Solvent Extraction
Overview
- Isolates and concentrates the products
- Partially purifies the product by removing similar chemical species
- Based on the solubility differences of the compound in one phase relative to the other
- Transfer of a solute from one phase to another
- liquid-liquid
- solid-liquid
- Solute is distributed or partitioned between the solvents
- Increase in solute in one phase
- Depletion of solute from the second phase
Ideal Solvent
- Nontoxic
- Selective
- Inexpensive
- Immiscible with feed
Equations
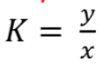
- K = distribution coefficient or partition coefficient
- y = concentration of solute or product in solvent
- x = concentration of solute or product in raffinate
- residual feed after extraction with solvent
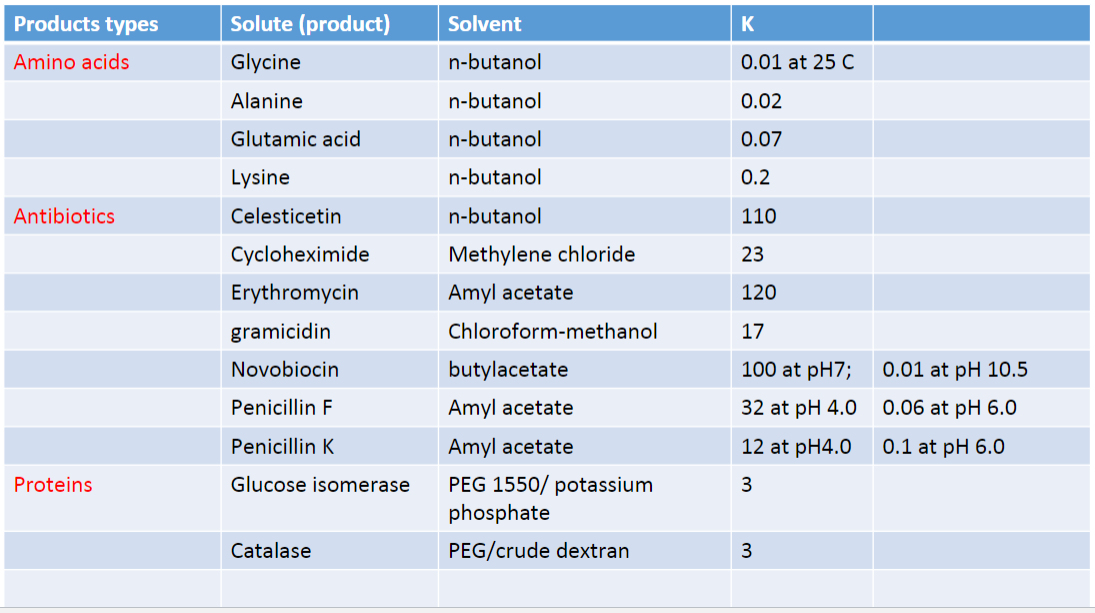
Partition Coefficient K
- based on partitioning of solute between two immiscible solvents
- citric acid is more soluble in methyl amyl ketone than H20 at pH 4.0
- Penicillin dissolved readily in amyl acetate than H20 at pH 5.5
- Catalase has higher conc. in polyethylene glycol-rich solution that dextran-rich solution
- for lowest volume of extract to be used, K must be very large
- If K <= 1
- large volumes of solvent and multiple extraction will be required to recover the product
- If K = 0
- extraction is impossible
Factors
- molecular size of solute
- pH
- types of solvent
- temperature
- For aqueous two-phase extraction:
- concentration of polymers
- molecular weight of polymers
Determining best solvent
- no reliable thermodynamic theory to predict the best choice of solvent
- rely on solubility parameters
Examples
- Change solute ion pairs
- Lactic acid (hydroxypropionic acid, (CH3CHOHCOOH) production
- Convert lactic acid to calcium lactate which is insoluble in water and therefore can be easily recovered
- Change in solute pH
- eg Novobiocin at pH 7
- the K value in butylacetate is 100
- at pH 10.5 the K value is 0.01
Batch Extraction
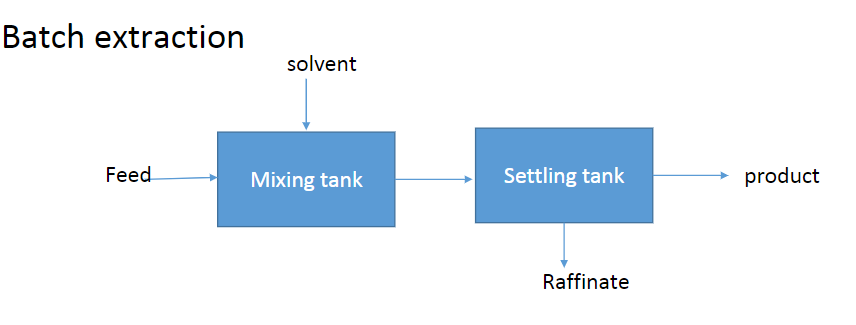
Equations

 Steps
Steps
- Perform solute mass balance around the extractor
- Assume solvent and feed are immiscible
- Equilibrium
- Dilute solution
Continuous Extraction
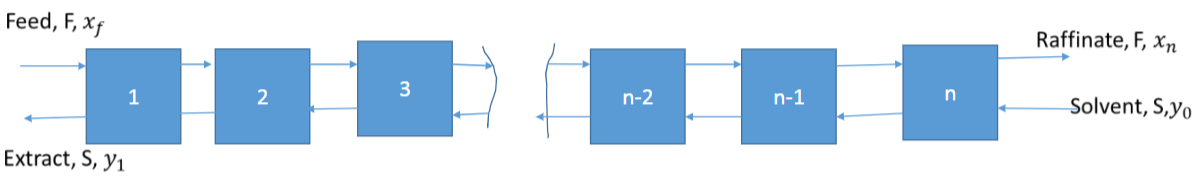
Assumptions
- The solvent and feed are immiscible
- The solute concentration is sufficiently low that the flowrates of raffinate and extract are constant
- The streams leaving each stage are in equilibrium
- The second assumption is true if the concentrations of the bioproducts are low eg 10 g/l but often it is less than 1 g/l
Design of solvent extractors
- In the chemical industry there are several types of extractors, but in \n the biotechnology industry there are only a few extractors. The two \n common extractors used in the biotech industry are: \n • Agitated extraction columns (reciprocating-plate extraction column) \n • Centrifugal extractors (used mostly for antibiotics extraction) \n • For column extraction design, the Height Equivalent to Theoretical \n stage (HETS) is commonly used or the overall stage efficiency is used. \n • HETS = (height of extractor)/(number of theoretical stages (n)) \n • n = {Ln[xf/xn(E-1)+1]/[Ln(E)]} -1