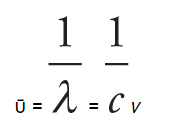Lab Techniques- Spectroscopy
5.4 Spectroscopy
Molecules normally exist in their lowest energy form, known as a ground state.
When a molecule is exposed to light, it may absorb a photon (provided that the energy of the photon matches the energy between two of the fixed electronic energy levels of the molecule)
- The absorption of a photon causes the molecule to be in an excited state
Molecules prefer to be in ground state rather an excited states, but in order for them to return to a ground state- they must lose the energy they gained
- The loss of energy can occur by heat emission or the emission of light
In absorption spectroscopy, scientists induct the absorption of energy by a sample of molecules by exposing the sample to various forms of light, thus exciting the molecule to a higher energy state. Scientists then measure the energy that is released when molecules return to a ground state.
- the measure of energy release can reveal structural features of the molecule within a sample
Different forms of light induce different transitions in ground state molecules to different excited states of the molecules and allow for the acquisition of different structural information about the molecules.
Mass Spectrometry
Allows researchers to determine the mass of compounds within a sample.
How it works:
molecules are ionized in a high vacuum, usually by bombarding them with high energy electrons..
once ionized, compounds enter a region of the spectrometer where they are acted on by magnetic field
the field causes the flight path of the charged species to alter, and the degree to which the path is changed is determined by the mass of the ion. This difference is detected and translated into a mass readout in the detector.
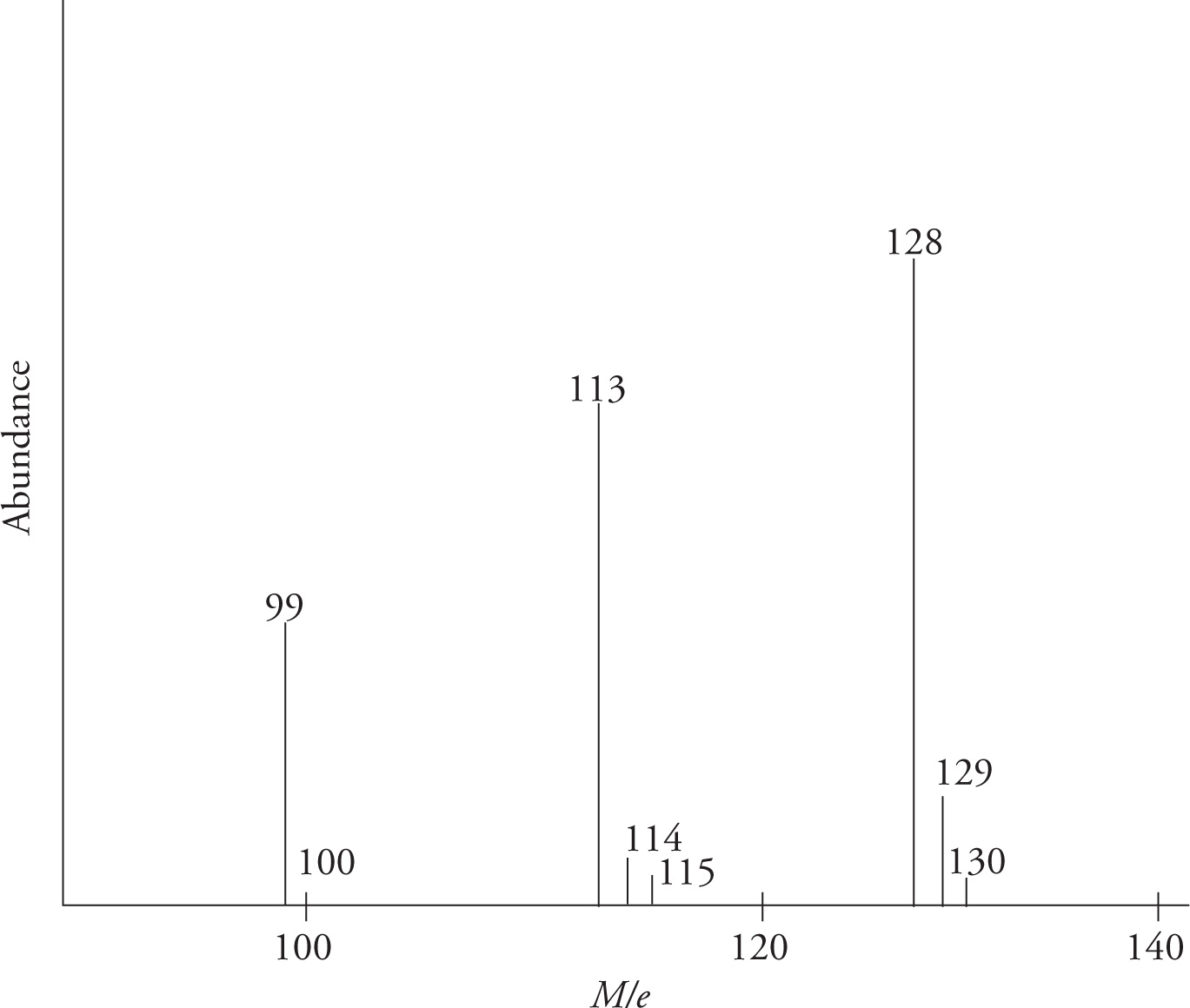
Reading mass spectrum type A
- Axis
- Y axis: abundance (typically in percentage)
