Topic 5: Formulae,
Equations and Amounts of
Substance
Reacting Masses:
1.
Equation
2. Moles of known
3. Ratio of known: unknown (moles)
4. moles of unknown (from ratio)
5. mass of unknown
Topic 5: Formulae, Equations and Amounts of Substance
Reacting Solutions:

Topic 5: Formulae, Equations and Amounts of Substance
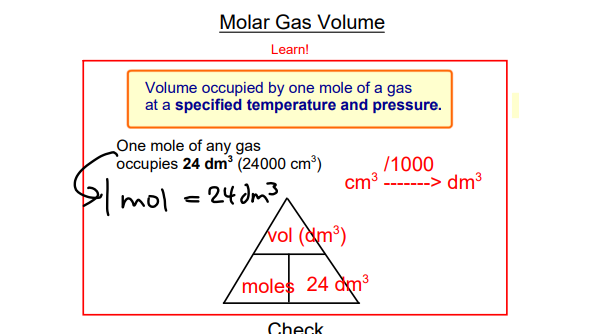
Gas Volumes
V1/n1 = V2/n2

Topic 5: Formulae, Equations and Amounts of Substance b. Determining Molar Volume of a Gas
Method 1: Measuring Cylinder
1. Pour 30 cm3 of 1 mol dm3 hydrochloric acid into the boiling tube. 2. Weigh out 0.05 g of calcium carbonate using a balance. 3. Remove the bung from the boiling tube and tip the calcium carbonate in. Quickly replace bung. 4. Once reaction is over, measure the volume of gas collected in the measuring cylinder. 5. Repeat using increasing masses of calcium carbonate (0.10 g, 0.15 g, 0.20 g etc up to 0.40 g)
Method 2: Gas Syringe:
2. Pour 30 cm3 of 1 mol dm3 hydrochloric acid into the conical flask. 3. Weigh out 0.05 g of calcium carbonate using a balance. Place in a small vial.
4. Remove the bung from the conical flask and place the vial in without it tipping over. Replace bung. 5. Shake the conical flask to tip the vial over. 6. Once the reaction is complete, measure the volume of gas collected in the gas syringe. 6. Repeat using increasing masses of calcium carbonate (0.10 g, 0.15 g, 0.20 g etc up to 0.40 g)


