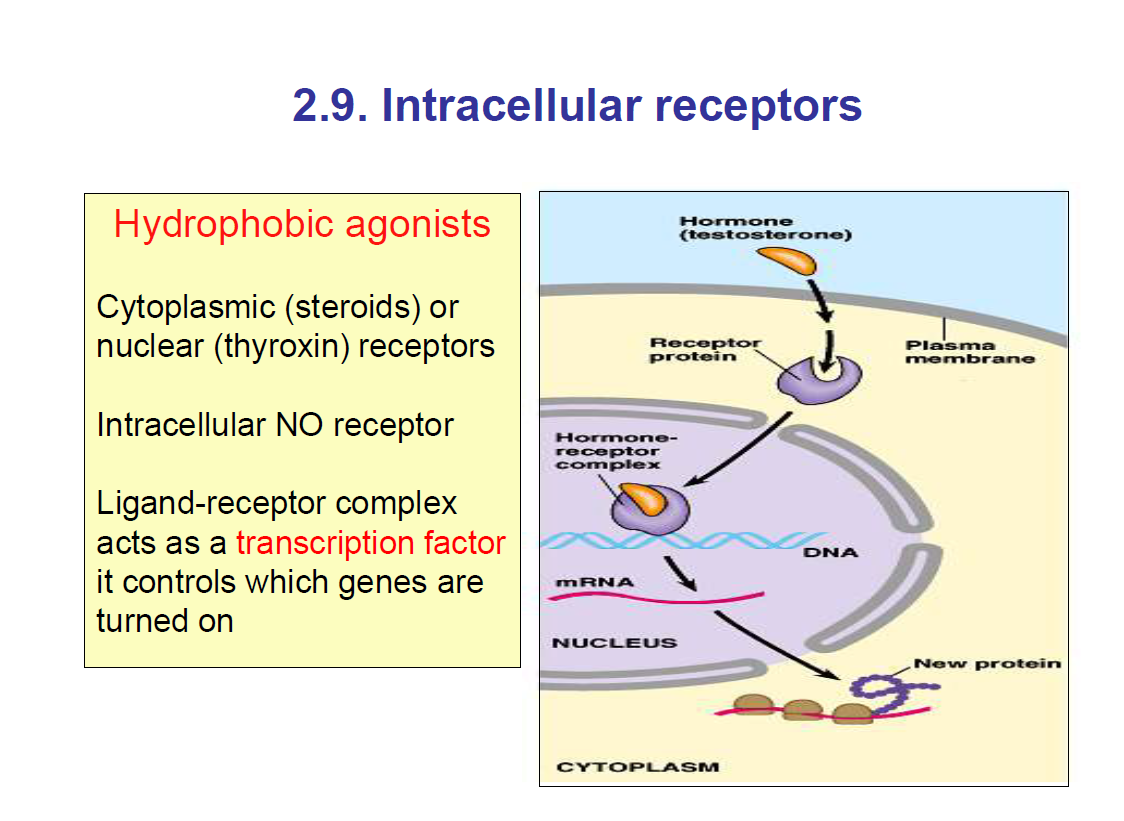
Intracellular receptors are inside the cell, either in the cytoplasm or in the nucleus
They act as transcription factors.
They are hydrophobic, so they can pass the lipid bilayer by simple diffusion.
