PHYSIO MIDTERM (I WANT TO KMS💚💚)
2.2 Somatic Senses
Definition
Somatic senses are the mechanisms by which the nervous system collects sensory information from different parts of the body.
They are distinct from special senses (vision, hearing, scent, taste, balance).
Types of Somatic Senses
Classifications Based on Physiological Cell Types:
Mechanoreceptive Somatic Senses:
Tactile Senses: Touch, pressure, vibration, tickle.
Position Senses: Static position and rate of movement.
Thermoreceptive Senses: Detect temperature changes.
Pain Sense: Detect noxious stimuli.
Sensory Receptors of the Skin
Skin receptors encode tactile information such as fine touch, pressure, vibrations, temperature, and pain.
Approximately 1 million sensory nerve fibers innervate the skin. Key receptor types include:
Fine-tactile:
Type Aα (Group I)
Type Aβ (Group II)
Pain:
Type Aδ (Group III)
Type C (Group IV)
Temperature:
Type Aδ (Group III)
Type C (Group IV)
Large myelinated fibers (Type A) encode touch, vibration, and pressure.
Type C fibers are associated with crude touch, slow pain, and thermal sensations.
Dorsal Root Ganglion
Sensory fibers for touch, pressure, vibration, proprioception, pain, and temperature are peripheral projections of pseudo unipolar neurons. Their central axons enter the spinal cord or brain stem.
These sensory ganglion neurons are the primary efferent or first order neurons of the somatosensory system.
The cell bodies of these pseudo unipolar neurons are clustered in the dorsal roots of the spinal cord, forming the dorsal root ganglion.
The cell bodies of these pseudo unipolar neurons are clustered in the dorsal roots of the spinal cord, forming the dorsal root ganglion.
Each first-order neuron in the dorsal root ganglion is part of a three-neuron chain that reaches the cerebral cortex via the thalamus.
The dorsal root ganglion or the trigeminal ganglion neurons are the primary afferent neurons in the somatosensory system
Two classes of dorsal root ganglion neurons:
Large dorsal root ganglion neurons:
Receive input from group I and II fibers.
Have large diameter myelinated fibers (Type Aα and beta).
Mediate fine touch and proprioception.
Have large cell bodies.
Small dorsal root ganglion neurons:
Receive input from group III and group IV fibers.
Include Aδ and C fibers (1 myelinated, 1 unmyelinated).
Mediate pain, itch, temperature, and crude touch.
Ascending Pathways in the Spinal Cord
Two Major Pathways:
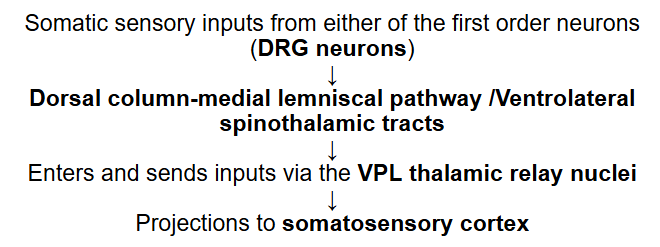
Dorsal Column – Medial Lemniscal Pathway:
Carries sensory input from the large dorsal root ganglion neurons.
Carries fine touch, pressure, and proprioceptive sensations.
Structures involved include:
Nucleus Gracilis: Receives from lower body.
Nucleus Cuneatus: Receives from upper body.
Fibers ascend ipsilaterally and decussate at the medulla.
PATHWAY:
First-Order Neuron:
Sensory neurons in the dorsal root ganglia (pseudounipolar neurons) transmit signals from the periphery to the spinal cord.
Dorsal Columns: In the spinal cord, the axons of these neurons travel in the dorsal columns (fasciculus cuneatus and fasciculus gracilis).
Medulla Oblongata: The axons synapse in the dorsal column nuclei in the medulla oblongata.
Medial Lemniscus: Second-order neurons then cross the midline and ascend through the brainstem in the medial lemniscus.
Thalamus: The medial lemniscus terminates in the thalamus, specifically the ventral posterolateral nucleus (VPL).
Third-Order Neuron: Third-order neurons in the thalamus project to the primary somatosensory cortex in the brain.
Ventrolateral Spinothalamic Tract:
Carries sensory input from the smaller dorsal root ganglion neurons.
Carries pain and temperature sensations.
Fibers decussate within the spinal cord at the level of entry.
Functions for sharp pain, temperature, and crude touch. Converges spinal responses to higher brain centers.
PATHWAY:
First-order neurons: Sensory neurons in the dorsal root ganglia.
Second-order neurons: Synapse in the dorsal horn of the spinal cord, where the axons cross the midline and ascend in the anterolateral spinal cord as the spinothalamic tract.
Third-order neurons: Synapse in the thalamus and project to the primary somatosensory cortex.
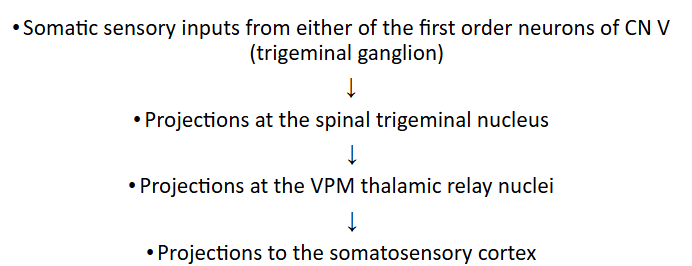
PATHWAY: (face region)
first order neurons: reside in the trigeminal ganglion.
The axonal terminals of the trigeminal ganglion neurons synapse with neurons of the trigeminal sensory nuclei
Then projects to the ventral posteromedial (VPM) thalamic nuclei
The 3rd order neurons from the ventrobasal complex (VPL + VPM), finally, project to the somatosensory cortex.
Thalamus
A paired gray matter structure in the dorsal part of the diencephalon located near the center of the brain
Allows nerve fiber connections to the cerebral cortex in all directions
Serves as a relay for sensory information to the cortex. receiving separate ascending pathways from the sense organs
For sight, hearing, equilibrium, taste, and somatic senses
These signals would then project to, separately, the various primary sensory cortices
Visual, auditory, and somatosensory cortices
Other thalamic nuclei also have synaptic connections with the cerebellum and the basal ganglia for the control of movement, and cognitive and emotional circuit loops
Involved in the regulation of consciousness, sleep, and alertness
Trunk/Limbs:
Face/head Region:
Sensory Homunculus
Topographic representation of different areas of the body in somatosensory area I (somatosensory cortex)
Fibers carrying inputs from the lower part of the body are located medially
Fibers carrying inputs from the upper part of the body are located laterally
This kind of somatotopic organization of nerve projections at the spinal cord is continued through the thalamus and the somatosensory cortex.
Because of decussations of ascending pathways, each lateral side of the cortex receives sensory inputs almost exclusively from the opposite side of the body.
Some areas in the body are represented by large areas in the somatosensory cortex.
Trunk and lower part of the body are represented by smaller areas
Other Ascending Pathways of the Somatosensory System
The collateral pathways arising from the main spinothalamic tract:
Spinoreticular Pathway
“Spinoreticulothalamic Pathway”
Sends collateral signals from ascending pain pathways to the brainstem reticular formation, and from the reticular formation to the limbic system and the rest of the cerebral cortex via the thalamus as part of the Reticular Activating System (ascending arousal system).
Function:
Concerned with the affective component of pain
Arouses the organism/human to deal with painful condition
Spinomesencephalic Pathway
Ascending pathway for the activation of the brainstem descending pathway inhibition system from the periaqueductal gray.
Function:
Serves as afferent limb for descending pain control activation
Spinocerebellar Pathway
For unconscious proprioception
Carry unconscious proprioceptive signals from muscle spindles and golgi tendon organs and other joint receptors to the cerebellum.
Function:
For motor control
Major Cutaneous Mechanoreceptors
What are Mechanoreceptors?
Mechanoreceptors are sensory receptors that react to mechanical stimulation. This means they're activated by things like:
Touch: A gentle brush or a firm press.