- X axis: ratio of mass (M) to charge (e or z)
- most cases, e=+1
- peaks could be viewed as molecular mass
- Values
- The value of 128 (or the value that has the highest abundance) is the molecular weight of molecule
- Values >128 (or small peaks that are larger than typical molecular weight)= masses of other isotopes
- Values <128 (peaks that are less than typical molecule weight)= masses of molecular fragments
- Ex: the 113 value represents the loss of terminal CH3 group (MW CH3=15, 128-15=113)
- Ex: The 99 value represents the loss of CH2CH3 group
- Bromine and Chlorine
- both elements have two naturally occurring isotopes
- Bromine (79 and 81): will have two major peaks equal in height and approx. 2amu apart
- Chlorine (35 and 37): will have a peak 2amu heavier than the main peak about about 1/3 its height
- 35 has 75% natural abundance
- 37 has 25% natural abundance
Reading mass spectrum type B
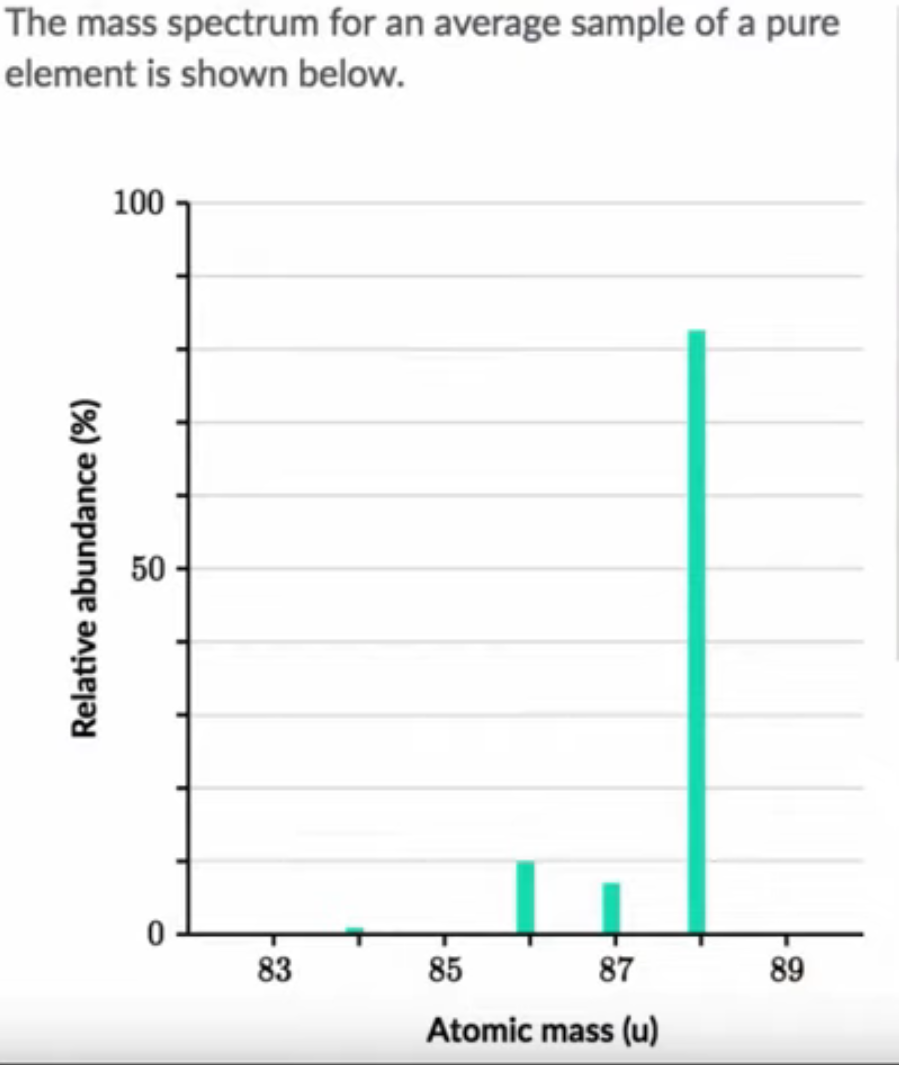
- Most abundant isotope above for (ELEMENT)
- Abundant type in picture above is 88 atomic mass (no mass/charge ratio here)
- Could either do math by adding together all masses* %abundance
- Or look for an element on the periodic table that is a little below 88
- this is due to other elements being present that are lower in mass (mostly between 86-88)
- compare masses on the periodic table to identify the unknown element at SR (AM=87.62)
Ultraviolet/Visible (UV/Vis) Spectroscopy
A type of absorption spectroscopy that is similar to IR, but instead focuses on the slightly shorter, more energetic wavelengths of radiation in the ultraviolent and visible area of the spectrum.
Used to monitor complexes of transition metals and to study highly conjugated organic systems