Intracellular Ca2+ as second messenger
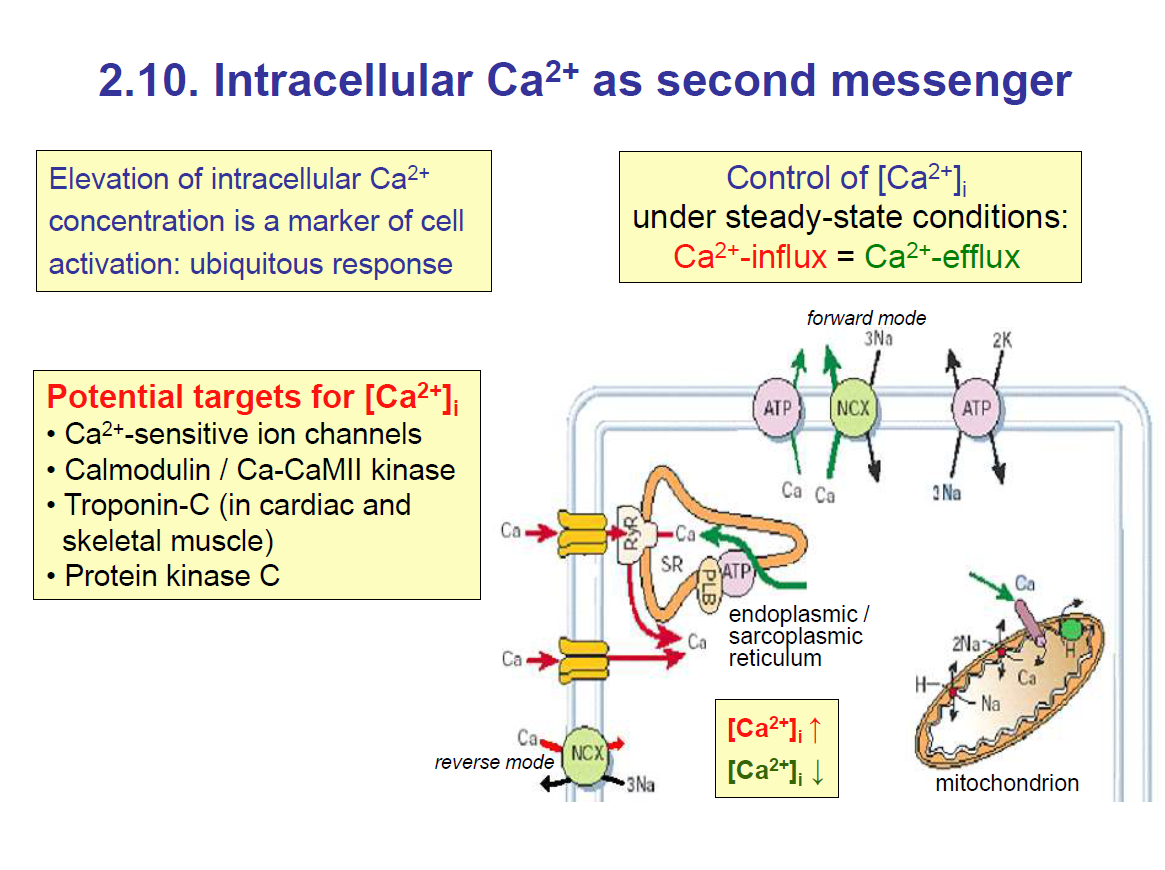
There are various second messengers such as: Ca2+, cGMP, cAMP, etc.
The Ca2+ ion has different functions in different targets
Ca2+ ion level has to be kept constant/steady because if it is too high, it can cause cell death.
Control of Ca2+ entry
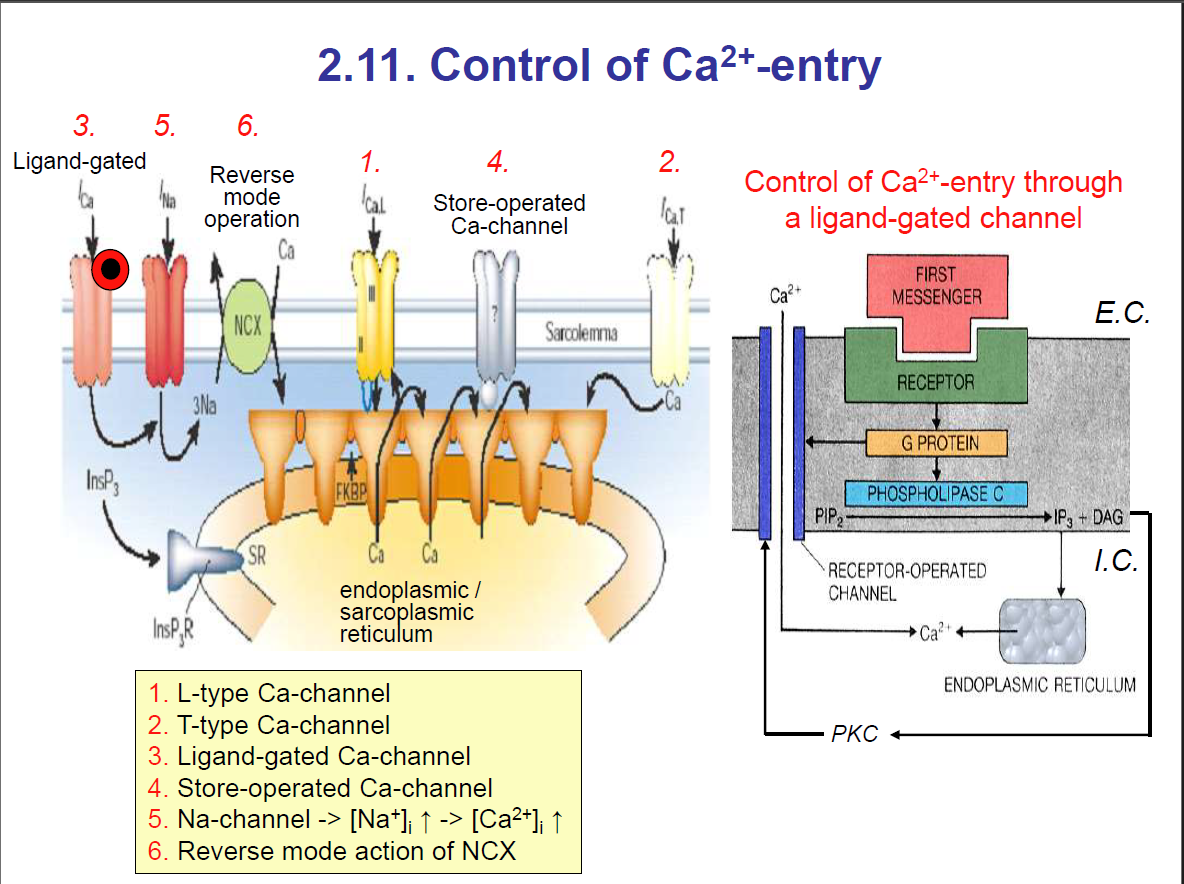
Types of Ca2+ channels
1. L-type Ca-channel: (Long-lasting)
Found in cardiac muscle, smooth muscle, skeletal muscle, and neurons.
Opens during depolarisation → allows a sustained Ca²⁺ influx.
Important for the plateau phase of cardiac AP, excitation–contraction coupling in muscle.
Blocked by drugs like verapamil and nifedipine (calcium channel blockers).
2. T-type Ca-channel: transient; they are open for a short time
Found in pacemaker cells (SA node, thalamic neurons).
Opens briefly → short, rapid Ca²⁺ entry.
Important for pacemaker potentials (automaticity).
3. Ligand-gated Ca-channel: Ligand binding to the receptor
Opened by ligand binding (not voltage).
Example: NMDA receptor in neurons (activated by glutamate + glycine → allows Ca²⁺ and Na⁺ influx).