Pressure: A sustained force on the skin.
Vibration: Rapidly repeated pressure.
→ These receptors are crucial for everything from feeling a light breeze to holding a heavy object.
Key Cutaneous Mechanoreceptors:
Pacinian Corpuscles:
These are found deep in the dermis and hypodermis.
They are very sensitive to deep pressure and high-frequency vibration.
Pacinian corpuscles are rapidly adapting, which makes them ideal for detecting vibrations, like the buzz of a phone.
Meissner's Corpuscles:
These are also found in the upper layers of the skin, especially in areas sensitive to touch like the fingertips and lips.
They are specialized for detecting light touch and low-frequency vibration.
Meissner's corpuscles are rapidly adapting, meaning they respond strongly to the initial stimulus but quickly stop firing if the stimulus is maintained. This makes them good at detecting changes in touch.
Ruffini Endings:
These are located deeper in the skin.
They are sensitive to skin stretch and sustained pressure.
Ruffini endings are slow-adapting and contribute to our sense of proprioception (body position).
Merkel's Disks:
These are located in the upper layers of the skin.
They are sensitive to light touch and sustained pressure.
Merkel's disks are particularly important for fine tactile discrimination, like reading Braille or feeling the texture of fabric.
They are slow-adapting, meaning they continue to fire signals as long as the stimulus is present.
Hair Follicle Endings:
These are nerve endings that wrap around hair follicles.
They are sensitive to touch and movement of the hair.
Hair follicle endings are rapidly adapting, allowing us to detect things like a gentle breeze or an insect crawling on our skin.
Cutaneous Thermoreceptors
Temperature sensation is encoded by:
Cold receptors (cold fibers)
Free nerve endings of Group III (A𝛿) and Group IV (C)fibers.
Warm receptors (warm fibers)
Free nerve endings of Group IV (C) fibers
There are 4-10 times as many cold receptors as warm receptors.
Receptive fields of thermoreceptors are very small spots.
1mm in diameter in glabrous skin 3-5 mm in hairy skin,
3-4 of which are innervated by a single primary axon
Both cold and warm thermoreceptors give information about the body temperature by changing its firing rate.
Skin temperature: decreases
Cold fiber: transiently increases its firing rate
Warm fiber: transiently decrease its firing rate
Skin temperature: increases
Cold fiber: transiently decreases its firing rate
Warm fiber: transiently increases its firing rate
Nociceptors, Fast and Slow Pain Pathways
Two types of pain sensation result from the application of a strong, noxious stimulus to the skin:
First (fast) pain vs. Second (slow) pain
Nociceptors according to type of pain and fiber types
Dual Pain Pathways in the Cord and Brainstem
First Pain: Fast (initial) pain
Also known as: “Epicritic Pain”
Discrete, well-localized pinprick sensation that results from activating the nociceptors on the A𝛿 fibers
A𝛿 nociceptors
(A𝛿) 2-5 microns
12-35 m/s C.V. or A𝛿 nociceptors
Second Pain: Slow (delayed) pain
Also known as: “Protopathic pain”
A poorly localized, dull, burning sensation that results from activating the nociceptors on the C fibers.
C-nociceptors (C fibers)
0.4-1.2 microns
0.5-2 m/s C.V.
Dual Pain Pathways in the Cord and Brainstem
Neospinothalamic tract (fast pain system)
Conveyed by the A𝛿 fibers
Ascend the main ascending pathway of the ventrolateral spinothalamic tract, passing through the specific thalamic relay nuclei to reach the cerebral cortex
Part of the ventrolateral spinothalamic tract
To remind the type of pain which it conveys to the somatosensory cortex via the specific thalamic relay nuclei
Sending fast pain signal to somatosensory cortex is the reason why fast pain is easier to localize than slow pain
Paleospinothalamic tract for (slow pain system)
Many painful conditions mediated by slow pain mechanisms via the slowly conducting C fibers gives poorly localizable slow burning sensation
Aka Spinoreticulothalamic pathway or spinoreticular tract
Majority of pain fibers from the spinothalamic tract project to the brainstem reticular formation
Peripheral Mechanisms of Pain (Receptor to Brain)
Fast pain: Type A-delta δ fibers of dorsal root ganglion neuron via spinothalamic tract (neospinothalamic tract)
Reaches cortex via ventrobasal complex of thalamus (whether ventroposterolateral thalamic nuclei or ventroposterior medial thalamic nuclei
Topographically represented in the cortex, which accounts for ability to encode the more precise location of the stimulus producing fast pain
Aδ fibers synapse with 2nd order neurons in Laminae I of dorsal horn
Slow pain: Type C fibers or dorsal root ganglion (1st order neurons) via spinoreticulothalamic system or (Paleospinothalamic tract)
1st order neurons (Type C fibers) synapse with 2nd order neurons in Laminae II and III; signals reach cortex via intralaminar thalamic nuclei (after passing through brainstem reticular formation)
Slow pain, which has a more diffuse pathway, travels to the brain through the spinoreticulothalamic system; projects to the brain via
Collaterals of this system pass through the reticular formation to activate fiber tracts that connect to the limbic system and produce the emotional perceptions accompanying pain sensations.
These pathways account for the intense unpleasantness associated with slow pain.
Other tracts activated are those:
Connected with the brainstem
Ascending arousal system which project to the intralaminar thalamic nuclei
Later on projects with parts of cerebral cortex to produce behavioral arousal
Cutaneous Sensations and Introductory Pain Physiology
Types of cutaneous nociceptors according to adequate stimuli:
Mechano-nociceptor (Type Aδ, C fibers) – respond to strong pressure from damaging stimulus/sharp object
Thermal nociceptors (Type Aδ, C fibers)- respond to changes in skin temperature > 42 Celsius, or severe cold
Chemo-sensitive nociceptors (Type C) – respond to the release of tissue chemicals from damaged tissues such as Bradykinin, histamine, H+ and other environmental irritants
Polymodal nociceptors (Type C fibers)- respond to mechanical, thermal or chemical noxious stimuli
It has also been found that there are nociceptors which respond to both mechanical and thermal stimuli
This is possibly distinct from the polymodal receptors or variations of polymodal nociceptors which respond to all types of damaging noxious stimuli.
Glutamate as an Excitatory Neurotransmitter for A𝛿 and C- fibers
A major excitatory neurotransmitter released from A-delta or C fibers when stimulated.
Substance P is released from C fibers in response to tissue injury or to intense stimulation of peripheral nerves.
Substance P is released together with neurokinin A and CGRP (calcitonin gene related peptide), and other excitatory amino acids (Glutamate and Aspartate) during prolonged stimulation of C fibers simulating the conditions of chronic pain.
Glutamate and other neuropeptides are released together from primary efferent terminals and have distinct physiologic actions on postsynaptic neurons.
Substance P enhances and prolongs the actions of glutamate.
A𝛿 vs. C- fiber Stimulation
A delta fibers: fast peaking pain sensation
C fibers: slow burning pain sensation
When A delta fibers (first order neurons) are stimulated, action potentials are produced in the postsynaptic second order projection neurons which shows that the excitatory neurotransmitters are released to excite the postsynaptic membrane.
Glutamate is the principal excitatory neurotransmitter released from the central terminals of A delta fibers.
Increasing the number of stimulus applied to the A delta fibers shows the same number of action potentials produced in the postsynaptic neuron
Stimulating C fibers produces an increase in excitability in the postsynaptic neuron. As the number of stimuli applied to the C fibers are increased, so does the number of spikes produced after repeated stimulation.
Augmented response in the postsynaptic neuron
Substance P and other neuropeptides like neurokinin A or neurokinin A augment the fast excitatory effects of glutamate on the postsynaptic neurons
Cutaneous Nociceptor (Pain Receptors)
Cutaneous nociceptors that respond to noxious stimulus:
Free nerve endings of small myelinated A fibers (Group III)
Free nerve endings of small unmyelinated C fibers (Group IV)
Specific to painful stimuli
Responds to damaging or potentially damaging mechanical, chemical and thermal stimuli
What happens when touch receptors (Pacinians, Merkel’s Ruffini’s, Meissner’s) are stimulated intensely?
