5.1: The Chemist's View of Fatty Acids and Triglycerides
Lipids (3):
triglycerides
phospholipids
sterols
characterized by: insolubility in water
fat soluble vitamins
composed of CHO (carbon, hydrogen, oxygen)
more carbons & hydrogens than oxygens
more energy per gram
Chemical Structure of Triglycerides: E
1 glycerol
3 fatty acids (chains of carbon atoms) attached
4-24 (even numbers) carbons long
18 carbon long ones are most common in foods and abundant in food supply
stearic acid: simplest of the 18 carbon fatty acids, bonds between its carbons are all alike, saturated fatty acid
saturated or unsaturated
1 or more points of unsaturation: monounsaturated or polyunsaturated
18 carbon polyunsaturated fatty acids:
omega-3 fatty acids, linolenic acid
omega-6 fatty acids, linoleic acid
essential fatty acids that the body cannot make
primary member of a family of longer chain fatty acids
regulate blood pressure, blood clotting, and other body health functions
Fatty Acids
all have the same basic structure
one end: chain of carbon & hydrogen atoms with an acid group (COOH)
other end: methyl group (CH3)
differ from one another
length of their carbon chains
number & location of their double bonds
Length of the Carbon Chain
naturally occurring fatty acids contain even numbers of carbons in their chains
up to 24 carbons in length
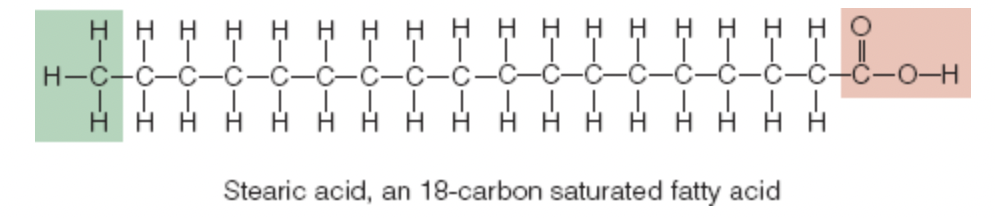
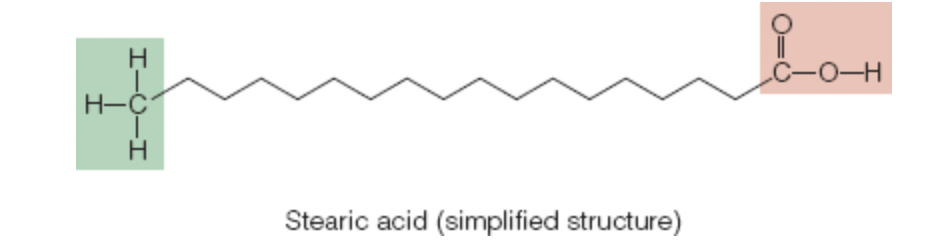
Long chain (more than 12 carbons) fatty acids:
meats, seafood, and vegetable oils most common in diet
Smaller amounts of medium chain (8-12 carbons) & short chain (up to 6 carbons) fatty acids:
dairy products
The Number of Double Bonds
saturated fatty acid:
fully loaded with hydrogen atoms
contains only single bonds between its carbon atoms
every carbon must have 4 bonds
2 carbons form 1 double bond, yielding oleic acid
point of unsaturation:
the double bond of a fatty acid
hydrogens are missing
at least 1 double bond is an unsaturated fatty acid
there is an oxygen attached to a carbon instead of a hydrogen
oleic acid:
18 carbon monounsaturated fatty acid
abundant in olive and canola oil
polyunsaturated fatty acid
2 or more carbon-to-carbon double bonds
linoleic acid
linolenic acid
The Locations of Double Bonds
fatty acids differ in:
length of their chains
degrees of saturation
locations of their double bonds
chemists identify polyunsaturated fatty acids by:
position of the double bond closest to the methyl (CH3) end of the carbon chain (omega number)
omega-3 fatty acid: a polyunsaturated fatty acid in which the closest double bond to the methyl (CH3) end of the carbon chains is 3 carbons away
omega-6 fatty acid: a polyunsaturated fatty acid in which the closest double bond to the methyl (CH3) end of the carbon chain is 6 carbons away
Omega-3 and Omega-6 Fatty Acids Similarities and Differences
similarities
location of the double bond closest to the methyl end
differences
lengths and numbers of double bonds
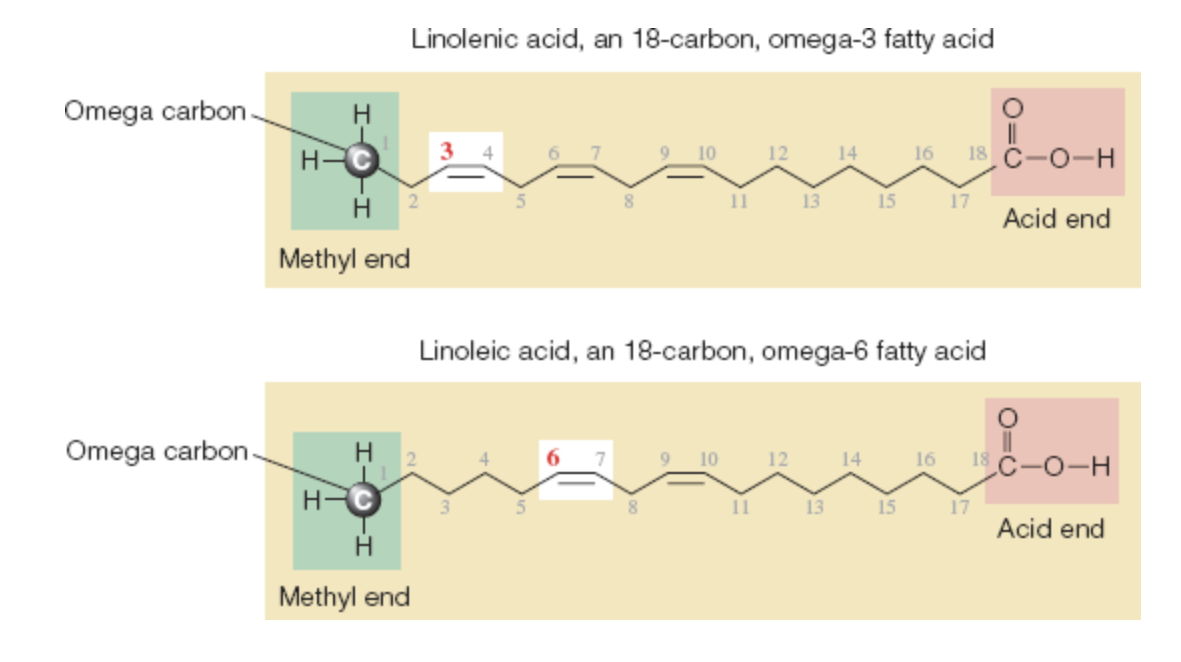
monounsaturated fatty acids
belong to omega-9 group
closest and only double bond 9 carbons away from the methyl end
oleic acid
most predominant monounsaturated fatty acid in the diet
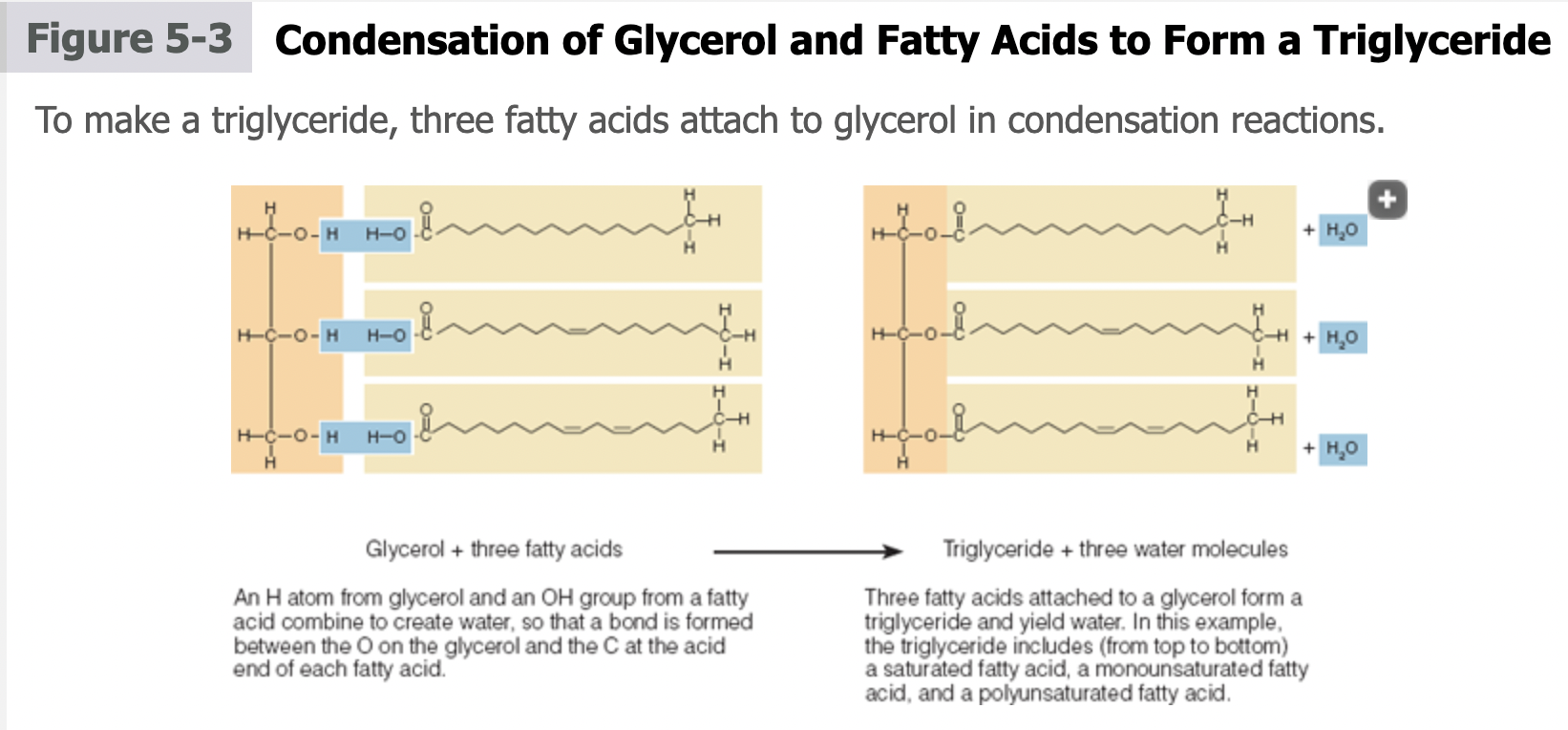
Triglycerides
triglycerides
lipids composed of 3 fatty acids attached to a glycerol molecule
made in a series of condensation reactions
combine hydrogen (H) from glycerol & hydroxyl group (OH) from a fatty acid
contain a mixture of more than one type of fatty acid
Characteristics of Solid Fats and Oils
3 chemical features of a fatty acid affect the characteristics of foods and the health of the body-
short or long
saturated or unsaturated
closet double bond is at carbon 3 or carbon 6
Firmness
degree of unsaturation influences the firmness of fats at room temperature
vegetable fats
polyunsaturated liquids (oils)
cocoa butter, palm oil, palm kernel oil, and coconut oil are saturated
firmer than most vegetable oils because of their saturation
softer than animal fats because of their shorter carbon chains (8-14 carbons long)
saturated animal fats
solid
the shorter the carbon chain → the softer a fat is at room temperature