Example: nicotinic ACh receptor (though mainly Na⁺/K⁺, some allow Ca²⁺).
4. Store-operated Ca-channel:
Activated when ER/SR Ca²⁺ stores are depleted.
Sensor: STIM1 (in ER membrane).
Channel: Orai1 (in plasma membrane).
Important in immune cells (T-lymphocyte activation).
Other channels
Na+ channel:
Voltage-gated Na⁺ channels → open during depolarisation → cause the upstroke of the action potential (phase 0 in nerve/skeletal/cardiac fast AP).
Inactivated rapidly (gives the refractory period).
Blocked by tetrodotoxin (TTX), lidocaine (local anaesthetics).
Reverse mode action of NCX:
Normal mode (forward):
Moves 3 Na⁺ in / 1 Ca²⁺ out → extrudes Ca²⁺ from cell.
Helps relax muscles after contraction.
Driven by the Na+⁺ electrochemical gradient (Na+⁺ higher outside).
Reverse mode:
It can operate backwards (3 Na⁺ out / 1 Ca²⁺ in) when intracellular Na⁺ is high or during strong depolarisation.
Seen in ischemia or digitalis toxicity (because Na⁺/K⁺-ATPase is blocked → ↑ intracellular Na⁺).
This brings Ca²⁺ into the cell → may increase contractility but can also cause Ca²⁺ overload.
