M3L4 Epithelial-stromal crosstalk in cancer
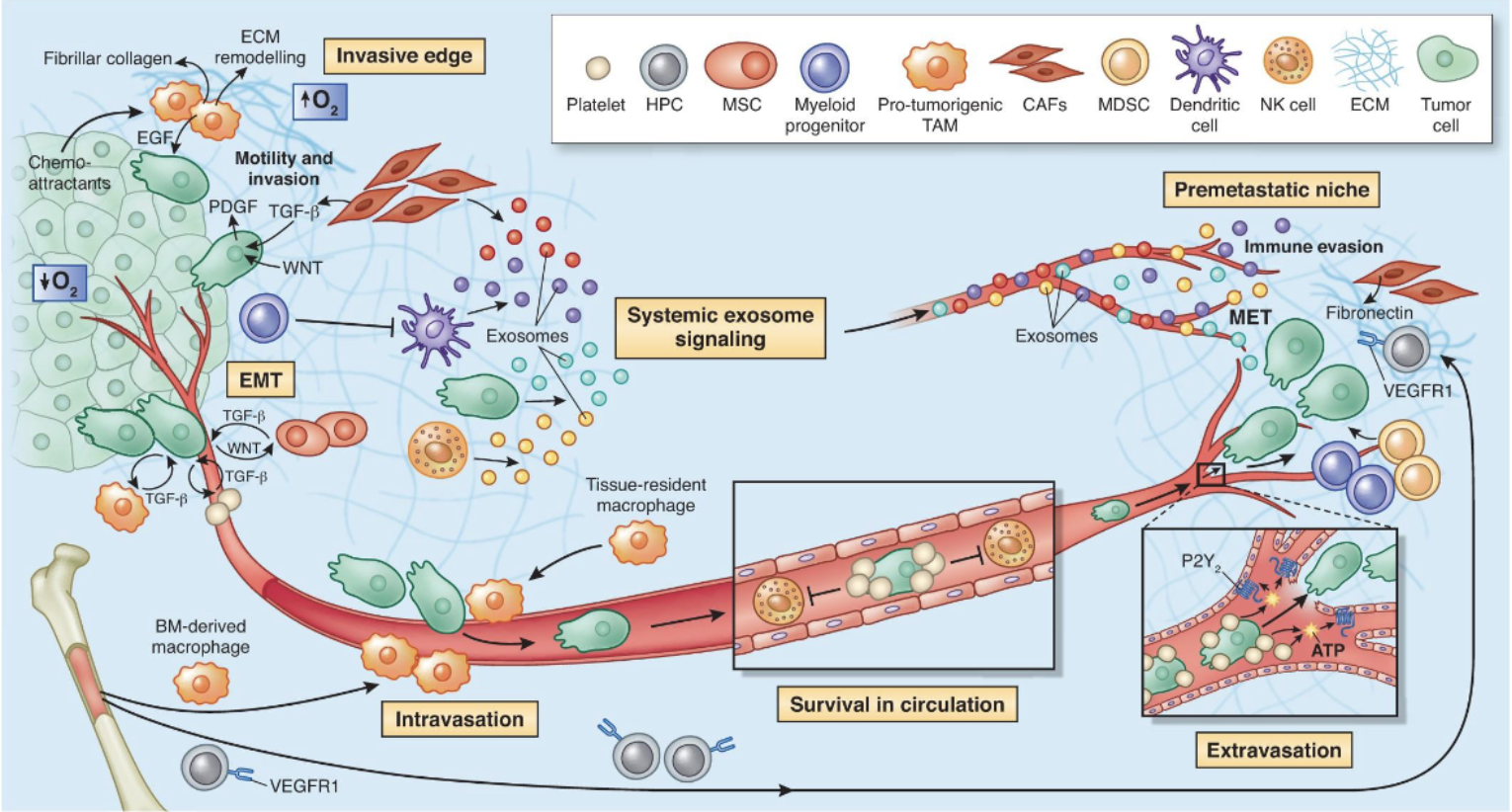
Microenvironment promotes dissemination and colonisation\
Cancer associated fibroblast (CAF)
Mesenchymal (-ve for epithelial, endothelial, leukocyte markers)
No cancer mutations
Elongated shape
Different types of CAFs
May may depend of their origin
Differ in responsiveness to growth factors and drivers of CAF phenotype
Multiple functions
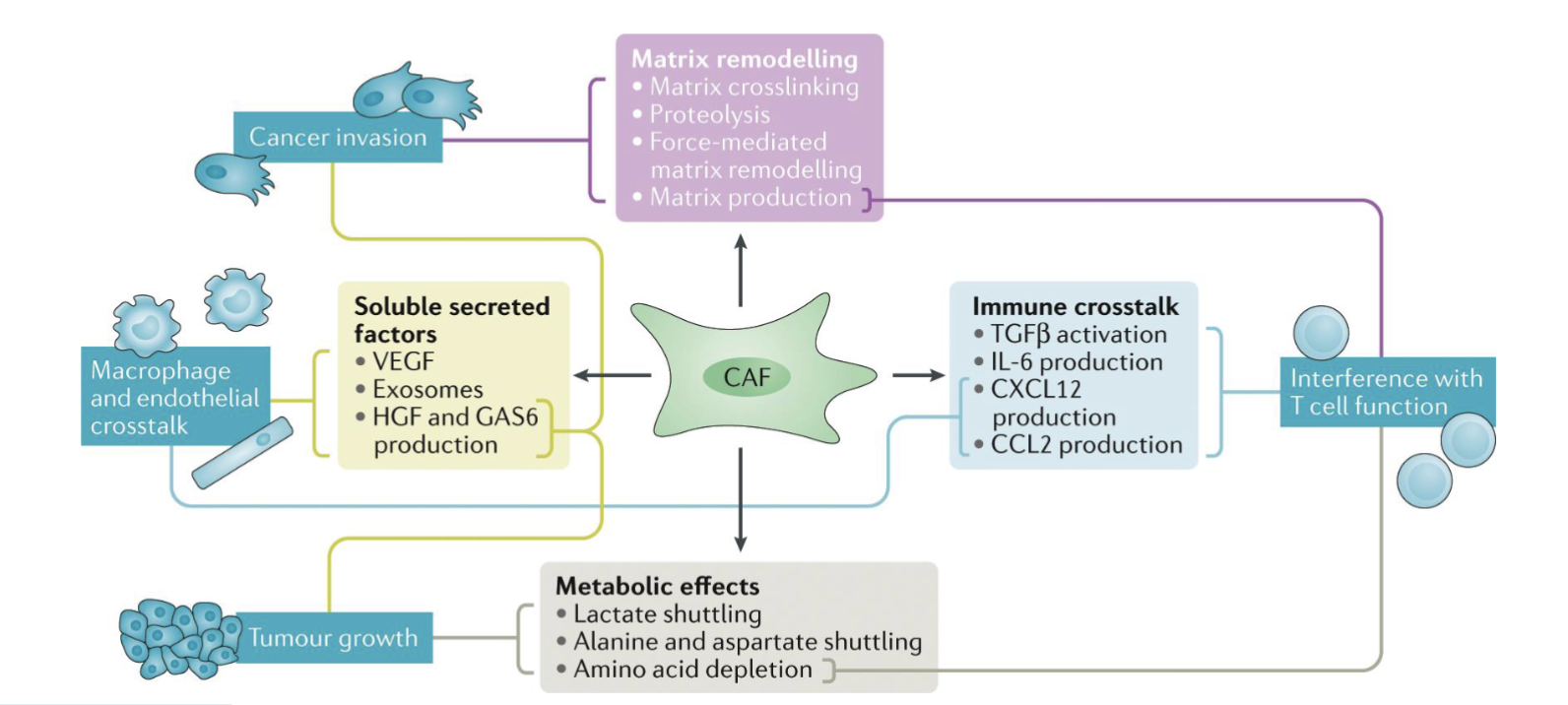
CAFs promote malignant transformation
Loss of TGFb2 receptor in CAFs can drive prostate cancer progression
CAFs ‘lead the way’ for tumour cells