- The promotion of electrons from ground to excited states in closely spaced d-orbitals of many transition metals gives them their bright color (by absorbing wavelengths in the visible regions) and since many of these promotions involve energies in the UV range, these promotions allow study of these species
- when molecules have conjugated pi-systems, orbitals form many bonding, non-bonding, and anti-bonding orbitals. These orbitals can be reasonable close together in energy, and allow promotion of electrons between electronic states through absorption of UV or visible photons
- The more extensive the conjugated system is, the longer the wavelength of maximum absorption will be
- I.e addition of an aromatic ring causes the conjugated system to be long and increases the maximum absorption wavelength
The wavelength maximums correlate to a particular color of light
- 800nm-650nm= red
- 650-600nm= orange
- 600-560nm= yellow
- 560-490nm= green
- 490-430nm= blue
- 430-400nm= purple
If a molecule absorbs a certain color, it will appear visibly as the absorption’s complementary color
- if a molecule absorbs red, it will visibly appear as green
If a molecule absorbs only UV radiation, ALL visible wavelengths will be reflected- thus the compound will appear white or colorless
Infrared (IR) Spectroscopy
Electromagnetic radiation in the infrared range λ = 2.5 to 20 µm has the proper energy to cause bonds in organic moleculars to become vibrationally excited. When a sample of an organic compound is irradiated with inrared radition in the region between 2.5 and 20µm, its covalent bonds will begin to vibrate at distinct energy levels (wavelengths, frequencies) within the region. These wavelengths correspond to frequencies in the range of 1.5 × 1013 Hz to 1.2 × 1014 Hz.
In IR spectroscopy, vibrational frequencies are more commonly given in terms of the wavenumber.
The wavenumber (ῡ) is simple the reciprocal of wavelength:
- Wavenumber is directly proportional to both frequency and energy of radiation.
