materials
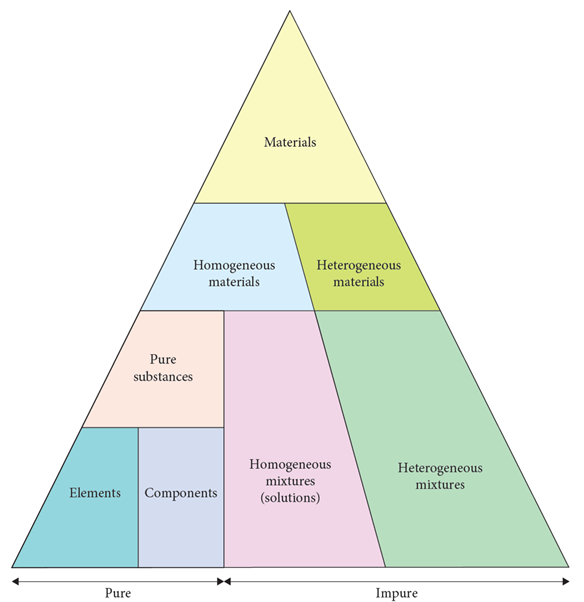
TWO TYPES OF CLASSIFICATION FOR MATERIALS:
PURE SUBSTANCES:
Materials with distinct measurable properties ie melting and boiling points, reactivity, strength, density, etc.
They contain only ONE type of molecule/particle e.g., Argon, O2, and water. Therefore every single molecule/particle is the SAME.
MIXTURES:
Materials with properties that are dependent on the identity and relative amounts of the substances that make up the mixture.
They contain MORE than one type of molecule/particle e.g., saltwater, air and ink. Therefore every molecule/particle AREN’T the same.
HETEROGENEOUS VS HOMOGENEOUS:
HETEROGENEOUS MATERIALS:
2 or more DIFFERENT substances, proportions can vary and mixtures are NON UNIFORM
HOMOGENEOUS MATERIALS:
UNIFORM composition, pure substances like elements/compounds and a solution (the only mixture that can be homogeneous)
PHYSICAL PROPERTIES VS CHEMICAL PROPERTIES:
physical properties
can be determined WITHOUT changing the chemical compositions. These differences then allow for the identification of different materials. The properties of the components in a mixture then determine the technique used to separate them.
physical change ——> changes in the physical properties (colour, density, and state) WITHOUT a change in chemical composition e.g. tearing paper
chemical properties
relate to the reactions that form new substances such as decomposition by heat, the effect of light, and reactions with water, oxygen, bases, acids, and other substances)
chemical change ——> at least one new substance being formed with DIFFERENT chemical and physical properties e.g., digestion and burning. These changes are recognised by a solid (precipitate) forming, a gas being produced, colour change, a significant change in temp, and/or an insoluble solid disappears.
SEPARATION TECHNIQUES:
Particle size:
1 = SIEVING ————>
used to separate a DRY mixture that contains substances of different particle sizes by passing it through a sieve.
the mixture of solids with different sized particles passes through a mesh which allows the smaller sized particles to pass through whilst the larger ones will remain behind
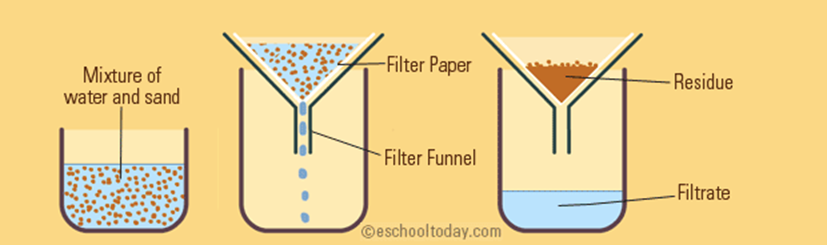
2 = FILTRATION ————>
the method used to separate on the basis of a difference in particle sizes in wet suspension IE it’s used to separate solid particles from a liquid or gas.
2 TYPES, gravitational filtration and vacuum filtration
Boiling points:
1 = VAPOURISATION/EVAPORATION ——>
solids that dissolve (solute) in liquids (solvent) to form a solution CAN’T be separated using filtration, meaning that the solvent MUST be evaporated or boiled off in order to recover the solid solute
used when ONLY the SOLUTE is valuable
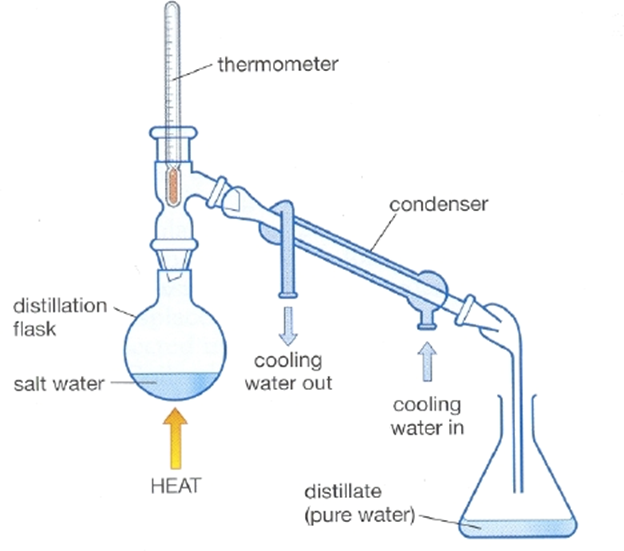
2 = DISTILLATION ———————>
an important technique when the SOLVENT is valuable, meaning you use a condenser IE the recovery of water from saline solutions.
it uses the same principle as evaporation BUT is performed in an apparatus in such a way that the evaporated liquid CAN be recovered.