Remodeling the ECM, making it easier for cancer cells to move through
Some CAFs are irreversibly altered, while others are reversible
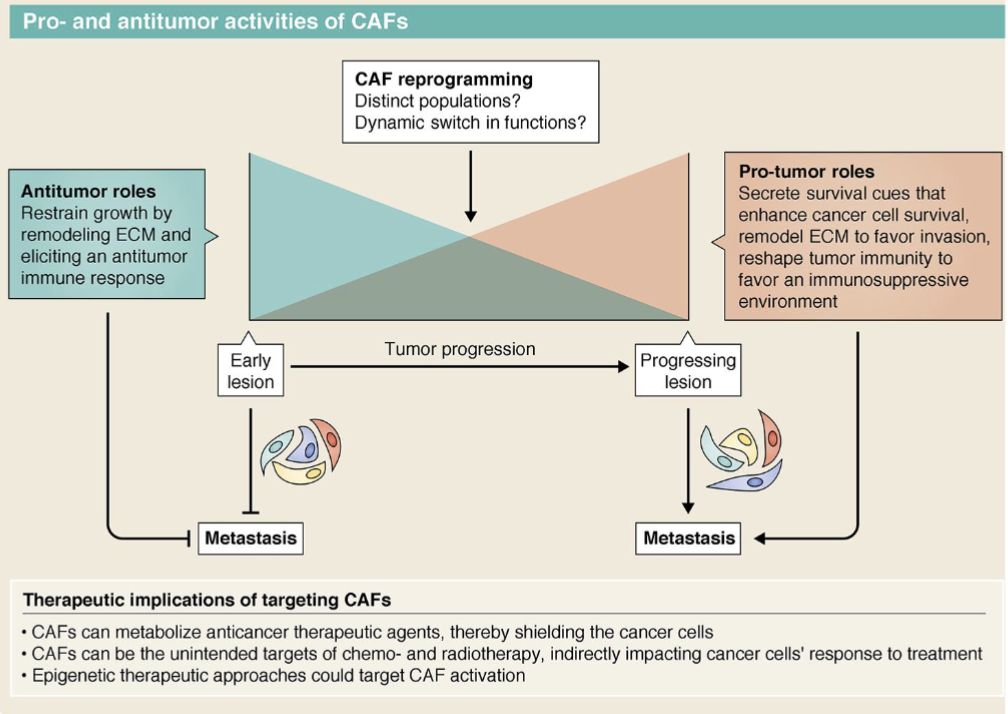
At early stages of disease CAFs can activate the immune system and suppress cancer, though mostly afterwards they take on a pro-tumour role
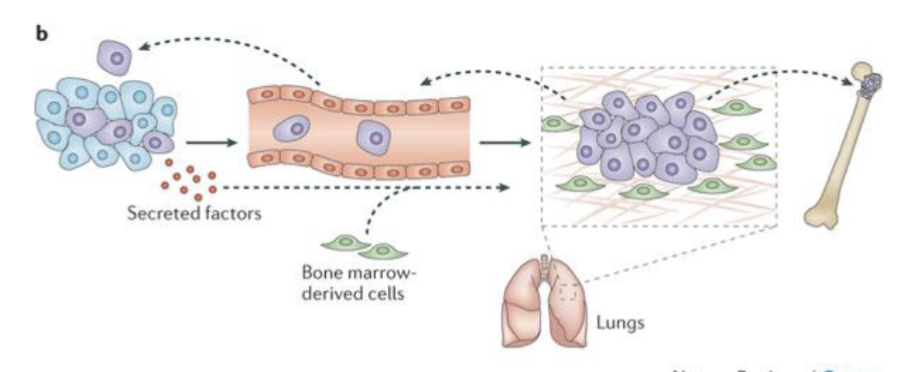
Pre-metastatic niche - cancer cells can release factors to prime the pre-metastatic site to make it more favourable for establishing metastases
Drug resistance - site of the stromal cells affect drug resistance
Most studies show that stromal cells promote drug resistance
Stromal-induced EMT induces drug resistance
Cancer cells express bone specific markers (osteomimicry) to resist therapy, possibly to disguise as a body cell
BMP signalling enriched in stromal progenitors in transcriptomics of cell populations in MM bone microenvironment
MM bone disease reversed by BMP blockade
BMP-induced prevented by BMSCs, but not seen in cancer cells cultured with stromal cells
CAFs and BMSCs promote mitochondrial transfer to cancer cells to alter metabolism via formation of tunnelling nanotubules, EVs or connexin channels
Reverse Warburg effect??