- The high the wavenumber, the higher the frequency, and the greater the energy
- typically expressed in reciprocal centimeters, cm−1
- For the MCAT IR spectra will typically range between 4000-1000cm−1
Radiation source
When a bond absorbs IR radiation of a specific frequency, that frequency is not recorded by the detector and is thus seen as a peak in the IR spectrum (since low transmittance corresponds, naturally, to absorbance)
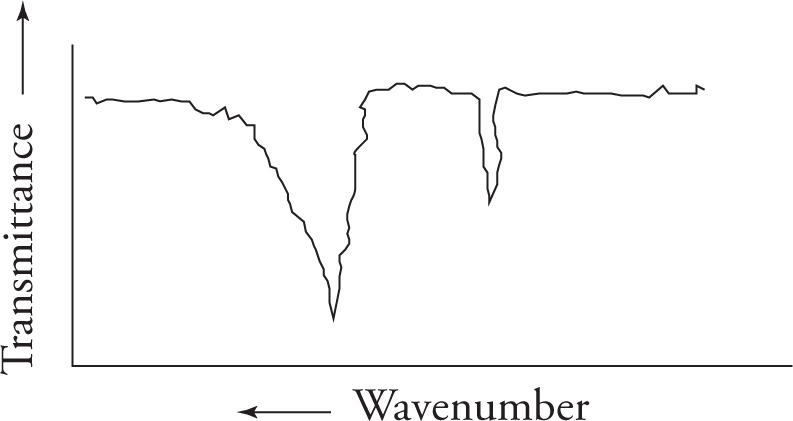
Important stretching frequencies
Double bond stretches
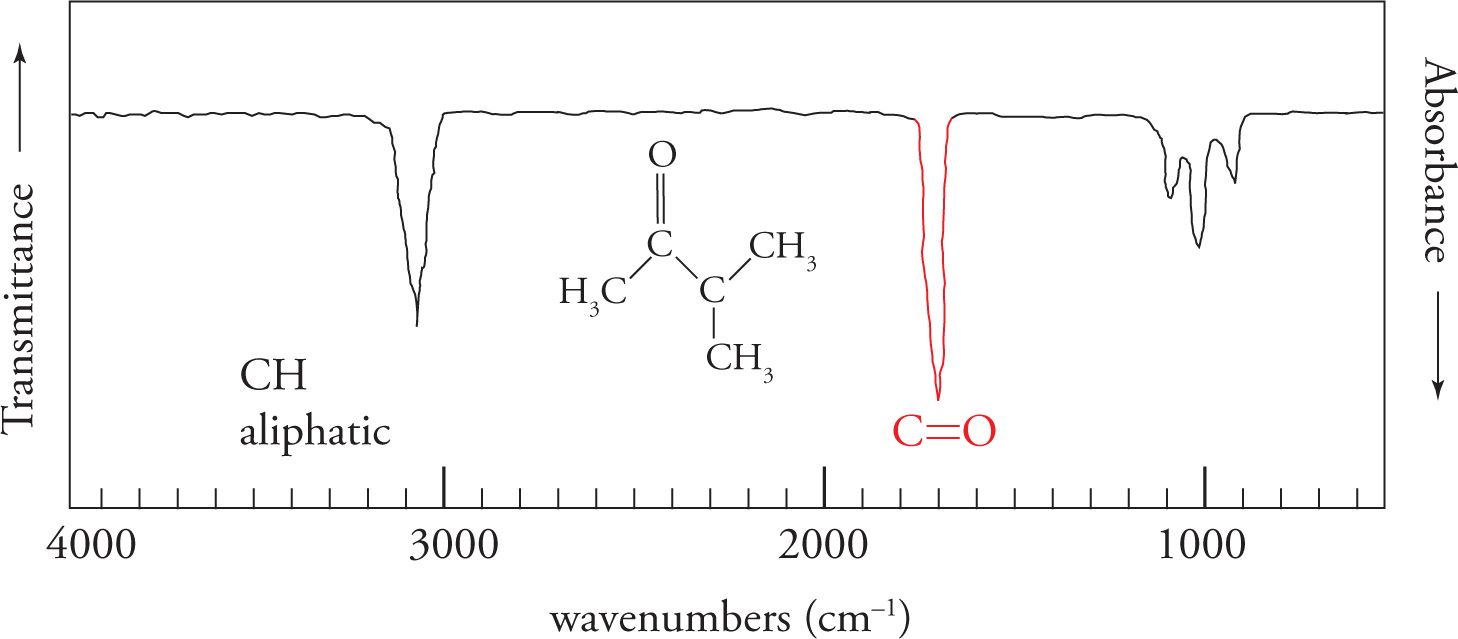
carbonyl/ C=O stretch
- centered around 1700cm-1
- very strong (%transmittance) and intense (sharpness/distinctiveness of spike in ‘V’ shape)
