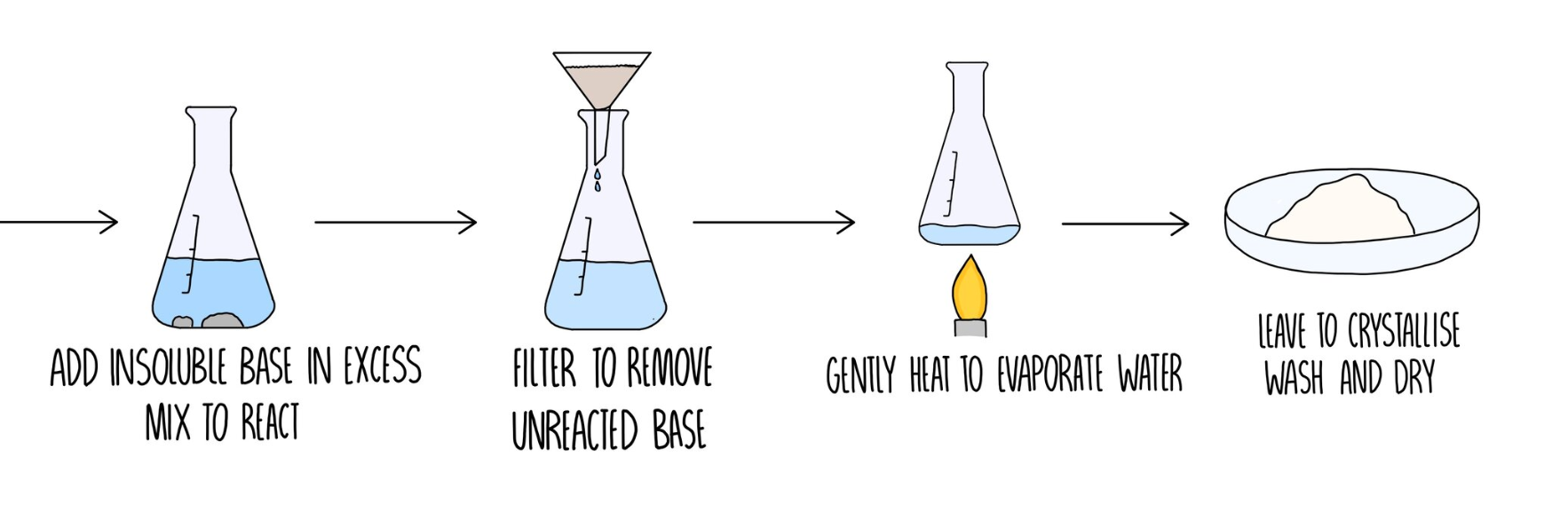
Making salts with a Soluble Base
Acid + Base → Salt + Water
if the base is soluble, do a TITRATION
- Can’t add excess base, because the base is soluble.
- Can’t filter out excess base.
- Therefore, you must add the base until exactly neutral.
\ The acid that you will use will depend on what salt you want:
- Hydrochloric acid produces chloride salts
- Sulfuric acid produces sulfate salts
- Nitric acid produces nitrate salts
\ \ Making an insoluble salt experiment
- Do the titration WITH an indicator (to find the volume of the base)
- Repeat with NO indicator (left with only base + water)
- Evaporate about half of the solution - until a crystal forms on the end of a glass rod.
- Turn of the heat and let the solution cool.
- Filter off the crystals from the solution.
\