Tumour promotes bone and bone promotes tumour (??)
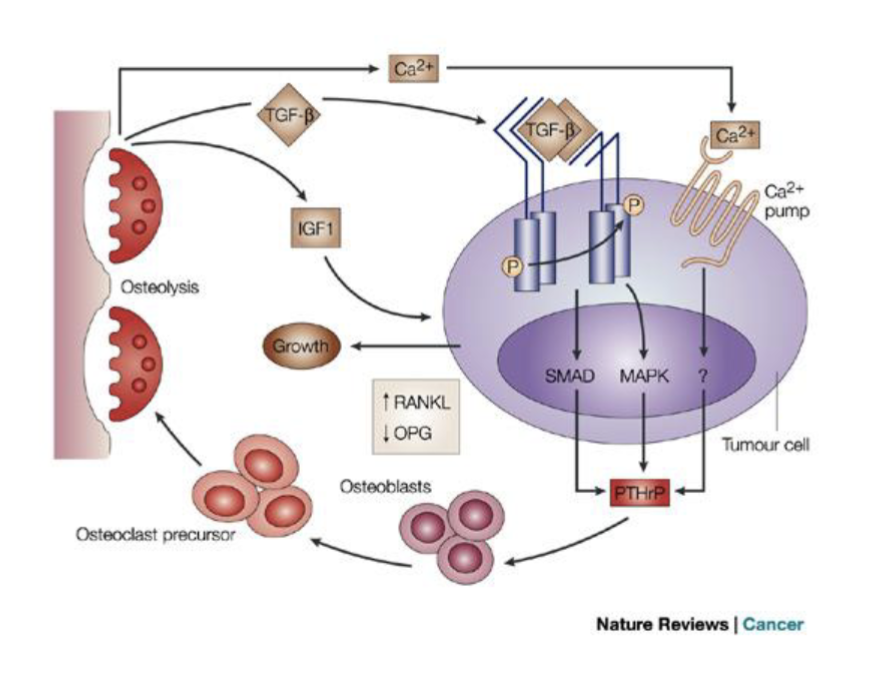
Tumour cells release factors that activate osteoclasts and osteoclasts which then activate tumour cells, mediated by RANKL
Anti-RANKL - treatment of cencer induced bone disease
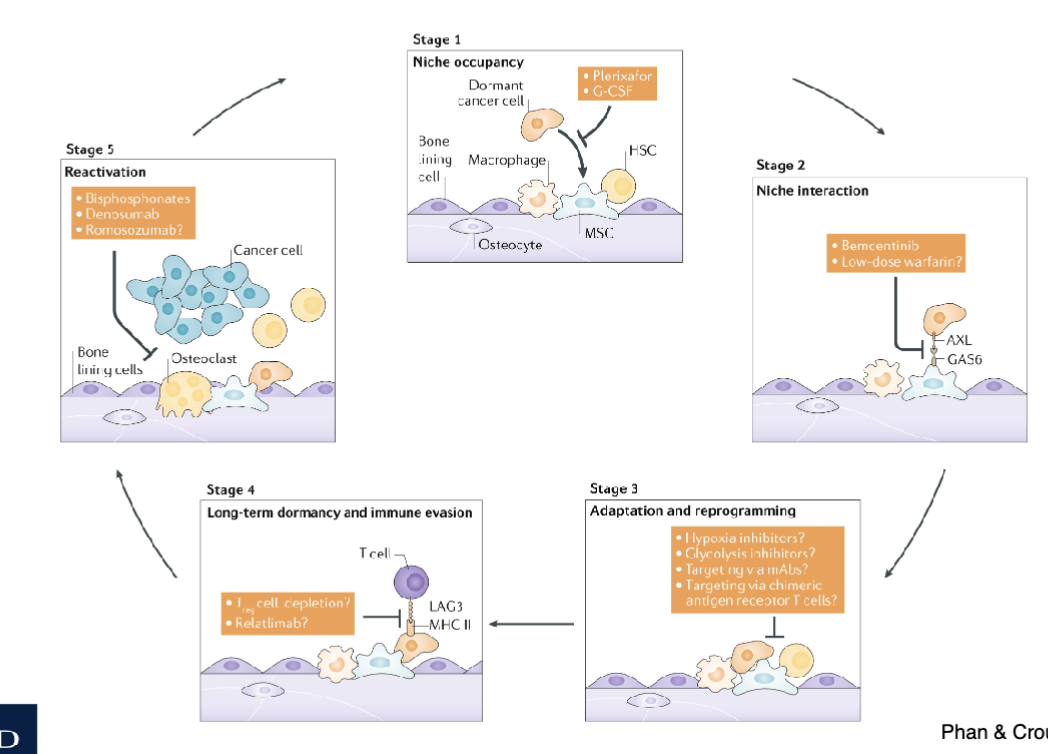
PCa cells compete with HSCs for endosteal HSC niche, increasing niche size promotes metastasis and HSC mobilisation