STEPS:
1) the solution is heated in a distillation flask to then vaporise the liquid.
2) The vapor passes through a condenser = a tube cooled with running water and cools the vapor, making it condense back into a liquid forcing droplets to form along the inside of the condenser.
3) The condensed liquid drips out of the condenser and into a second flask = the receiving flask.
This received liquid = the distillate
It’s usually used with large differences in boiling points BUT can be used with substances that have a closer range but the SOLVENT will be very contaminated
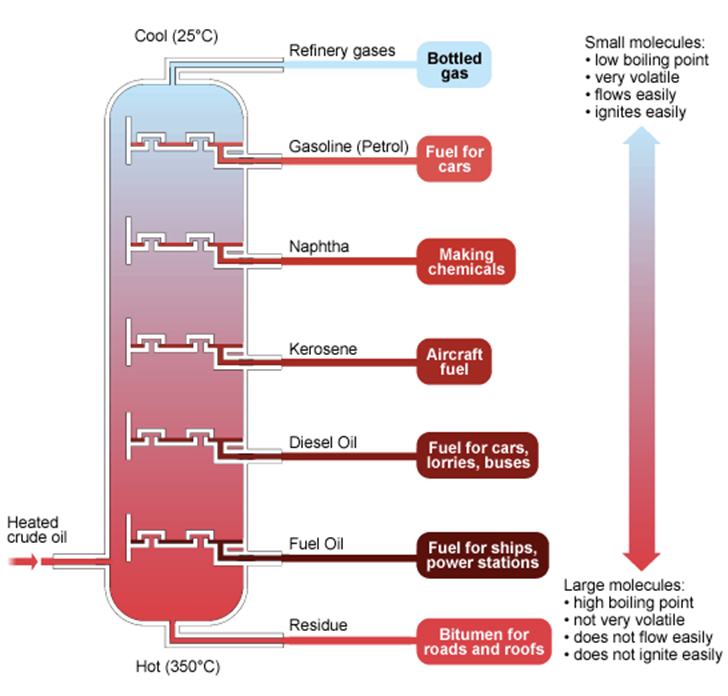
3 = FRACTIONAL DISTILLATION —————>
taking a very complex mixture and separating it into ALL the different fractions available for it. Meaning that it’s used to separate miscible (can mix) liquids with similar boiling points
It involves the cycling of condensation and evaporation that eventually leads to the final solutions being very pure (because it exists and its boiling point and ISN’T extracted straight away.)
Density and solubility:
the separation is formed when the immiscible (can’t mix) liquids separate in layers due to differences in density e.g., oil and water.
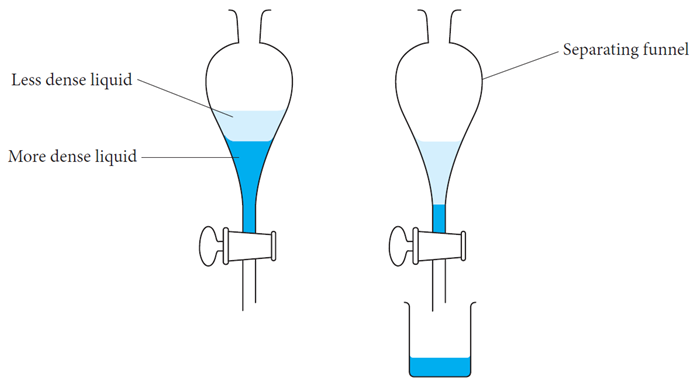
1 = SEPARATION FUNNELS ——————>
if two liquids have different densities and are immiscible then a separation funnel can be used.
when placed into a flask, 2 layers will form - the less dense liquid floats to the top whilst the denser liquid will sink to the bottom and will then flow out first.
Making the process itself a liquid-liquid extraction.
Magnetism and electrostatic attraction:
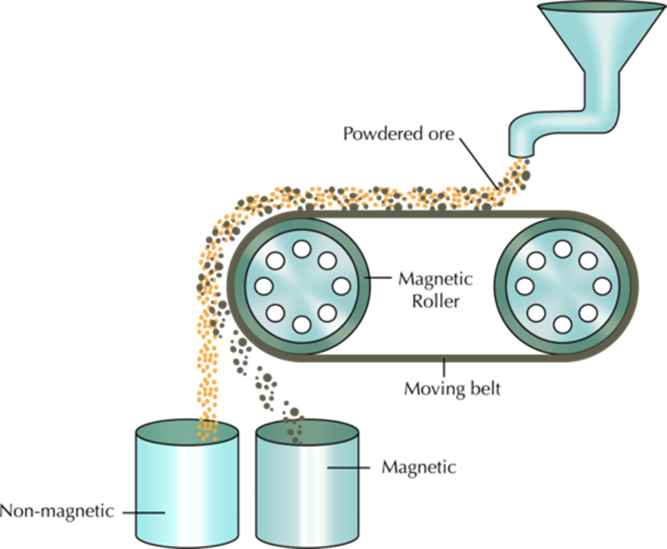
1 = MAGNETIC SEPARATION ———>
uses the degree of attraction to a magnetic field to separate the substance e.g., iron, cobalt and nickel.
2 = ELECTROSTATIC SEPARATION ————>
relies upon the differences in electrical charge e.g., mineral sands containing zircon, rutile and monazite.
NANOTECHNOLOGY:
involves the study of nanoparticles and structures as well as their manufacture and potential use.
A nanometer = 1/1000 mm
nanotechnology size = 1-100nm
These are important for 3 reasons.
Their extremely small size - even in comparison to biological cells (meaning they are useful in science)
Their large surface area due to their small size, meaning they are helpful in engineering
The quantum effect (they don’t behave as expected due to their extremely small size e.g., light) that occurs due to their extremely small size meaning they can be used in labs.