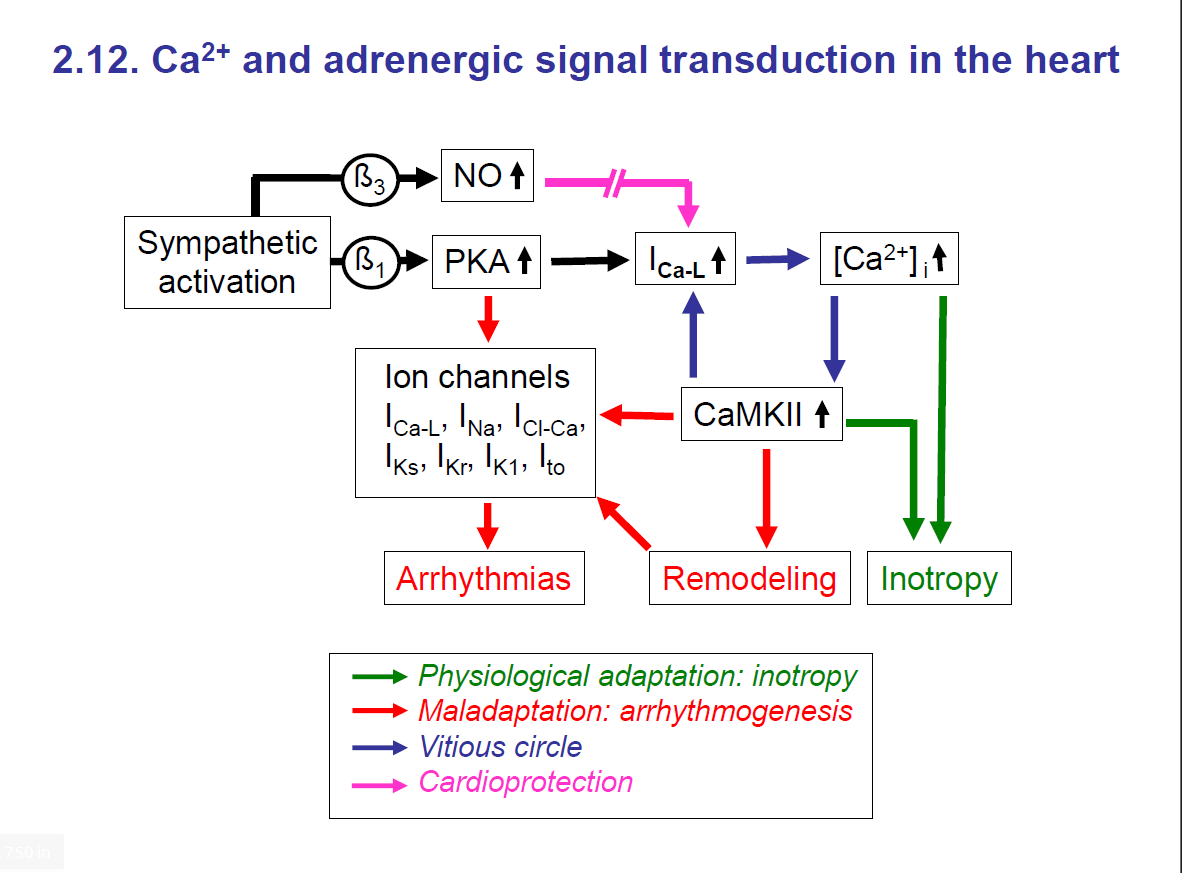
Compartmentalisation
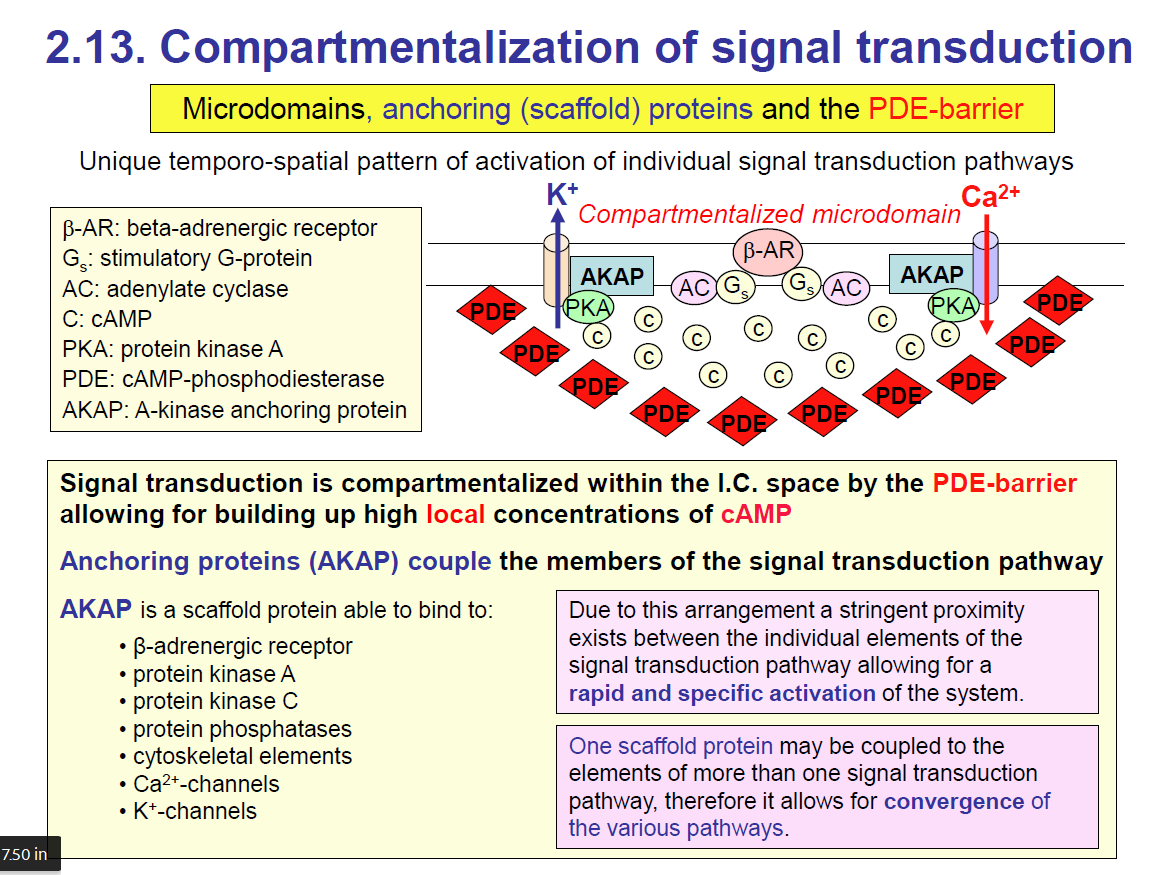
🔑 Big Idea of the Slide
Cells don’t want cAMP signalling to just spread everywhere in the cytoplasm — it would be too messy and unspecific. Instead, they compartmentalise signalling into microdomains, using scaffold proteins (AKAPs) and enzymes (PDEs) to keep everything localised and controlled.
Simply remember: Compartmentalisation keeps the signal localised in one area. An example of compartmentalisation is the PDE wall. It surrounds the signalling pathway. The PDEs break down cAMP outside the microdomain so cAMP stays concentrated only where it is needed
Key Components on the Slide
β-AR (β-adrenergic receptor): GPCR that activates Gs → stimulates adenylate cyclase (AC) → makes cAMP.
cAMP: Second messenger.
PKA (protein kinase A): Main effector of cAMP.
PDE (phosphodiesterase): Breaks down cAMP → prevents it from spreading everywhere.
AKAP (A-kinase anchoring protein): A Scaffold protein that anchors PKA and other signalling molecules close to the receptor.
K⁺/Ca²⁺ channels: Targets for PKA phosphorylation.
How Compartmentalisation Works
Microdomains
Near the β-adrenergic receptor, adenylate cyclase makes cAMP by breaking down ATP to cAMP + phosphate.
Instead of diffusing everywhere, cAMP is trapped in a small local zone by surrounding PDEs (the “PDE-barrier”).
This creates a compartment of high cAMP concentration only around the receptor.
AKAPs as scaffolds
AKAP proteins bring together all the key players: receptor, PKA, PKC, phosphatases, and ion channels.
This ensures signaling is fast, efficient, and specific → PKA can phosphorylate the nearest channel without delay.
Advantages
Specificity: Only the correct nearby proteins are phosphorylated.
Speed: Everything is physically close, so response is rapid.
Convergence: One AKAP can bind components from multiple pathways → allows cross-talk between signaling systems.
Important Notes (from slide text)
PDE-barrier: Keeps cAMP local by degrading it outside the microdomain → prevents “cAMP flooding” of the entire cytoplasm.
Anchoring proteins (AKAPs): Bring β-AR, PKA, kinases, phosphatases, cytoskeleton, and ion channels together.
Stringent proximity: Because everything is close, activation is rapid and specific.
Convergence: One AKAP can organize multiple pathways → signaling pathways can integrate.
✅ Simplified summary for exams:
“Signal transduction is compartmentalized into microdomains by PDEs and scaffold proteins (AKAPs). This ensures high local cAMP concentrations, allowing PKA to act rapidly and specifically on nearby targets like Ca²⁺ and K⁺ channels, while avoiding uncontrolled signaling throughout the cell.”