protocols mobilise PCa cells ffrom niche to circulation (??) to target via chemo
Reactivation of dormant cells
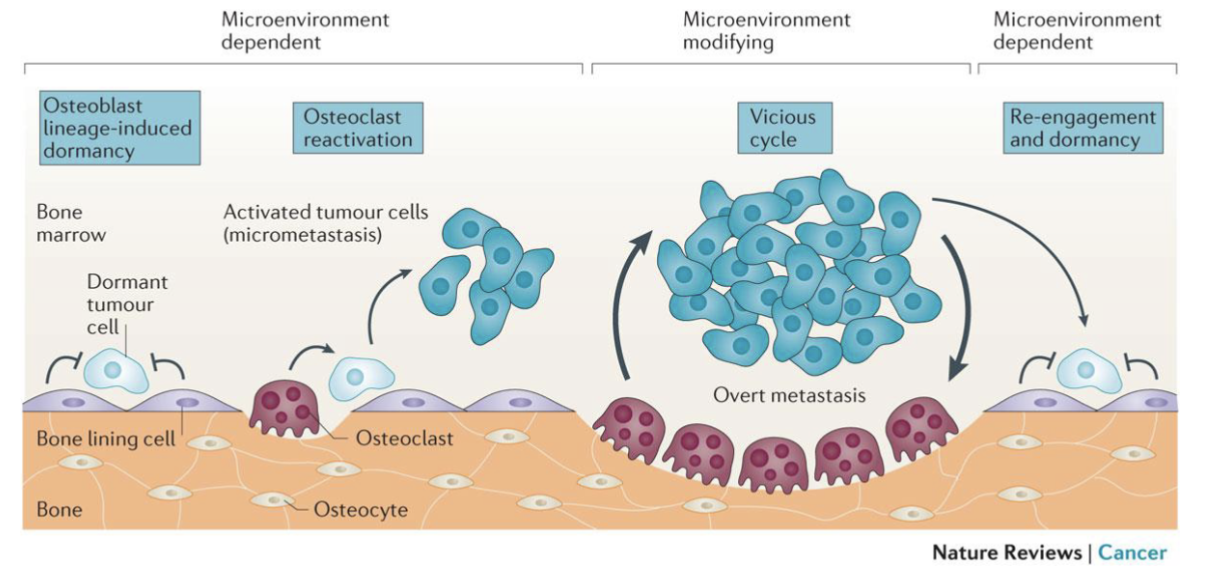
Blocking dormant niche interactions
Senescence in TME
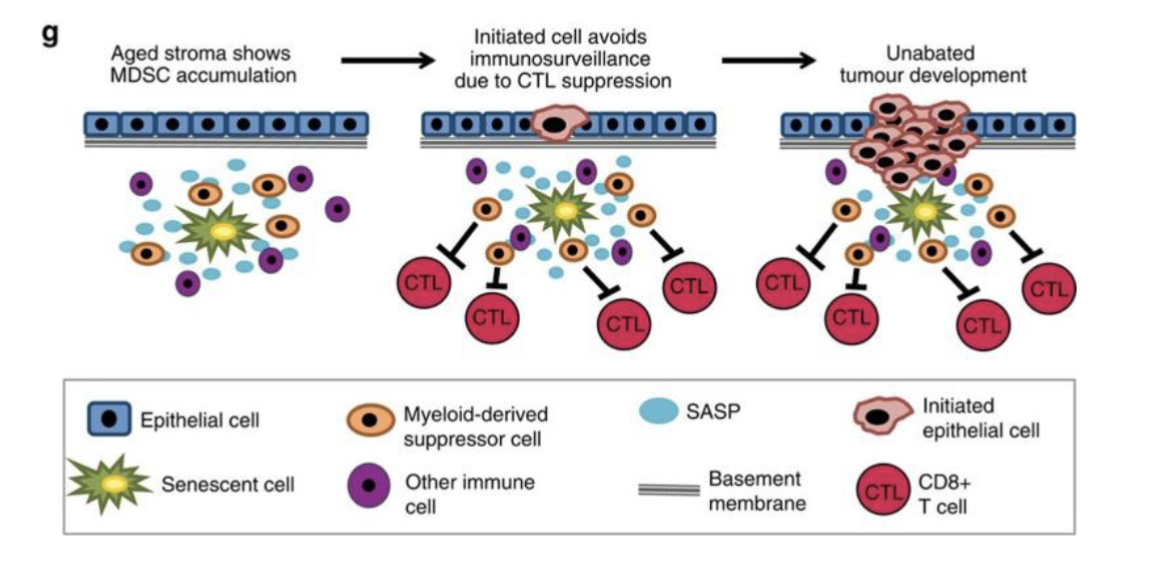
Aged stroma shows MDSC (myeloid derived suppressor cell) accumulation
Suppresses cytotoxic T cells and avoids immunosurveillance
In vitro methods - difficult to study TME
