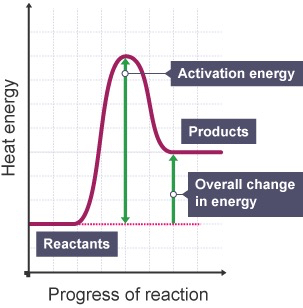
Reaction profiles/ energy level diagrams
Show energy changes of the reactants and products in a reaction
H = energy change (+ H for endothermic, - H for exo)
AE (activation energy) = minimum energy needed to start a reaction ,arrow always from reactants to the peak of products