ISOTOPE GEOLOGY
ABUNDANCE OF ELEMENTS
H,He are most abundant in solar system and H/He=12.5
First 50 elements: Exponentially decreases with increasing Z
(We can see progressive decrease in abundance with increase in atomic number but there is not uniformity in that, generally Zig-Zag pattern is observed)
After 50 elements: Do not vary appreciably with increasing Z
Even atomic number are more abundant than neighbour odd ones
Abundance Li,Be and B is anomalously low
Abundance of Fe is anomalously higher
Tc and Pm do not occur naturally in our solar system (because all
of their isotopes are unstable and decay rapidly)
element with atomic number > 83 (Bi) have no stable isotopes but
occur naturally at low abundances
CHONDRITES AND METEORITES
Chondrite corresponds to bulk composition of silicate earth
Fe-meteorite corresponds to core of earth
Chondrites are undifferentiated, unmelted having smaller masses
NUCLEAR BINDING ENERGY CURVE
Fe is the most abundant element of earth
Fe is the highest atomic number that can be synthesized
For synthesizing element having higher atomic number greater than Fe, we need greater energy like Supernova explosion
HERTZSPRUNG-RUSSEL DIAGRAM
Sun is a star which is in main sequence stage right now, but it will not remain there forever because it has an evolutionary path, it will end up as white dwarf
Sun - Red giant - White dwarf
Chandrasekhar limit : limits the mass of star which can undergo explosive events like supernova
Sun has a mass which is below chandrasekhar limit
NUCLEOSYNTHESIS
Process of creation of different chemical Elements
Process of creating new atomic nuclei from pre-existing nucleons (protons + neutrons)
H-burning and He-burning are very long duration processes
S-process : slow process but rapid than H and He-burning
r-process: Extremely rapid process
Other processes
e-process : silicon burning
p-process : proton capture
r and s-processes : neutron capture
x-process : light element synthesis
NUCLEAR STABILITY AND DECAY
1.Neutron - Have mass but no charge
2.Proton - Posses mass and positively charged
Mass of both proton and neutron = 1.67*10 -23
Mass of Electron = 1/1836.1 of mass of Proton
Isotopes: Nuclides with Same Z, different N
Isotones : Nuclides with Same N, different Z.
Isobars: Nuclides with Same A, different Z and N
Out of 2500 known nuclides only 270 are stable
Nuclides having magic proton number or magic neutron numbers are unsually stable
FUNDAMENTAL PARTICLES
Proton and Neutrons are composite particles i.e they can be broken down further
Proton : Consist of two up quarks and one down quark
Neutron : Consist of two down quark and one up quark
W-boson is extremely unstable and immediately decays to electron as beta-particles
RADIOACTIVE DECAY
Process by which unstable nuclides are converted into stable nuclides
Nuclear transmutation process
TYPES OF DECAY MECHANISM
Beta Negatron : neutron rich
N —> P + e (Transformation of neutron to a proton and electron)
Z+1 and N-1
Atomic no. increased by 1 and Neutron no. reduced by 1
eg : 37 Rb ——> 38 Sr
Beta Positron : proton rich
P ——> N + Positron + Neutrino (Transformation of a proton to neutron, positron and neutrino)
Z-1 and N+1
eg: 9 F ——→ 8 O
Mass is conserved in case of beta decay
Electron-capture decay
Reaction between extranuclear electron and proton gives rise to neutron and neutrino
e + P ———→ N + neutrino + X-ray
X-ray is the product
Alpha decay : proton rich
Alpha particle is the He-nuclei ( 24He)
Z-2 and N-2
Overall atomic mass reduced by 4
eg: 92 U ——→ 82 Th + 24He + Q
Nuclear Fission