When one stimulates fine tactile receptors, despite any intensity, it will not produce pain.
One will only experience sensations produced by tactile receptors
Visceral and Deep Tissue Nociceptors
All visceral pain that originates in the thoracic and abdominal
cavities => transmitted through small type C pain fibers
Produces chronic, aching, suffering type of pain
- Manifested as REFERRED PAIN
Many nociceptors in:
Periosteum, joint surfaces;
Blood vessel walls (arteries and veins)
Brain itself has no nociceptors
However, there are plenty of nociceptors in the meninges (dura mater, falx cerebri/falx cerebelli, tentorium of cranial vault) and the periosteum of bone
Periosteum and bone marrow cavity are innervated by small diameter slow conducting units with free nerve endings, typical of nociceptors
There are both type A and type C nerve endings with biochemical markers present here
Visceral Pain Pathways vs. Parietal Pain Pathways
Visceral Pain Pathways
True visceral pain is difficult to localize, usually (expressed as referred pain).
Visceral pain afferents travel along autonomic nerves, joining dorsal root ganglion, and converge on spinal projection neurons receiving inputs from somatic afferents.
Parietal Pain Pathways
Parietal pain sensations arising from body wall
Conducted directly onto spinal nerves
Sensations localized directly from painful area
Usually on the body wall above affected organ
Referred Pain
Visceral pain is poorly localized as it is referred to areas of the skin which are supplied by the same sensory ganglia and spinal cord segments as the nerve fiber innervates the internal organs.
• Visceral Nociceptors Excitation
Referred as sensation coming from an area of the body surface (in same dermatomal segments from which originated in the embryo)
Chronic Pain Mechanisms
Chronic pain:
A sensation of pain that endures long after the stimulus is removed, and the injury is healed
It is an extremely debilitating condition that is difficult to treat
May result from the spontaneous
One kind: Neuropathic pain - caused by injury to pain fibers.
Reverberating Circuit hypothesis:
Chronic pain is supposed to result from spontaneous activity within the CNS
Reverberating circuits that develop because of continuous pain input may fail to stop firing when the input is removed.
As a result of a continuous Cycle of interneurons augmenting the flow of signals from the T3 to CNS
The closest to this rc is the wind-up phenomenon involving glutamate and nitric oxide in ascending pain pathways.
Neuropathic Pain:
Arising from nerve injury affecting pain fibers
Normal state: nociceptive nerve endings are not hyper excitable unless directly stimulated; even if norepinephrine is circulating, there
is still no abnormal; excitation of the uninjured peripheral nerve fibers including pain fibers.
After nerve injury, there is sprouting and overgrowth of sympathetic nerve fibers around the dorsal root ganglia of the sensory nerve including pain fibers from injured area
The adrenoceptors or epinephrine are expressed to the neuronal
membrane of the nociceptive peripheral endings and cell bodies of
injured cell fibers surrounding uninjured cell fibers
Sympathetic discharge around the injured and surrounding nerve
fibers makes them hyperexcitable which brings some pain in the
presence of circulating norepinephrine
3 sites where nociceptors become responsive to sympathetic
stimulation following partial nerve lesion:
Cell on the membrane of the soma of dorsal root ganglion
Axonal stamp of the injured nerve fibers
The peripheral nerve endings of uninjured adjacent nerve fibers
This causes continuous discharge of the nociceptive fibers contributing to hyperexcitability of the postsynaptic neurons in the spinal cord and other supraspinal centers.
Other Conditions of Chronic Pain:
Denervation Supersensitivity Concept
• May develop in pain centers subsequent to the removal of a pain fiber input
• an increased sensitivity to circulating neurotransmitters that occurs when the normal synaptic input is removed from a neuron, due to upregulation of postsynaptic receptors after sensory denervation.
• Example: Phantom pain
present in many amputees who have had their limbs cut off due to disease/trauma
Anterolateral Cordotomy
• Severing the ascending pathway of the ventral column of spinothalamic tract to alleviate pain
• Blocks the transmission of pain, thermal, and crude touch sensations
• Fine tactile pressure and contraceptive remains intact as the dorsal column is unaffected.
• Used for patients with severe intractable pain, such as those suffering from terminal cancer where use of opiates are proven inadequate; Anterolateral cordotomy becomes an option for pain management.
Brown-Sequard Syndrome
• What happens to your sensory modalities below the level of the spinal cord hemisection?
For pain, temperature, and crude touch
Pain and temperature senses from the opposite side of the body, at least 2 segments and more below the hemisection, is lost as the sensory inputs are carried over by second-order spinal projection neurons of the spinothalamic tract which crosses over to the opposite side of the spinal cord (below the hemisection)
The transection blocks the transmission of signal along the second-order spinal projection neurons of the spinothalamic tract
Pain or temperature sense form the side of the hemisection, but sufficiently below it, would be UNAFFECTED as their ascending spinothalamic projections have already decussated to the unaffected contralateral side of the spinal cord
For fine touch and proprioception
Fine touch, vibrations, two-point discrimination (2PD) and proprioceptive sense signals passing along the ipsilateral dorsal column below the level if the spinal cord hemisection is impaired as transmission is blocked.
The dorsal column fibers only terminate at the dorsal column nuclei at the level of the medulla, to synapse with the second-order neurons which decussate to the opposite side before it projects upward towards the thalamic relay nuclei.
The sensory inputs to the contralateral dorsal column would be unaffected by the left-sided hemisection.
tangina parusa tong somatic senses, ilang beses ako na stuck dito
2.3 Special Senses
Vision 👀
Definition:
The ability to bend light.
Measurement: Measured in Diopters, which is the reciprocal of the focal distance in meters.
Eye Composition: 59 diopters of refractive power total
Cornea: Contributes 2/3 of the refractive power with fixed refractive properties.
Lens: Contributes 1/3 of the refractive power and is variable, altered by the suspensory ligaments (zonula fibers).
Lens Accommodation Process
Ciliary Muscles Relaxed: Increases tension on suspensory ligaments, resulting in a flat lens.
Ciliary Muscles Contracted: Decreased tension on suspensory ligaments leads to a spherical lens.
Refractive Errors
Myopia (Near-Sightedness): Light rays converge in front of the retina due to a long eyeball.
Hyperopia (Far-Sightedness): Light rays converge behind the retina due to a short eyeball.
Astigmatism: Irregular curvature of the cornea leads to multiple convergence points on the retina.
Presbyopia: Age-related loss of accommodation; difficulty in reading due to decreased ciliary body contraction ability.
Corrective Lenses
Types:
Biconcave Lenses for myopia.
Convex Lenses for hyperopia.
Cylindrical Lenses for astigmatism.
Retinal Cell Functions:
Types of Retinal Cells:
Rods: Sensitive to low-intensity light (night vision).
Cones: Sensitive to high-intensity light (day, color vision).
Bipolar Cells
Intermediate between photoreceptor cells and ganglion cells
Play a role in contrast detection.
Steps in Photoreception of Rods:
Vitamin A regenerates 11-cis rhodopsin/retinal.
Photons (light particles) converts 11-cis rhodopsin/retinal to all-trans rhodopsin/retinal.
Several intermediates from all-trans rhodopsin are formed. The most important: Metarhodopsin II.
Metarhodopsin II activates a Gt or Transducin. Transducin activates Phosphodiesterase.
Phosphodiesterase converts cGMP to 5’GMP decrease cGMP levels.
Decreased cGMP causes closure of Na+ channels. Decreases Na influx leading to hyperpolarization. The brighter the light, the greater the hyperpolarization.
Hyperpolarized photoreceptors lead to decreased glutamate
Decreased glutamate → Excitatory ionotropic glutamate receptors in bipolar and horizontal cells are inhibited.
Decreased glutamate → Inhibitory metabotropic glutamate receptors in bipolar and horizontal cells are excited and depolarized.