- always look for this stretch first (can eliminate many compounds that contain carbonyl groups if not present like aldehydes, ketones, carboxylic acids, acid chlorides, esters, amides, and anhydrides)
Alkene center/C=C stretch
- centered around 1650cm-1
Triple bond stretches
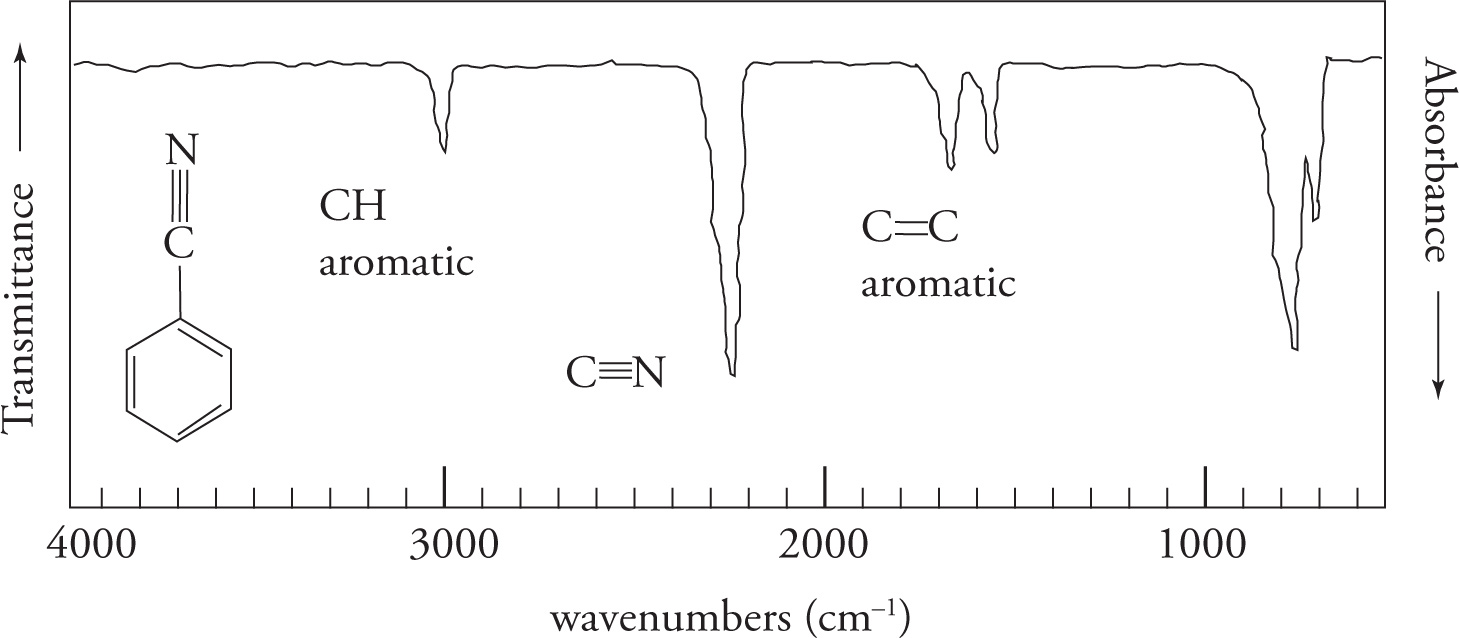
- C triple bonded to C
- centered between 2260-2100cm-1
- C triple bonded to N
- 2260-2100cm-1
Hydroxyl/O-H stretch
- strong and broad (makes a ‘U’ shape due to hydrogen bonding)
- centered between 3600-3200cm-1
C-H stretches
C-H sp3= 3000-2850cm-1
C-H sp2= 3150-3000 cm-1
C-H sp= 3300cm-1
Aromatic CH bonds >3000cm-1
1H NMR
Commonly called proton NMR, this absorption spectroscopy uses light from the radio frequency range of the electromagnetic spectrum to induce energy absorptions.