Heaviest isotopes are decay by this Nuclear fission
Elements with greater atomic number than uranium and transuranic elements
the fission products have excess neutrons and therefore
undergo further decay by β- and gamma-ray emission until a
stable nuclide is formed
FORMULAE
1) D= D0+ N (e λt-1)
2) M= eλt-1 , (M=Slope)
3) N/No = e λt-1
4) N/No = 1/2 n , (n= No. of half lives) , n=t/t1/2
5) t1/2= 0.693/λ, (t1/2= Half life, λ= Decay constant)
6) t1/2 = 0.693* Mean life
7) N1/t1/2(P) = N2/t1/2 (D), N1λ1=N2λ2 (Secular Equilibrium)
8) D= No-N
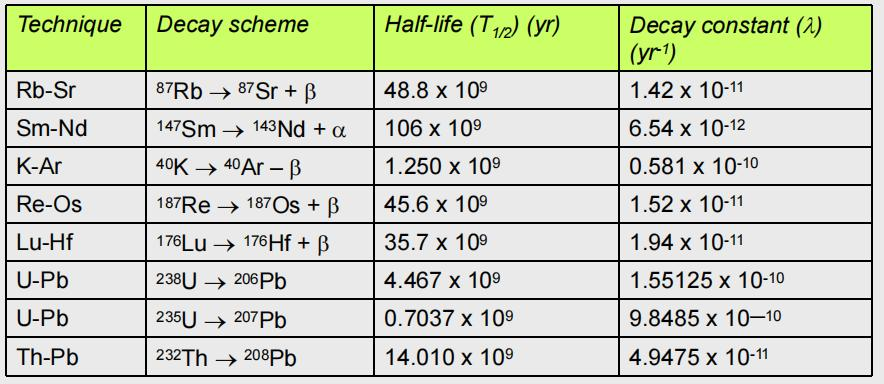
DECAY SCHEME AND THEIR HALF LIVES
K-Ar method:
used for potteris
date cenozoic/mesozoic volcanic rocks (younger volcanic rocks)
used to date metamorphic events
Rb-Sr method:
dating acidic and intermediate igneous rocks such as rhyolite, dacite, granite etc.
U-Th-Pb method:
Zircon dating
C-N method
date recent events, Holocene and Pleistocene rocks
MASS SPECTROMETRY
Instrument that we used to measure age of earth
Mass spectrometer separate ions of different mass
All these parts are evacuated to the pressure order of 10-6 to 10-9
SOURCES OF MASS SPECTROMETER
TIMS (Thermal Inonization MS)
Pure form of the element is deposited in metal filament made up of Re,Ta, W
Which is electrically heated to volatilize and ionize the element into positively charged ion by stripping outer most electron
PLASMA SOURCE ( ICP-MS)
also a form of thermal ionisation
restricted to filament under vaccum - technique
A plasma is generated by an electrically excited charge
SECONDARY ION (SIMS)
a secondary target mineral surface is bombarded insitu
with a high energy beams of ion to split or remove secondary ions
extracted positive ions are accelerated and analyse in a double focusing mass spectrometer
Here we don’t need to digest the sample
GAS SOURCE ( NOBLE GAS MS)
Extracted under ultra high vaccum
gas extraction is done using a furnace or infra-red laser
ACCELERATED MS (AMS)
Particularly used for samples which are very low in abundances
SAMPLE PROCESSING
Non-invasive technique : (SIMS, LA-ICP-MS)
Suitable polished section should be prepared
Detailed petrographic study, Backscattered electron image (BSE) and Cathodoluminiscene image (CL) are needed
Minerals such as Zircon, Titanite, etc can be separated from > 5 kg rock sample crushed to 180 micro m
Mozley superpanner or Wilfley table may be used
Invasive technique (TIMS, ICP-MS)
Rock needs to be digested by attacking HF+HNO3+Hcl acids
obtaining 70 micro m powder for digestion
Most rock forming mineral dissolve above 100 C
Resistant mineral like Zircon needs temp upto 220 C
Low procedural blank
ISOTOPE DILUTION
Digested sample split into two weighted aliquots
One of which is spiked with an enriched isotope and homogenized for isotope dilution analyis
Other unspiked aliquot is used for determination of isotropic compostion or isotropic ratio
K-Ar Method
daughter element Ar is an inert gas
parent element potassium is an alakali metal
K had been the dominant contributor to the heat budget
DISTRUBUTION OF K
Highest conc in crust being an alkali metal
It is incmpatible
In bulk mantle, K is an accessory or trace element
K has 3 isotope among which 40 K is radiogenic
It has a brached decay
40 K decays to 40 Ar (EC)and 40 Ca (beta)
Braching ratio i.e EC/Beta = 0.117
APPLICATION OF K-Ar METHOD
Contact metamorphic zones
Retention sequence :
K-Feldspar < Biotite (350) < Muscovite (450) < Hornblene (550)
Hornblende has better retention of Ar
Biotite has lower retention of Ar
Date cenozoic/mesozoic volcanic rocks
Date ocean mounds and ocean islands