Increases when light strikes the eye: activity of transducin
Visual Pathways
Visual Field
Divided into left and right visual fields
Further subdivisions: nasal and temporal visual fields
Light coming from the temporal visual fields can be received by the nasal portion of the retina
On the other hand, light coming from the nasal visual field will be received by the temporal portion of the retina.
After which, signals will travel to the optic nerve → as they reach the optic chiasm, fibers coming from the nasal half of the retina will cross to the opposite side until they reach the optic tract.
Fibers from the optic tract will synapse at the dorsal lateral geniculate nucleus of the thalamus, and from there, signals will be sent to the primary visual cortex via the optic radiation.
All of the optic nerves terminate in the dorsal lateral geniculate nucleus which is located at the dorsal end of the nucleus.
Lateral Geniculate Nucleus (LGN):
Relays visual information form the optic tract to the visual cortex by way of the optic radiation.
Very accurate feature
Half of the fibers in each optic tract after passing the optic chiasm are derived from one eye and the other half is derived from the other eye
Therefore, it represents corresponding points from the two retina.
However, the signals from the two eyes are kept apart in the dorsal lateral geniculate nucleus by its 6 layers.
Another way of dividing the 6 layers:
Magnocellular Layers
Layers I & II
Receives inputs entirely from type Y ganglion cells
Provide rapidly conducting pathway to the visual cortex
Color blind
Parvocellular Layers
Receives inputs from type X ganglion cells
Transmits color
Control the transmission of signals to the visual cortex (acts as a gate to control how much signals is allowed to pass the visual cortex)
Corticofugal fibers of the occipital lobe
Reticular are of the mesencephalon
Both of which send inhibitory retrograde signals to the dorsal lateral geniculate nucleus and can turn off transmission of selected areas of the dorsal lateral geniculate nucleus.
Visual Cortex
From the DLGN, fibers will travel to the visual cortex at the occipital area.
1. Primary Visual Cortex
where direct visual signals terminate Lies along the Calcarine Fissure and up until the medial portion of the occipital cortex.
Signals coming from the macular area of the retina, terminate at the most lateral aspect of the primary visual cortex, while signals coming from the peripheral portions of the retina terminate at the medial aspect of the cortex.
The larger area of the cortex represents the macula or the center most portion of the retina.
2. Secondary Visual Cortex
Visual association area
Anatomically surrounds the primary visual cortex
Analyze the meaning of the things that we see
Clinical correlations:
Complete Blindness of the Affected Eye
Any condition that can damage or impinge the optic nerve
Bitemporal Hemianopsia
Destruction of the optic chiasm
Fibers coming from the nasal portion of the retina will be affected
Light rays received by the nasal portion of the retina is from the temporal aspect of the visual field, THEREFORE, patients will have tunnel vision, a condition called Bitemporal Hemianopsia
Homonymous Hemianopia
Destruction of the radiation
What would happen if the left optic tract or optic radiation were affected?
Following the visual pathway, signals that are transmitted at the left optic tract will be coming from the right visual field. → A person will only be able to see half of the image.
Eye Movement and Control
Fixation Movement of the Eye
Fixation is an important function of the eye because it enables us to look for specific objects in our field of vision and ultimately hold our gaze firm on those objects.
Fixation is controlled by 2 neuronal mechanisms:
Voluntary Fixation Mechanism
Controlled by the Premotor Region of the Frontal Lobe
Function: to move the eyes in order to look for the objects that they want to fix or focus on
Damage to this voluntary fixation mechanism results to the person being unable to unlock the eye easily from one fixation to the next.
Involuntary Fixation Mechanism
Controlled by the Secondary Visual Cortex
Function: to lock on objects once found by the eye
Damage to this area will result in difficulty in keeping the eye still
Patient is unable to direct the eye towards the desired object
Autonomic Control of the Pupillary
Parasympathetic
Parasympathetic preganglionic fiber of the vision: Edinger-Westphal Nucleus along the midbrain (visceral nucleus position of the third nerve).
Preganglionic fibers will synapse to postganglionic parasympathetic fibers along the ciliary nerves.
Ciliary muscles: focuses the lens
Sphincter of Iris: constricts the pupil
Pathway: Edinger Westphal Nucleus → Oculomotor Nerve → Ciliary ganglion → Ciliary nerve
Sympathetic
Sympathetic innervations of the eye will originate from the intermediolateral horn cells of T1 of the spine → travel upwards to the superior cervical ganglion → tributaries of carotid plexus until the postganglionic sympathetic fiber reaches the eyes directly.
Sympathetic fibers travel directly to the eye
Control of Pupillary Diameter
Miosis: decrease in pupillary aperture
Mydriasis: increase in pupillary aperture
Visual Neural Pathways and Visual Field Defects
Visual Neural Pathways
Visual Pathways allow visual information to travel from the retina to the occipital lobe
The left and right visual fields represent the entire area we can see peripherally when we focus on a central point
Temporal retina: portion of the retina that receives information from the nasal visual field
Nasal retina: portion of the retina that receives information from the temporal visual field (portion of retina closest to the nose)
Visual information from the retina is transmitted via ganglion cells forming the optic nerves, which extend to the optic chiasm.
Here, some optic nerves from the nasal retina cross to the opposite brain side.
The thalamus receives sensory information through optic tracts and lateral geniculate nuclei, relaying it to the primary visual cortex in the occipital lobe via optic radiations.
Some nerves also connect to the superior colliculi for pupillary reflexes.
Visual Pathway
Nerve (blue) on the left eyeball
captures images from the left nasal field
carries visual information to the left lateral geniculate body
synapses with another nerve that carries information to the occipital lobe
Nerve (blue) on the right eyeball
captures images from the right temporal field
carries visual information to the optic nerve, cross over the optic chiasm, and synapse with neurons of the left lateral geniculate body
the other nerves will carry information to the occipital lobe
Nerve (orange) on the left eyeball
capture images from the left temporal field
carries visual information by crossing over the optic chiasm, and synapse with neurons of the right lateral geniculate body
the other nerves will carry the information to the optic tract, then to the occipital lobe
Nerve (orange) on the right eyeball
capture images from the right nasal field
carries visual information to the right lateral geniculate body where it will synapse with another nerve
the other nerves will carry the information to the occipital lobe
Visual Lesions
Left Anopia
Location of the Lesion: on the left optic nerve
Cause: orange and blue optic nerves on the left side cannot bring information down
Clinical Manifestation: blindness on the left eye
Bitemporal Hemianopia
Location of the Lesion: on the optic chiasm
Cause: may be due to a pituitary tumor; nerves that cross over (especially nerves that capture images on the temporal field) cannot bring information down
Clinical Manifestation: blindness in the temporal visual field
Bitemporal Hemianopia
Location of the Lesion: on the optic chiasm
Cause: may be due to a pituitary tumor; nerves that cross over (especially nerves that capture images on the temporal field) cannot bring information down
Clinical Manifestation: blindness in the temporal visual field
Right Homonymous Hemianopia
Location of the Lesion: below the optic chiasm
Cause: loss of visual field that the blue nerves carry
Clinical Manifestation: loss of vision on the left nasal field and right temporal field
Right Homonymous Superior Quadrantanopia
Location of the Lesion: on the nerve tract bundles
Cause: nerves (black) cannot bring information down to the occipital lobe
Clinical Manifestation: field deficit in the superior field of both eyes on the same side
Other Visual Field Problems
Central Scotoma (Macular Degeneration)
loss of central vision or high quality vision because of a damaged macula
Constricted Visual Fields
macula is preserved but all other parts are damaged
manifests as tunnel vision (loss of peripheral vision)
seen in glaucoma and retinal pigmentosa
Light Reflex
The visual pathway consists of the eyeball, optic nerve, optic chiasm, optic tract, and the occipital lobe.
The midbrain contains structures and nuclei crucial for eye reflex, including the superior colliculus, pretectal nucleus, and the Edinger Westphal nucleus (where CN III originates).
CN III (oculomotor nerve) contains parasympathetic nerve fibers that travel to the ciliary ganglion and synapse with another neuron
Key structures in the eyeball for light reflex:
Ciliary muscles: change lens shape to control light entry.