4 essential molecule features can be deduced from its 1H NMR spectrum (MCAT will only focus on first 2)
- number of sets of peaks within the spectrum tells the number of chemically nonequivalent sets of protons within the molecule
- the splitting pattern of each set of peaks tells us how many protons are interacting with the protons within that set
- the mathematical integration of the sets of peaks indicates the relative numbers of protons in each set
- the chemical shift values of those sets of peaks give information about the environment of the protons in that set
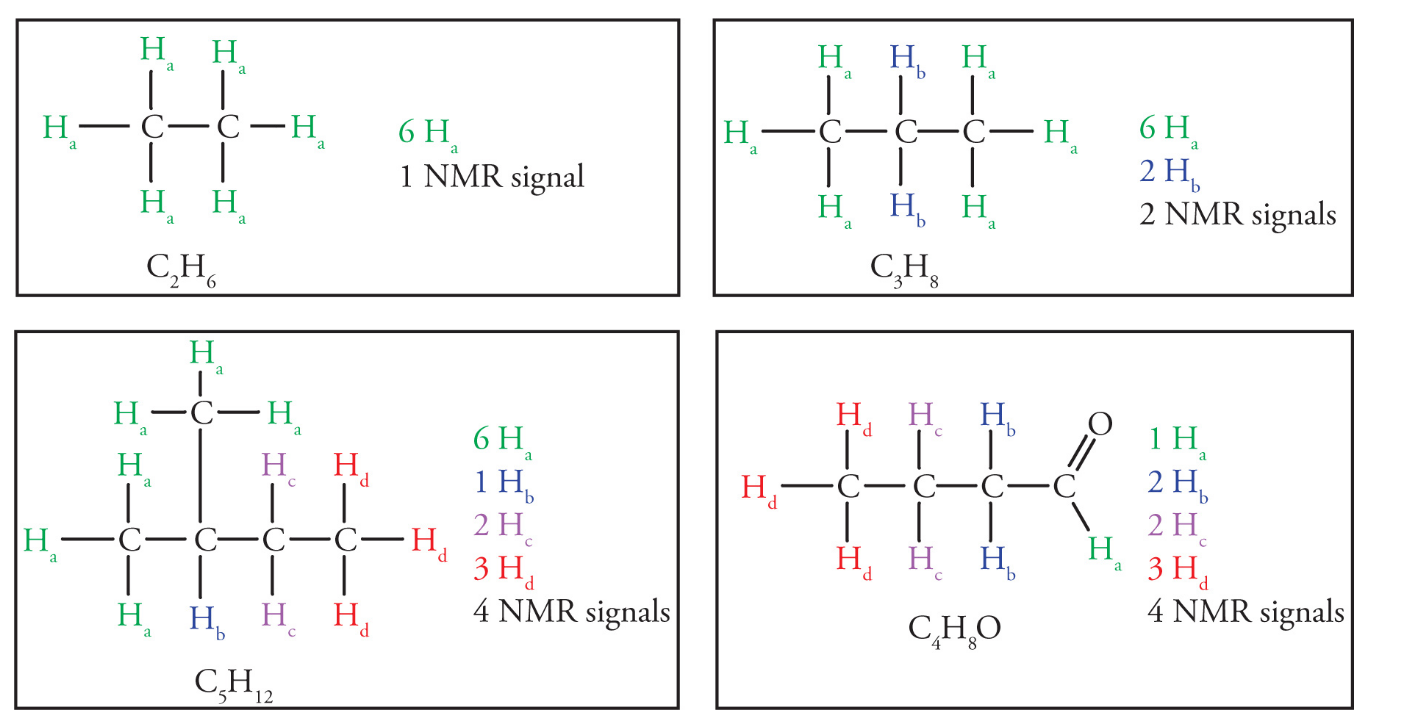
Chemically equivalent hydrogens
Equivalent hydrogens in a molecule are those that have identical electronic environments and will have identical locations within the 1H NMR spectrum
- therefore they are represented by the same signal or resonance
Nonequivalent hydrogens will have different locations within the spectrum and be represented by different signals
Hydrogens are considered equivalent if they can be interchanged by a free rotation or a symmetry operation (mirror plane or rotational axis)
Splitting/ spin-spin splitting phenomenon
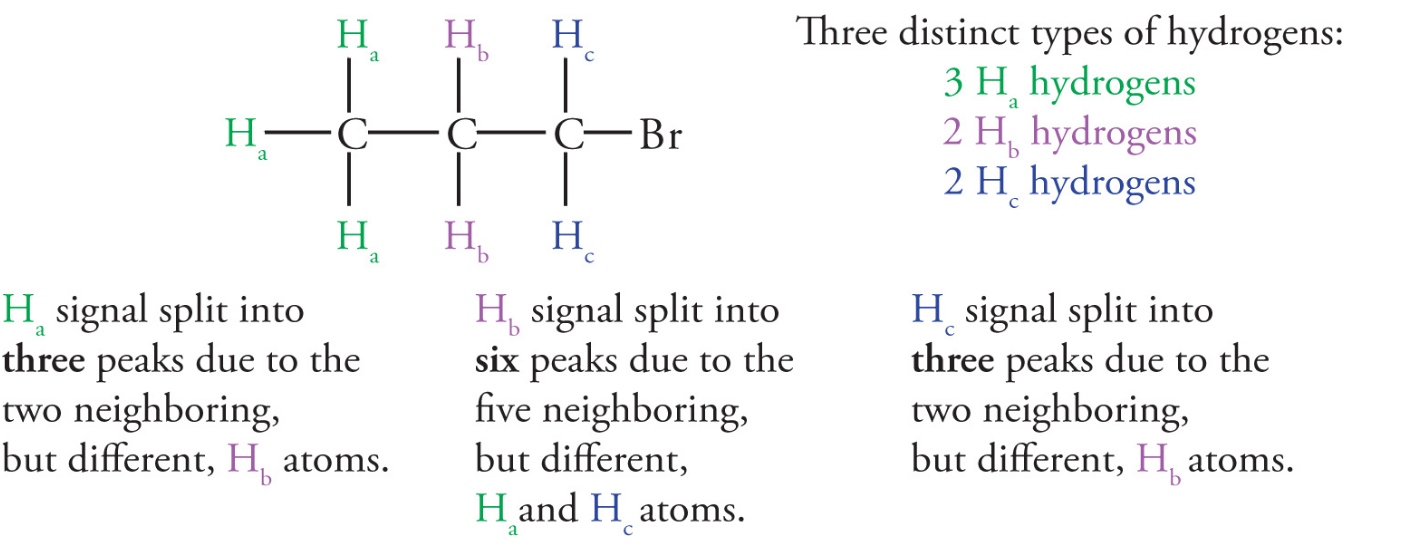