at room temperature-
saturated fats → solid
fats found in butter and other animal fats
unsaturated fats → liquid
fats found in vegetable oils
Stability
the degree of unsaturation influences stability
all fats become spoiled when exposed to oxygen
bad smell and taste of fat-
oxidation
exposure to heat and light
microbial growth
polyunsaturated fats-
spoil easily because their double bonds are unstable
monounsaturated fats-
less susceptible to spoil
saturated fats-
most resistant to oxidation and least likely to spoil
manufacturers can protect fat-containing products against rancidity-
products are sealed in airtight, nonmetallic containers, protected from light, and refrigerated
add antioxidants (additives BHA, BHT, vitamin E) to compete for the oxygen and protect the oil
products undergo hydrogenation
Hydrogenation
hydrogenation: some or all points of unsaturation and saturated by adding hydrogen molecules and creates trans-fatty acids
2 food-processing advantages-
protects against oxidation, prolonging shelf-life, by making polyunsaturated fats more saturated
alters the texture of foods by making vegetable oils more solid (margarine and shortening)
makes margarine spreadable, pie crusts flaky, and puddings creamy
most often, fat is partially hydrogenated (creating trans-fatty acid)
remaining double bonds after processing change their configuration from cis to trans
Trans-Fatty Acids
naturally, most double bonds are cis (hydrogens next to the double bonds are on the same side of the carbon chain)
trans-fatty acids-
only a few fatty acids (milk and meat products) naturally occur as this
hydrogens next to the double bonds are on opposite sides of the carbon chain
behave like saturated fats
raises blood cholesterol and the risk of heart disease
a relatively small amount of trans fat in the diet comes from natural sources
natural trans fat exert little effect on blood lipids
conjugated linoleic acids-
naturally occurring trans-fatty acids
offer health benefits
not counted as trans fat on food labels