Iris: contains muscles controlling pupil size.
Circular muscle: constricts pupil (miosis) via parasympathetic fibers of CN III.
Radial muscle: dilates pupil (mydriasis) via sympathetic nerve fibers.
Light Reflex Pathways:
Direct Pupillary Reflex: pupil size change in the eye with the light.
Consensual Pupillary Reflex: pupil size change in the opposite eye.
The Light Reflex Pathway:
Light on the left eyeball is transmitted by the optic nerve ipsilaterally to the left lateral geniculate body.
Some nerves cross over at the optic chiasm to the right lateral geniculate body (contralaterally).
Information travels along the optic tract to the occipital lobe.
Optic nerves also synapse with the pretectal nucleus in the midbrain.
The pretectal nucleus sends information to both Edinger Westphal nuclei.
Oculomotor nerves carry the signal to the ciliary ganglion on both sides.
Short ciliary neurons transmit the signal to the iris, causing pupil constriction.
Hearing and Equilibrium 👂
Ear Anatomy
Outer Ear: Pinna and auditory canal; involved in sound localization and collection.
Middle Ear: Contains the tympanic membrane and ossicles (malleus, incus, stapes) that inserts to the oval window; Functions to amplify sound via impedance matching
Inner Ear: Composed of the cochlea, vestibule, and semicircular canals; involved in sound detection and balance. Bony labyrinth (semicircular canals, cochlea, vestibule), Membranous labyrinth (series of ducts)
Endolymph is seen in the scala media/cochlear duct high in potassium
Perilymph is seen in the scala vestibuli and scala tympani high in sodium
Hearing Process
Sound waves cause the cochlea to vibrate, bending cilia on inner hair cells due to the stiffness difference between the basilar and tectorial membranes.
Depolarization of inner hair cells is caused by K+ influx (endolymph is rich in K+).
Bending cilia in one direction causes depolarization, and in the opposite direction causes hyperpolarization, resulting in an oscillating potential called the cochlear microphonic potential.
Outer Hair Cells:
Respond to sound like inner hair cells.
Contain the motor protein Prestin.
Depolarization shortens outer hair cells, hyperpolarization lengthens them.
Damage reduces sound clarity.
Place Theory of Hearing:
Inner hair cells near the base (oval and round windows): respond to high-frequency sounds.
Inner hair cells near the apex (helicotrema): respond to low-frequency sounds.
Damage near the oval window leads to inability to hear high-frequency sound.
Central Auditory Pathways:
Fibers ascend through the lateral lemniscus to the inferior colliculus to the medial geniculate nucleus of the thalamus to the auditory cortex.
Fibers can be crossed or uncrossed.
Lesions of the cochlea of one ear cause unilateral deafness.
Central unilateral lesions may not cause unilateral deafness due to the mixing of ascending auditory fibers.
(+) Tonotopic representation of all frequencies at all levels of the central auditory pathway.
Vestibular System 🫨
Allows reflex adjustments of the head, eyes, and postural muscles for stable visual image and steady posture.
Consists of: 3 Perpendicular Semicircular Canals: detect angular acceleration.
detect head position in space: Otolith organs:
Utricle: detects horizontal (linear) acceleration.
Saccule: detects vertical (linear) acceleration.
When the head is rotated to the right, both left and right eyes deviate towards the left.
Receptors: Hair Cells
Cupula: gelatinous structure
Kinocilium: single long cilium of hair cell
Stereocilia: smaller cilia of hair cell
Rule:
Stereocilia bends towards kinocilium: depolarization
Stereocilia bends away from kinocilium: hyperpolarization
Nystagmus:
Direction is frequently horizontal, but can be vertical or rotatory.
Direction of Nystagmus: same direction as head rotation.
Post-rotatory Nystagmus: occurs in the opposite direction of the head rotation.
Caused by continued movement of endolymph in the semicircular canals, bending the cupula and stimulating hair cells.
Olfaction 👃
Olfactory Neurons:
Capable of reproduction. (non-permanent cells)
Olfactory epithelium innervated by:
CN I (Olfactory Nerve): detects scent.
CN V (Trigeminal Nerve): detects noxious/painful stimuli (e.g., ammonia).
Odorant receptors are located on neurons in the olfactory epithelium and project to mitral cells, then directly to the olfactory cortex.
Olfaction Function:
Olfactory memories are found in the entorhinal cortex.
Cribriform plate fracture damages CN I but not CN V: results in anosmia but intact response to ammonia.
Mechanism of Action:
Odorant molecules bind to receptors in the cilia of olfactory receptor cells.
Golf are used as transducers to activate the cAMP second messenger system.
Increased cAMP opens Na+ channels, causing depolarizing receptor potential.
Action potentials are generated and propagated once the threshold is reached.
Taste👅
Taste Receptors: not true neurons, not synonymous with taste buds.
Taste receptor is a type of chemoreceptor.
Innervated by afferents of CN VII, IX, X
Anterior 2/3 of tongue: CN VII (chorda tympani - Facial Nerve)
Posterior 1/3 of tongue: CN IX (Glossopharyngeal Nerve)
Back of throat and epiglottis: CN X (Vagus Nerve)
Taste Pathway:
CN VII, CN IX, and CN X → enter medulla → ascend in the solitary tract → terminate on second-order taste neurons in the solitary nucleus → ascend ipsilaterally to the ventral posteromedial nucleus of the thalamus → end at the taste cortex.
Cause of dysgeusia (altered taste) following tooth extraction: damage to the gustatory afferent nerves.
2.4 Motor Cortex 🏃♂️➡️
Definition: Movement, whether voluntary or involuntary, is produced by contraction of muscles.
Types:
Involuntary: reflex movements
Voluntary: Result from the integrative activity of multiple neural inputs from spinal cord, brainstem, midbrain, and cortical levels, which regulate body posture and enable coordinated movements.
Voluntary Motor Control
Origins: Commands for voluntary movements originate from cortical association areas.
Involved Structures:
Cortex: Responsible for execution of movement; relayed by corticospinal and corticobulbar tracts to motor neurons.
Basal Ganglia: Works with the cerebellum and cortex to plan movements.
Cerebellum: Provides feedback to adjust and smoothen movements.
Key Areas in the Motor Cortex
Structure & Function
The motor cortex is divided into three subareas, each with a topographical representation of muscle groups:
Primary Motor Cortex (M1):
Located in the precentral gyrus of the frontal lobe; involved in executing movements.
Supplementary Motor Area (SMA):
Involved in organizing or planning movements; responsible for mental rehearsal.
Premotor Area:
Planning movement; sets posture at the start of planned movements.
Primary Somatosensory Area
Located in the parietal lobe; projects to the premotor cortex.
Contributes to descending pathways for movement execution.
Involved in discrete and detailed movements, especially of distal segments (hands, fingers).
Lateral Corticospinal Tract & Corticobulbar Tract
Composition: Fibers descending from the motor cortex; majority decussate (cross) at the lower medulla.
Lateral Corticospinal Tract (80%): Control of skilled voluntary movements of the contralateral side.
Anterior Corticospinal Tract (20%): Controls bilateral postural movements.
Diseases Affecting Motor Control
Identification of clinical findings:
Spastic paralysis, hyperreflexia, increased muscle tone, muscle weakness, disuse atrophy, and decreased speed of voluntary movements.
Lesions generally affect the CONTRALATERAL SIDE (e.g., right side lesion affects left side motor function).
Motor Pathways
Descending Motor Pathways:
Brainstem:
Medulla, Pons, Midbrain.
Acts as a command signal station for movement and equilibrium.
Medial Brainstem Pathways: Control of axial and proximal muscles.
Lateral Brainstem Pathways: Control of distal muscles.
Medial Brainstem Pathways:
Medial Brainstem Pathways Overview:
Work in concert with the ventral corticospinal tract.
Descend into the ipsilateral ventral columns of the spinal cord.
Terminate predominantly on interneurons and long propriospinal neurons in the ventromedial part of the ventral horn to control axial and proximal muscles.