The spin-spin splitting phenomenon occurs when nonequivalent hydrogens interact with each other
- this exists because the magnetic field felt by a proton is influenced by surrounding protons
- effect falls off with distance but can often extend over two adjacent carbons
Nearby protons that are nonequivalent to the proton in question will cause a splitting in observed 1H NMR signal
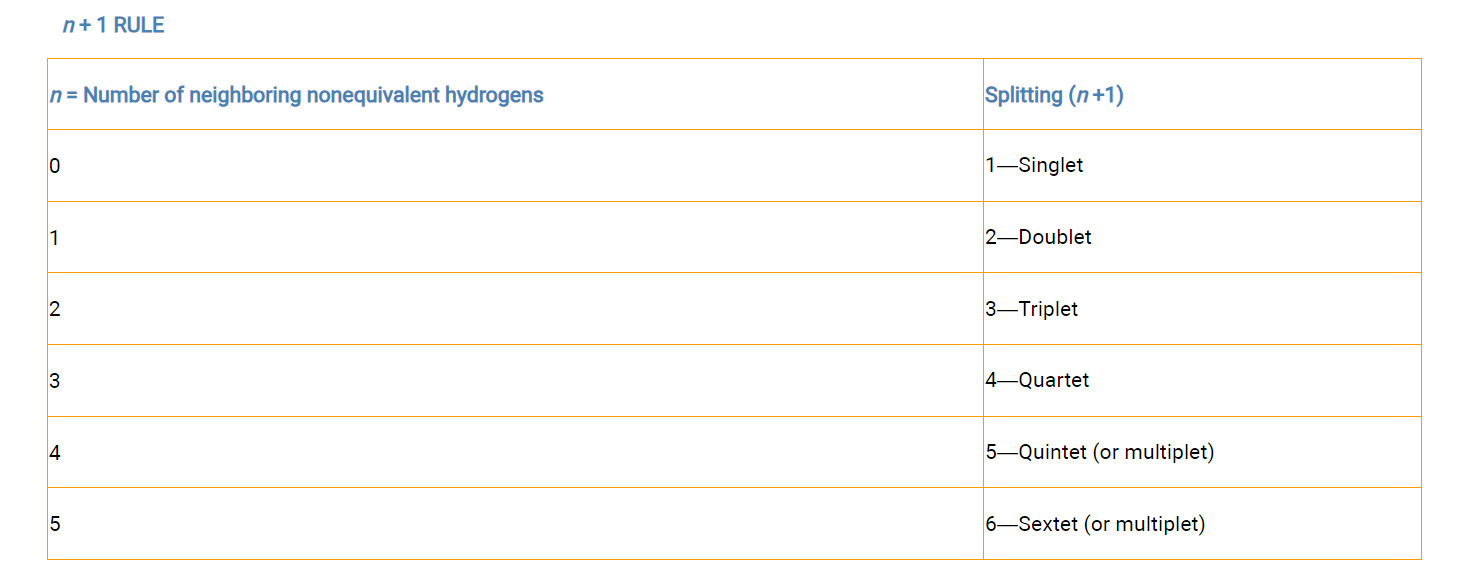
degree of splitting depends on the numbers of adjacent hydrogens
the signal will be split into n+1 lines, where n is the number of nonequivalent, neighboring (interacting) protons
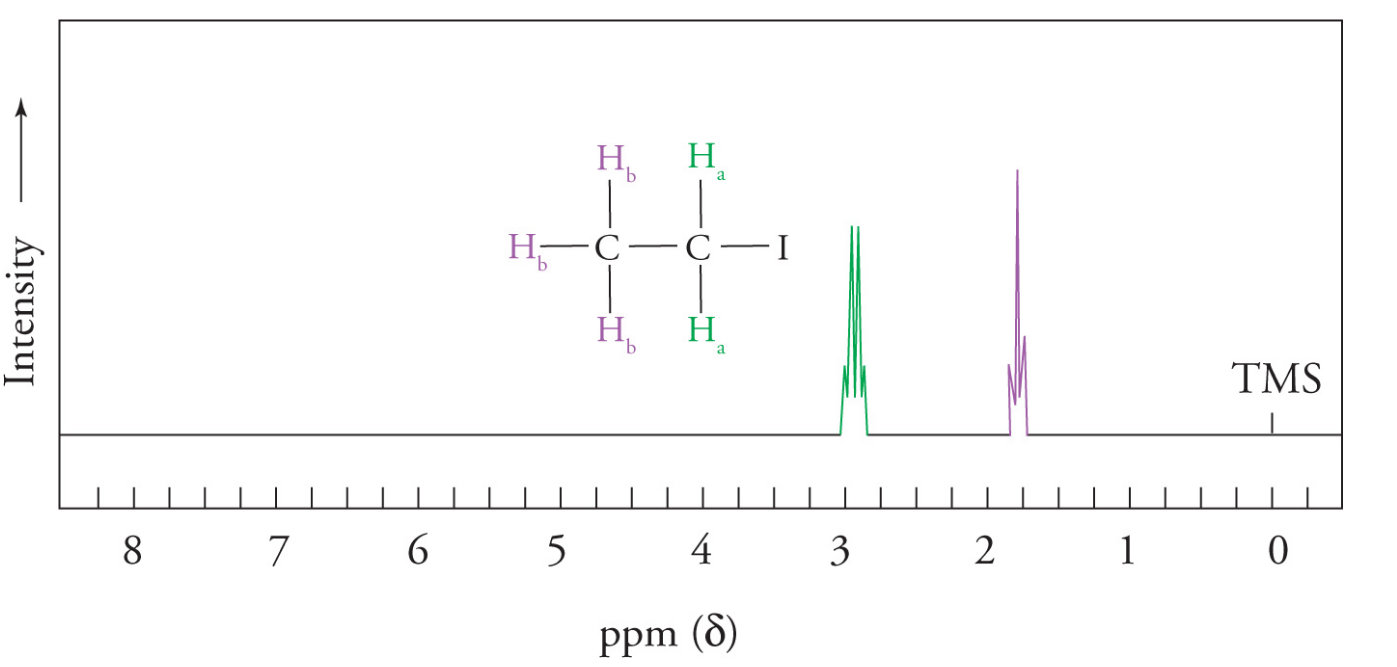
Integration
As the NMR instrument obtains a spectrum of the sample, it performs a mathematical calculation called an integration, thereby measuring the area under each absorption peak (resonance)
The calculated area under each peak is proportional to the relative number of protons giving rise to each peak. Thus, the integration indicates the relative number of protons in each set in the molecule
Chemical shifts
Chemical shift indicates the location of the resonance (the set of peaks) in the 1H NMR spectrum. Differences in the chemical shift values for different sets of protons in a molecule are the result of the differing electronic environments that different sets of protons experience
The magnetic field created by electrons near a proton will shield the nucleus from the applied magnetic field created by the instrument, shifting the resonance upfield
The more a proton is deshielded (the more distorted away from the atom the electron cloud is), the further downfield in an NMR spectrum it will appear
Downfield (to the left) = more deshielded
Upfield (to the right)= less deshielded
Factors that are involved in proton deshielding include:
electronegativity of neighboring atons
- if an electronegative atom is in close proximity to a proton, it will deshield it
hybridization
- the greater the s-orbital character, the more deshielded the set of protons is
acidity and hydrogen bonding
- protons attached to heteroatoms (oxygen and nitrogen) are quite deshielded
- acidic protons on a carboxylic acid contain a large downfield shift
- the more hydrogen bonding is present, the more the proton will be deshielded and the higher the chemical shift will be and varies between solvent and temperature