Radioactive Decay, Half-Life Calculations, and Balancing the Equations
Radioactive decay - process in which unstable nuclei of radioactive isotopes become stable by emitting charge particles and energy and becoming other elements.
- The nucleus of a radioactive atom gives off radiation in the form of energy and a particle of matter.
- Alpha decay is the least damaging, and gamma decay is the most damaging.
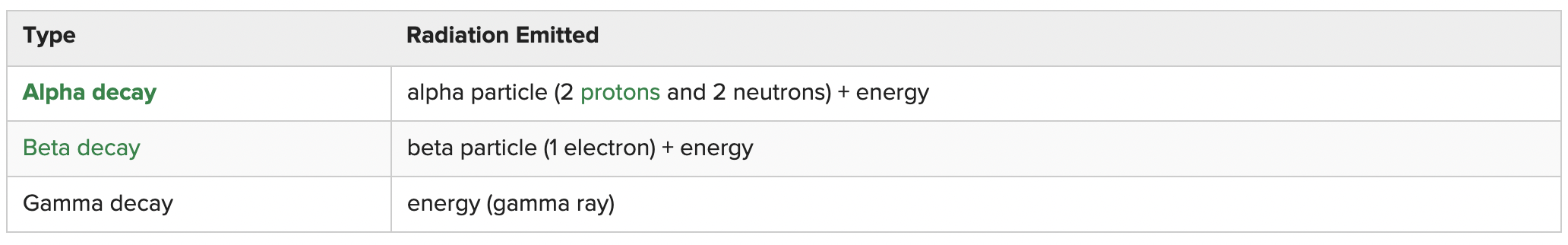
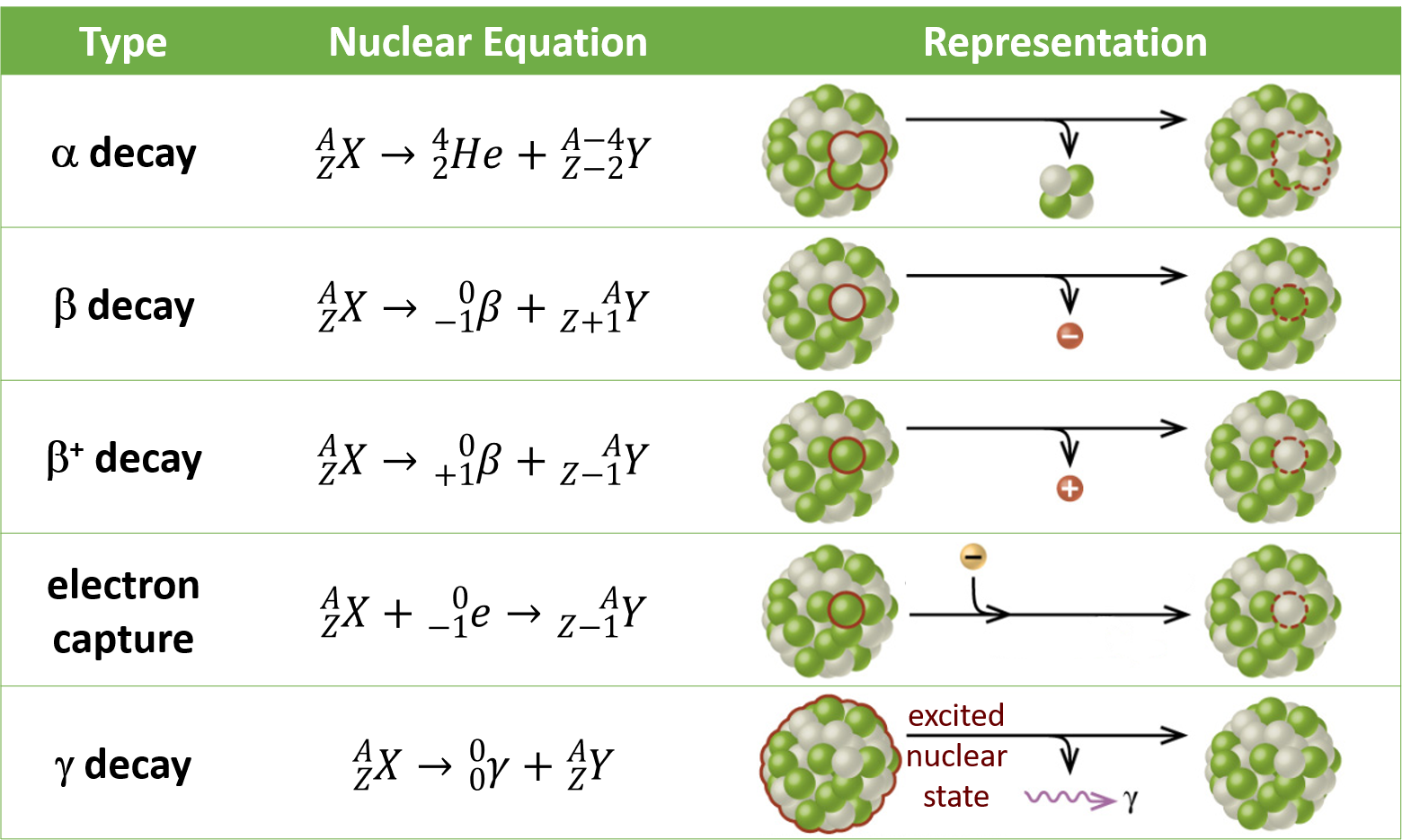
- Alpha decay occurs in atoms with too many protons.
* an alpha particle is a helium nucleus
*

- Beta decay occurs when an unstable nucleus emits a beta particle and energy
* beta particle is either an electron or a positron
* electron is negatively charged, positron is positively charged
* when the beta particle is an electron, the decay is called beta-minus decay; when the beta particle is a positron, the decay is called beta-plus decay
* beta-minus decay occurs when a nucleus has too many neutrons relative to protons, and beta-plus decay occurs when a nucleus has too few neutrons relative to protons.
*
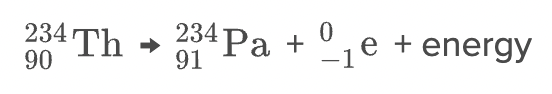
- Gamma rays are electromagnetic waves, and have the greatest amount of energy out of all the types of EM waves. They travel at the speed of light. * Gamma particles are photons of energy. * In alpha + beta decay, both particles and energy are emitted. In gamma decay, only energy, in the form of gamma rays, is emitted. * Gamma decay, in contrast, occurs when a nucleus is in an excited state and has too much energy to be stable. This often happens after alpha or beta decay has occurred.
\ Half-Life
- A radioactive isotope atom has as unstable nuclei, which decays by losing energy and particles of matter to become more stable. If they gain or lose protons as they decay, they become different elements. Over time, as the nuclei continue to decay, less and less of the original radioisotope remains.
- Half-Life - the amount of time it takes for half of a given amount of a radioisotope to decay, which is always the same for a given radioisotope regardless of other factors

\