A few medial pathway neurons synapse directly on motor neurons controlling axial muscles.
Specific Medial Brainstem Pathways
Medial and Lateral Vestibulospinal Tracts: Involved in vestibular function.
Medial vestibulospinal tract: Controls neck musculature.
Lateral vestibulospinal tract: Activates motor neurons to anti-gravity muscles to control posture and balance.
Pontine and Medullary Reticulospinal Tracts: Project to all spinal levels and are involved in the maintenance of posture and modulating muscle tone, especially via input to the gamma motor neurons.
Pontine reticulospinal tract: Excitatory.
Medullary reticulospinal tract: Inhibitory.
Tectospinal Tract: originates in the superior colliculus of the midbrain,
Project to the cervical spinal cord
Innervate motor neurons responsible for neck movements
Responsible for orienting the head and neck during eye movement
Lateral Brainstem Pathway Function: The main control of distal muscles arises from the lateral corticospinal tract, but neurons within the red nucleus in the midbrain also contribute. These cross the midline and project to interneurons in the dorsolateral part of the spinal ventral horn, influencing motor neurons that control distal limb muscles.
Rubrospinal Tract:
Originates in the red nucleus in the midbrain.
Projects to spinal cord interneurons and alpha motor neurons.
Stimulates flexor muscles and inhibits extensor muscles.
Clinical Relevance: The lateral brainstem pathway (specifically the rubrospinal tract) may play a role in the posture typical of decorticate rigidity.
Decorticate Posturing:
Can result from upper midbrain injury.
Involves interruption of pathways from the cortex (corticospinal and corticobulbar tracts).
With rubrospinal tracts intact (excitatory to limb flexors), it leads to:
Flexed upper limbs.
Extended lower limbs with toes pointed slightly inward.
Extended head.
Decerebrate Posturing:
Cause: Midcollicular lesion.
Mechanism:
Interruption of pathways from the cortex (corticospinal & corticobulbar tracts).
Interruption of pathways from the red nucleus (rubrospinal tract).
Pontine reticulospinal tracts remain intact and are excitatory to limb extensors.
Resulting Posture:
Upper limbs: Extended with fingers flexed.
Lower limbs: Extended with toes pointing inward.
Head & Neck: Extended
Effects of Transection at the Spinal Cord
Paraplegia: Loss of voluntary movements below the level of the lesion.
Loss of conscious sensation: Below the level of the lesion.
Spinal Shock:
Absent reflexes.
Limbs flaccid immediately after transection.
Partial recovery may occur after some time (e.g., several hours to a few weeks).
Reflexes are NOT chronically suppressed after spinal cord transection.
Level-Specific Effects:
C7 transection: HR and BP decreases.
C3 transection: Breathing stops.
C1 transection: Death
Lesions above Lateral Vestibular Nucleus: decerebrate rigidity
Lesions between Pontine Reticular Formation and Midbrain: decerebrate rigidity
Decerebrate rigidity is spasticity due to facilitation of myotatic stretch reflex
Lesions above Red Nucleus: decorticate posturing and intact tonic neck reflexes
Basal Ganglia & Cerebellum
Basal Ganglia: Function and Structure
Functions:
Initiation and execution of voluntary movement; cognitive motor control.
Components:(5)
Caudate nucleus (Striatum)
Putamen (striatum, lentiform nucleus)
Globus pallidus (lentiform nucleus)
Globus pallidus externa (GPe)
Globus pallidus interna (GPi)
Subthalamic nucleus. (STN)
Substantia nigra (SN)
Substantia nigra pars compacta (SNc)
Substantia nigra pars reticulata (SNr)
Neurotransmitters: GABA (inhibitory), glutamate (excitatory), dopamine (both inhibitory and excitatory based on receptor).
Disorders of the Basal Ganglia
Parkinson's Disease: Result from loss of dopaminergic neurons in the substantia nigra; leads to hypokinetic movement disorder. Destruction of SNc neurons
Huntington Disease: Damage to the striatum; leads to hyperkinetic movement disorders with abnormal movement patterns.
Other disorders include
athetosis - (globus pallidus) spontaneous and often continuous writhing movements of the hand, arm, neck, or face
hemiballismus - (STN) sudden flailing movements of an entire limb
chorea. - (putamen)
The Role of the Cerebellum
Functions as an accessory motor system; establishes balance and coordination.
Anatomical Divisions:
Anterior, Posterior, Flocculonodular lobes.
Functional Divisions:
Vestibulocerebellum: For balance and eye movements
Spinocerebellum: For synergy of movement.
Cerebrocerebellum: For planning and initiation of movement.
Cytoarchitectural layers:
Molecular layer: External, contains axons from granule cells, Purkinje cell dendrites, basket cells, and stellate cells.
Purkinje cell layer: One cell thick, contains Purkinje cells (major inhibitory output pathway).
Granular layer: Internal, contains granule cells and Golgi cells; receives input from Mossy fibers.
Cell Types in the Cerebellum
Purkinje Cells: Large neurons, send inhibitory (GABA-ergic) projections to deep cerebellar nuclei.
Granule Cells: Receive excitatory input from mossy fibers and excite Purkinje cells (release glutamate).
Interneurons: Include Basket and stellate cells (molecular layer) and Golgi cells (granular layer).
Fundamental circuits:
Climbing fibers: From inferior olivary nuclei, provide strong excitatory input to Purkinje cells.
Mossy fibers: Provide proprioceptive input from the body and cortex via pontine nuclei, weakly excite Purkinje cells via granule cells.
Main inhibitory output: Purkinje cells inhibit deep cerebellar nuclei.
Cerebellar Dysfunction and Clinical Conditions
Ataxia: Inability to coordinate movements. (dysdiadochokinesia)
Intention Tremor: Tremors (<5 Hz) that increase as a limb approaches a target.
Dysmetria: Overshooting targeted movements; a consequence of cerebellar lesions.
Clinical Relevance: Cerebellar disorders typically express symptoms ipsilaterally; resulting in lack of coordination in actions.
masakit ang ulo pero mas masakit pag walang ulo 🥀🥀🥀
2.5 Reflex
Definition of Terms
2 Types of Motor Output:
Reflex (involuntary)
Voluntary
Reflex:
Mechanism by which sensory impulse is automatically converted into a motor effect through the involvement of the CNS.
Reflex Arc
The reflex arc is the basic unit of integrated reflex activity.
Components of Reflex Arc:
Sense organ
Afferent neuron
One or more synapses within a central integrating station
Efferent neuron
Effector organ
Activity begins with a sensory receptor that generates a receptor potential proportional to stimulus strength.
This potential generates all-or-none action potentials in the afferent nerve, with action potential frequency proportional to the receptor potential strength.
In the CNS, responses are graded based on excitatory postsynaptic potentials (EPSPs) and inhibitory postsynaptic potentials (IPSPs) at synaptic junctions.
When action potentials reach the effector, a graded response occurs; if sufficient, action potentials trigger muscle contraction. Multiple inputs can modify reflex arc activity.
Types of Reflex Arcs
Monosynaptic Reflex:
Involves a single synapse between afferent and efferent neurons.
Examples include stretch reflex and inverse stretch reflex.
Polysynaptic Reflex:
Includes interneurons interspersed between afferent and efferent neurons; can have 2 to 100 synapses.
Adequate Stimulus
Reflex arc activity magnitude is proportional to stimulus strength, leading to all-or-none action potentials in the afferent nerve.
Stereotyped Response
Reflex responses are highly specific; a particular stimulus elicits a particular response.
Muscle Spindle
Muscle Spindle:
Fusiform structures within the muscle belly, detecting mechanical stretch, consist of specialized receptors that aid in myotatic reflexes.
Essential Elements of a Muscle Spindle:
Intrafusal Fibers: Specialized fibers with contracted polar ends.
Afferent Nerves: Type 1 & 2 myelinated fibers from intrafusal fibers.
Efferent Nerves: Myelinated efferent nerves for intrafusal fiber regions.
Intrafusal Fibers
Intrafusal fibers are one of the three essential elements of a muscle spindle.
