20.1 : Bronsted-Lowry acids and bases
key terms:
Bronsted-lowry acid is a proton donor
Bronsted-lowry base is a proton acceptor
Conjugate acid-base pair: contains 2 species that can be interconverted by transfer of a proton (related to eachother by the gain or loss of a single proton)
Conjugate acid-base pairs
definition- contains 2 species that can be interconverted by transfer of a proton (related to each other by the gain or loss of a single proton)
example:
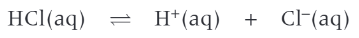
even through the equilibrium sign is shown HCl is still a strong acid
example: HCl + H₂O ⇌ H₃O⁺ + Cl⁻
HCl and Cl- act as an acid and base pair as they differ by H+ (called acid 1 and base 1)
H2O and H3O+ act as the 2nd acid and base pair (called acid 2 and base 2)
see pink book for more