Tips for Drawing Lewis Dot Structures
Decide on the sequence/organization of atoms
The element listed first is typically the central atom
For ABn formulas, the single atom usually is the central atom in the molecule
* Exceptions: H and F are never central and other halogens seldom are
Compounds with symmetrical formulas often adopt corresponding structures
Determine the number of valence electrons to place around each atom:
Recall the trend to quickly predict the number of valence electrons
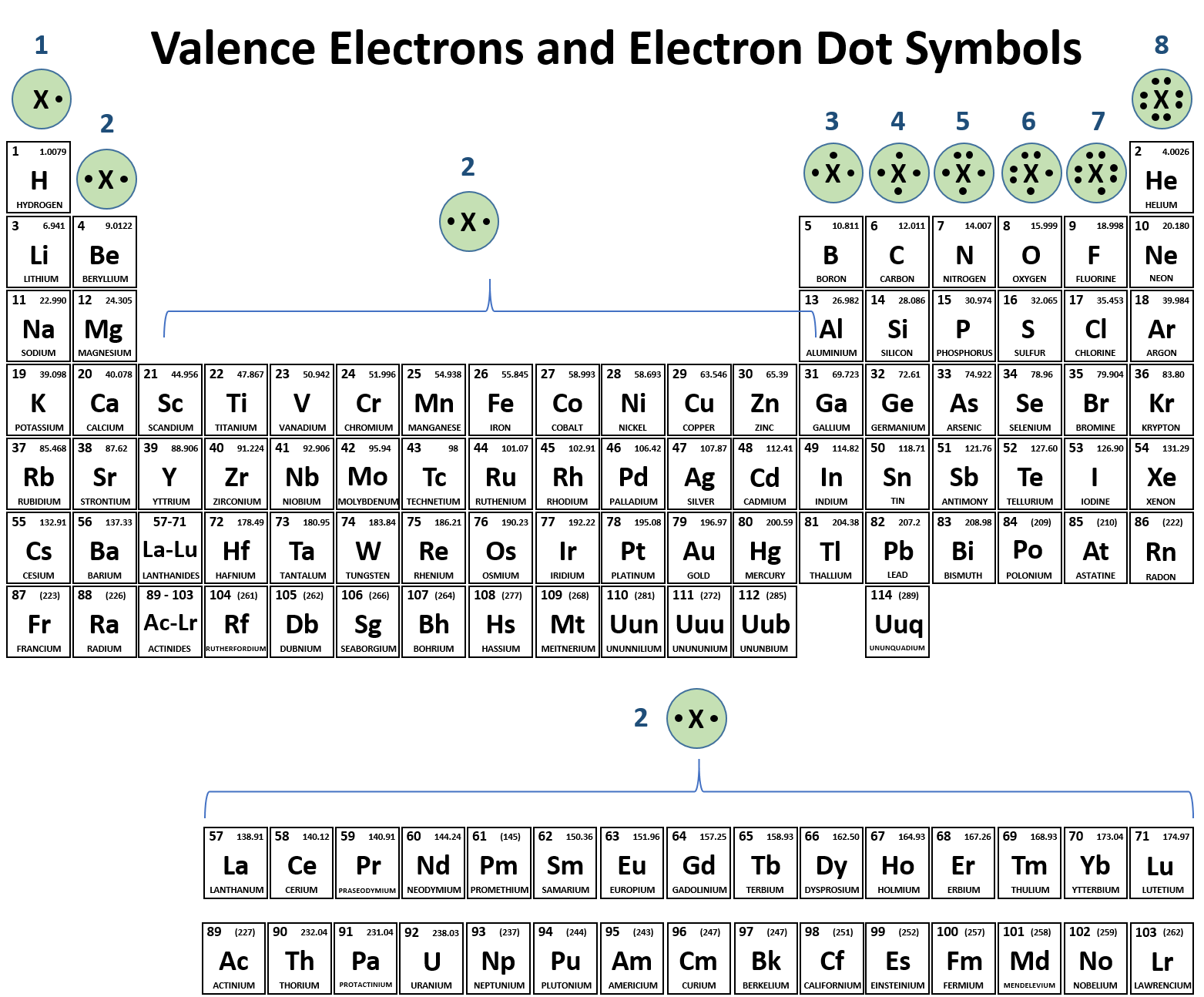
Predict how many bonds the atom is likely to make to achieve a stable octet (noble gas configuration)
Bond all perimeter atoms to the central atom once by circling the pair of electrons to be shared. See who has achieved an octet and who is likely to bond again
Some molecules require more than single bonds to provide each atom with the required octet.
Multiple bonds are formed primarily by the carbon, nitrogen, oxygen, and sulfur atoms.
Know how to alter your structure if an ionic charge is present:
Adding an extra electron for every -1 charge. For example, the NO3- ion has a total of 24 valence electrons: five from the N and 3 × 6 for the three O’s, plus one for the negative charge
Subtracting an electron for every +1 charge. For example, the NF3+ ion has a total of 32 electrons: five from the N, 4 × 7 for the four Fs, minus one for the positive charge.
Limit the number of electrons to eight around any second-period element (Li - F). These elements only take on an octet of valence electrons and their Lewis structure must reflect this.
If appropriate, more than eight electrons can be put around elements from the third or higher period. For example, Phosphorous can use it’ three unpaired electrons to form species such has PCl3, in which there are 10 electrons around the P.
Other elements have a d-sublevel available to bond with and can therefore have an “expanded octet”
EX: P can form 5 bonds to have 10 valence e-
S can form 6 bonds to have 12 valence e-
Common expanded octetes: phosphorous, sulfur, selenium, tellurium, the halogens (except for fluorine) and select noble gases; krypton, xenon, and radon