They are specialized intrafusal muscle fibers with contractile polar ends and a non-contractile center.
Intrafusal fibers are positioned in parallel to the extrafusal fibers, with the ends of the spindle capsule attached to the tendons at either end of the muscle.
Function:
They do not contribute to the overall contractile force of the muscle.
They serve a pure sensory function.
They can contract with the extrafusal muscle fibers.
Nuclear Bag Fiber:
Contains many nuclei in a dilated central region.
Subtypes include dynamic and static nuclear bag fibers.
Nuclear Chain Fiber:
Thinner and shorter, lacking a definite bag structure.
Afferent Nerves
Primary Ending (Group Ia):
Wraps around dynamic/static fibers to relay fiber length and velocity information.
Secondary Ending (Group II):
Special flower spray endings that function primarily based on static length.
Efferent Nerves
Alpha (somatic) Motor Neurons:
Innervate extrafusal fibers, crucial for muscle contraction.
Directly command muscle contractions
Form the final common pathway for neural influences affecting muscle contraction.
Gamma Motor Neurons:
innervates and contracts intrafusal muscle fibers to improve the sensitivity of these fibers and consequently, resulting in the myotatic reflex
α-γ Coactivation:
Adjusts muscle spindle sensitivity during contractions, ensuring accurate responses.
Monosynaptic Reflexes
Definition: The simplest reflex arc, involving a single synapse between the afferent and efferent neurons.
Involved in: Stretch reflex and inverse stretch reflex.
Example: The myotatic reflex (stretch reflex) is monosynaptic. It is stimulated by a sudden passive stretch of muscle belly fibers and produces a muscle contraction in the agonist muscle or muscle group with simultaneous agonist relaxation response.
Mechanism:
When a skeletal muscle with an intact nerve supply is stretched, it contracts.
The sense organ is the muscle spindle.
Impulses from the spindle are transmitted to the CNS by fast sensory fibers that pass directly to the motor neurons that supply the same muscle.
The neurons at the central synapse are glutamate.
Stretch Reflex Function: Muscle spindle reflexes oppose (correct for) increases in muscle length (stretch).
Stretch stimulates group Ia afferent fibers → synapse directly on α-motor neurons → innervates the homonymous muscle → contraction in the muscle → shortening & decreasing the stretch on the muscle spindle and returning it to its original length
Reciprocal Innervation:
In a stretch reflex, reciprocal innervation coordinates muscle action: synergistic muscles contract, and opposing (antagonist) muscles relax. Ia fibers from the stretched muscle inhibit antagonist motor neurons through inhibitory interneurons in the spinal cord.
Inverse Stretch Reflex:
Stimulus: muscle contraction.
Response: muscle relaxation.
When the tension becomes great enough, contraction suddenly ceases, and the muscle relaxes.
The sense organ/receptor used in the inverse stretch reflex is the Golgi tendon organ.
Golgi tendon organs are located within tendons and contain Ib sensory nerve fibers among tendon fascicles. These fibers, stimulated by both passive stretch and active contraction, detect muscle tension. When activated, Ib fibers trigger Inhibitory Postsynaptic Potentials (IPSPs) in motor neurons of the contracting muscle (via spinal cord interneurons) and excite motor neurons of antagonist muscles.
Excessive tension of the muscles → stimulates sensory receptors in the Golgi tendon organ → signals from the receptors are transmitted through a sensory afferent nerve Ib fiber → excites an inhibitory interneuron in the spinal cord → inhibiting anterior motor neuron activity → causing muscle relaxation → protecting the muscle against excessive tension
Polysynaptic Reflex
Definition: Reflex arcs in which interneurons are interposed between afferent and efferent neurons.
Characteristics:
Can involve one to many hundreds of synapses.
The withdrawal reflex is a typical polysynaptic reflex.
Withdrawal Reflex: Occurs in response to a noxious stimulus to the skin or subcutaneous tissue and muscle.
The response is flexor muscle contraction and inhibition of extensor muscles, so that the body part stimulated is flexed and withdrawn from the stimulus.
Crossed Extensor Response:
When a strong stimulus is applied to a limb, the response includes not only flexion and withdrawal of that limb but also extension of the opposite limb.
This crossed extensor response is part of the withdrawal reflex.
The withdrawal reflex serves a protective function as flexion of the stimulated limb gets it away from the source of irritation, and extension of the other limb supports the body.
Prolonged Response:
Strong stimuli can generate activity in the interneuron pool that spreads to all four extremities.
It also causes prolonged flexion and sometimes a series of flexion movements.
This prolonged response is due to prolonged, repeated firing of the motor neurons (after-discharge) that is due to continued bombardment of motor neurons by impulses arriving by complicated and circuitous polysynaptic paths.
Clinical Reflections
Knee Jerk Reflex:
Tapping the patellar tendon elicits the knee jerk, which is a stretch reflex of the quadriceps femoris muscle.
It's a deep tendon reflex (DTR) used in neurologic exams.
Absence can indicate abnormalities in the reflex arc (muscle spindle, Ia afferent nerve fibers, or motor neurons to the quadriceps).
A hyperactive reflex can signify interruption of corticospinal and other descending pathways that suppress the reflex arc.
Clonus:
Characterized by regular, repetitive, rhythmic muscle contractions due to increased γ-motor neuron activity.
Sustained clonus (5+ beats) is considered abnormal.
Ankle clonus involves rhythmic plantar flexion at the ankle, initiated by brisk, maintained dorsiflexion of the foot.
Hyperactive muscle spindles contribute to clonus.
Spinal Cord Injury:
Spinal cord transection leads to spinal shock, with depressed spinal reflex responses.
Reflexes return and become hyperactive after the spinal shock period.
Quadriplegia can result in a low threshold for the withdrawal reflex, causing prolonged withdrawal and flexion-extension patterns in limbs.
Noxious stimuli may trigger autonomic responses like bladder/rectum evacuation, sweating, pallor, and blood pressure swings (mass reflex).
di kasi pala pang kids yung physio🥀🥀🥀
2.6 Cerebral Cortex 🧠
Introduction
The human brain is complex, characterized by intricate neural circuits.
The cerebral cortex is the largest and most studied part of the nervous system, though many of its functions remain ambiguous.
Structure of the Cerebral Cortex
Functional Portion: Thin layer of neurons spanning the convoluted areas of the brain.
Neuron Count: Approximately 80 billion neurons.
Components:
Neuronal cell bodies
Axons from the cell bodies
Axon terminals in different cortex layers
Gray matter: The structure/layer that makes up the cerebral cerebral cortex (2-5 mm thick). Contains neuron cell bodies; densely packed due to infoldings and convolutions, increasing surface area for more neural circuits.
White matter: myelinated axons that transmit signals to/from the cortex.
Other areas with concentrated number of neuronal cell bodies are in the:
Subcortical deep nuclei of the brain
Gray matter of the spinal cord
Neocortex
Definition: The newest part of the cerebral cortex, evolved into a 6-layer structure that distinguishes mammals from other animals.
Layers: Six layers comprise about 90% of the cerebral cortex; humans possess the most expanded neocortex among mammals.
Allocortex: Comprises 10% of the cerebral cortex; contains 3-4 layers.
Cortical Association Areas
Function: Major areas processing signals from sensory and motor regions; include:
Parieto-occipitotemporal Association Area: (Aka posterior association area) Links visuospatial perception and language.
Prefrontal Association Area: (Aka frontal lobe association area, anterior association area or prefrontal cortex) Involved in motor planning, language, and judgment. Aka the central executive for working memory and receiving inputs from the rest of the cerebral cortex
Limbic Association Area: (Aka temporal association area or the limbic cortex) Associated with behavior, emotions, and motivation.
Functions of Specialized Areas
Broca's Area: Planning complex movements and thought elaboration; associated with word formation and speech production.
Wernicke's Area: Language comprehension; damage leads to difficulties in understanding language (Wernicke's aphasia).
both found in left hemisphere
Three Types of Subcortical Fibers
found in the white matter, Bundles of fibers which relay information to and from specific areas of the brain.