Monoculture - understanding cancer cells
Transwell - can separate cell types, understand contact/migration
Conditioned media - understanding secreted factors
Coculture - multiple cell types, understanding interactions
Organoids - better 3D architecture, cell-cell interactions modeling
In vivo -
Neghbour labelling cells
Allows stroma to be labeled by cancer cells they come into contact with
Understanding cell interactions rather than bulk populations
Intravital imaging
Imaging different sites and track over time
Barcodes can be used to label cancer cells to see which metastasised, understand the differences, which ones are proliferating, how they interact with stroma…
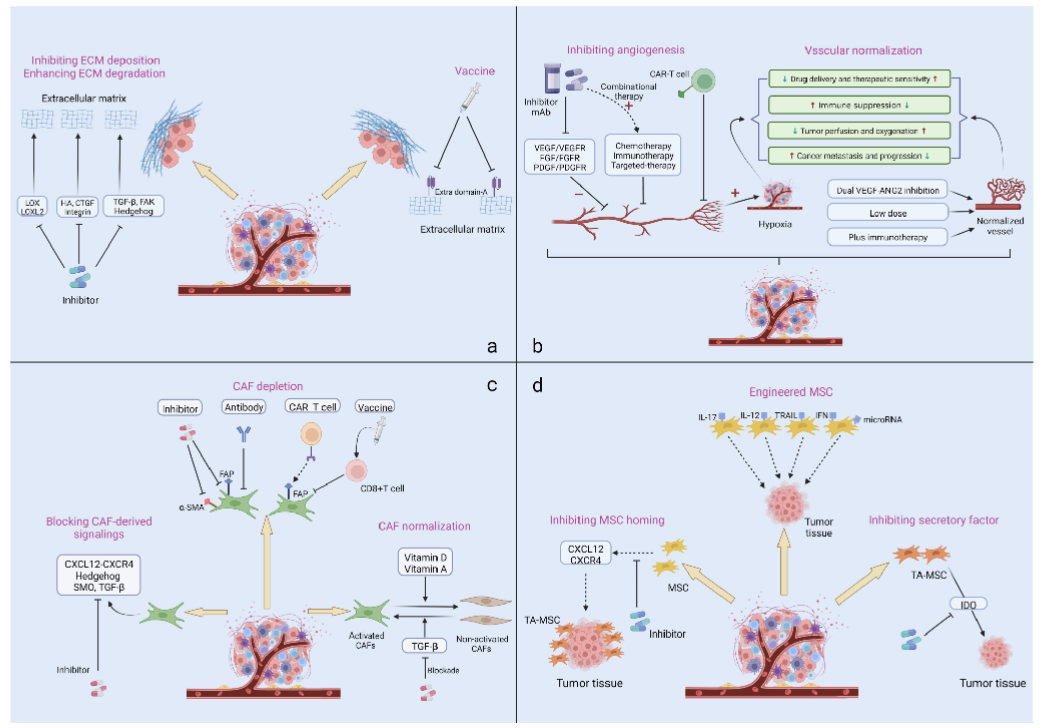
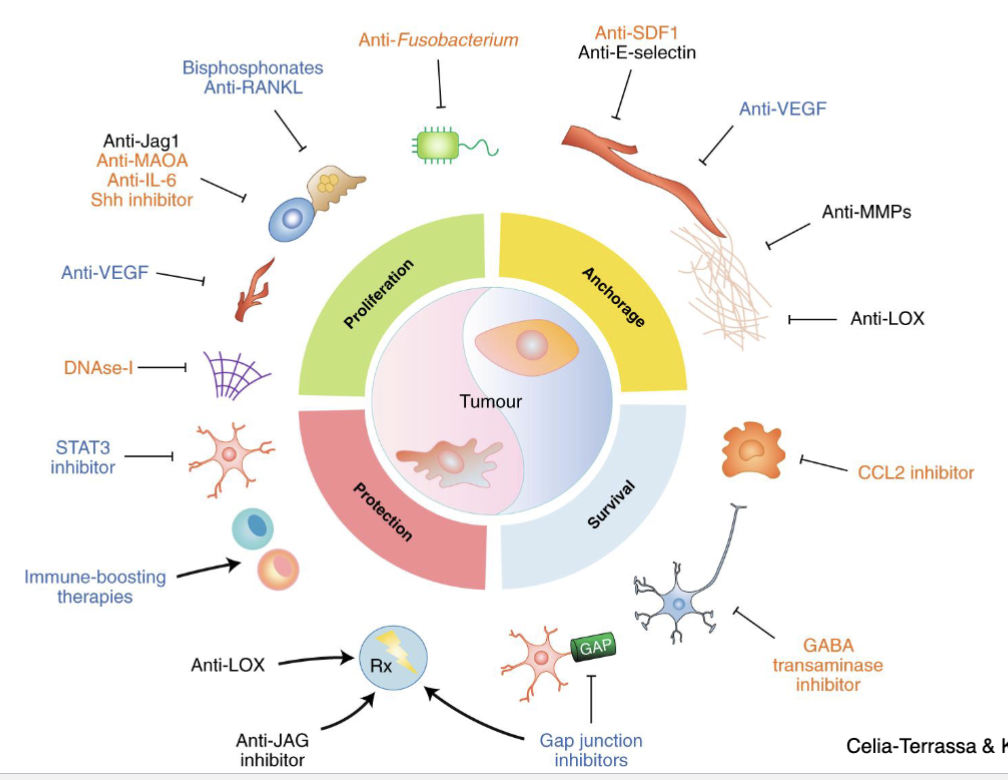
Targeting tumour-stroma
ECM
Angiogenesis
CAF depletion
Engineered mesenchymal stromal cell (MSC)