Note: Any lesions brought by disease, trauma, or removal of brain tissues may affect cell bodies of neurons and white matter axonal fibers, which connect one region of the brain to another
Association Fibers:
Interconnect regions of the brain within the same hemisphere.
Short association fibers connect areas in adjacent gyri.
Long association fibers connect areas far away from each other.
Commissural Fibers:
Interconnect areas of the cerebral cortex in one hemisphere with the same areas in the opposite hemisphere.
Allow coordination of cortical activity across the hemispheres.
Allow information received from the right hemisphere to be processed by the left hemisphere (such as in language, comprehension, and expression).
Projection Fibers:
Travel to and from the cerebral cortex.
May arise from the thalamus.
Descend from the cerebral cortex to the basal ganglia, brain stem, or the spinal cord.
Arise as ascending and descending fibers from all parts of the cerebral cortex (corona radiata) and converge into a compact bundle (internal capsule).
Corpus Callosum
The corpus callosum is the largest set of commissural fibers in the brain.
Commissural fibers interconnect areas of the cerebral cortex in one hemisphere with the same area in the opposite hemisphere.
It allows information received from one hemisphere to be processed by the other hemisphere, which is crucial for functions like language.
Neocortex Layers
Layer I: Molecular layer
Layer II: External granular layer
Layer III: External pyramidal layer
Layer IV: Internal granular layer
Layer V: Internal pyramidal layer; includes Betz cells, important for motor functions.
Layer VI: Multiform layer projecting to the thalamus.
Vertical Cortical Columns:
The neocortex (the main part of the cerebral cortex) is arranged into vertical cortical columns.
These columns are functional units of the neocortex.
They are connected with interneurons and receive inputs from:
Adjacent regions of the same hemisphere.
Inputs from the opposite hemisphere.
Working memory allows us to keep our sensory memories about our surroundings within our stream of consciousness at the present moment.
Columns:
200-800 um in diameter
About 2 million cortical columns in the human brain
Mini columns:
24-80 um in diameter
2x108 mini columns
Learning And Memory
Learning
Learning is the acquisition of information and skills through experience.
It results in a relatively permanent change in an organism’s behavior.
Learning is dependent on memory; without memory, learning is not possible.
Factors that affect learning and memory include:
Experience
Punishment or reward
Background of the person (mood and motivation)
Memory
Memory allows people to recall past events and information.
This includes recent or remote, episodic, and semantic memories.
It also enables the performance of previously learned skills without practice after time has passed.
Memory has a mechanism where an experience is incorporated into the organism, later causing an adaptive change in behavior.
Working memory allows us to keep sensory memories of our surroundings in our stream of consciousness.
Types of Memory
Declarative (Explicit) Memory:
Aka Explicit or Recognition
Involves conscious recall of events and facts, like faces and names.
Semantic Memory (FACTS): Words, rules, language, etc.
Episodic Memory (EVENTS): Can be converted into reflexive (long-term) memory by repetition ("training").
Structures involved: Hippocampus, medial temporal lobe, other parts of the neocortex, diencephalon.
Non-Declarative (Implicit) Memory:
Aka Implicit memory
Used in parallel, involving different neural structures.
Reflexive and effortless.
Does not involve conscious awareness.
Usually stored in long-term memory.
Not dependent on the hippocampus.
Example: driving a car becoming an effortless process.
Procedural Memory: Skills and habits; involves the striatum, motor cortex, cerebellum (e.g., playing an instrument, sports).
Associative Memory: Making connections between stimuli and behavioral responses (e.g., classical and operant conditioning); involves the amygdala and cerebellum.
Types:
Classical (Passive) Conditioning: conditioned reflex acquired by repeatedly pairing a conditioned stimulus to an unconditioned stimulus that produces a response
Operant (Instrumental) Conditioning: thought to perform task in order to obtain a reward or a punishment
One trial or Aversion Conditioning: process by which a noxious or unpleasant stimulus is paired with an undesired behavior
Complex Learning (This type of learning involves the neocortex.)
Imprinting: This involves a young animal forming a behavioral attachment to a parent, usually dependent on a stimulus and occurring during an early critical period.
Latent learning: This is learning that occurs in comfortable surroundings due to familiarity.
Priming: This refers to the facilitation of recognition of words or objects after prior exposure.
Vicarious or Observational learning: This is when humans imitate and follow others or learn from the experiences of others.
Non-Associative Learning:
Habituation: decrease in behavioral response with repeated stimulus.
Sensitization: enhancement of reflex response to stimuli.
Involves reflex pathways.
Memory Systems According to Durability
As classified by Ganong:
Immediate recall of ongoing events.
Short-term Memory: Recent memory of events occurring seconds to hours or days before; vulnerable to erasure; includes working memory (temporary storage for planning future action).
Long-term Memory: Remote memory of events from the distant past; resistant and persists despite brain damage; recalled by associations (sight, smell, words, emotions, etc.).
As classified by Schmidt:
Sensory Memory: First storage of sensory stimuli; information is extracted and encoded for short-term stores; forgetting begins immediately.
Primary Memory: Temporary storage of verbally coded materials; corresponds to short-term memory; non-verbal material is transferred to secondary memory.
Secondary Memory: Large long-term storage; information stored by "significance"; forgetting possible through proactive and retroactive inhibition (proactive inhibition is more important).
Tertiary Memory: Engrams (memory traces) for things like one's name and abilities used daily; result of years of practice; never forgotten.
Long Term Potentiation (LTP)
First discovered by Terje Lomo and Timothy Bliss.
Originally studied in the hippocampus.
The hippocampus is one of the neural substructures related to explicit or declarative memories.
LTP also operates outside the hippocampus, in areas like the neocortex, cerebellum, basal ganglia, and amygdala.
In humans, LTP is associated with declarative memories.
Declarative Memory System
Working Memory:
1. A form of short-term memory.
Keeps recent events or moment-to-moment results of mental processing "in mind."
Involves sensory and primary memories, lasting milliseconds to seconds.
Sensory and primary memories are subject to immediate recall or retrieval.
Information needed for ongoing tasks is picked up from sensory cortices, association areas, and long-term memories.
This information is brought into conscious awareness in working memory.
Prefrontal Cortex (PFC):
Controls the temporary memory space in working memory.
Acts as a central executive.
Medial Temporal Lobe:
Converts short-term memory into long-term or remote memories.
Long-term memories are stored in and retrieved from various parts of the neocortex.
Includes the hippocampus.
2. PFC as Central Executive of Working Memory:
Allows us to keep sensory and short-term memories about our surroundings.
Retrieves information from long-term memory for everyday tasks.
Different parts of the neocortex may be involved in a network for working memory.
Important for decision-making and everyday tasks.
Damage to the PFC results in difficulties with decision-making and regulating emotional responses.
Four Components of Working Memory:
Central Executive (in PFC): steers information into rehearsal systems and the episodic buffer.
Rehearsal Systems: temporary memory stores.
Verbal System (phonological loop): retains verbal memories.
Visuospatial System (visuospatial sketchpad): retains visual and spatial aspects.
Episodic Buffer: temporary storage, integrates information from various sources.
EEG Waves
Made up of alternating excitatory and inhibitory synaptic potentials in pyramidal cells of the cerebral cortex.
Cortical Evoked Potential: changes in the EEG that reflect synaptic potentials evoked in a large number of neurons.
Gamma Rhythm (30-80Hz) in the EEG may "bind" together diverse sensory information into a single percept and action.
Absence seizures are generalized nonconvulsive seizures with spike-and-wave discharge in the EEG.
Alpha rhythm/waves disappear when a patient's eyes are open.
Sleep
Due to an active inhibitory process, not just fatigue of reticular activating systems.
Possible cause: secretion of Muramyl Peptide.
Young Adults: 25% REM Sleep.
Newborns: 50% REM Sleep.
REM sleep duration decreases with age, alcohol, benzodiazepines, and amphetamines.
From NREM to Awake state:
Increase in: Norepinephrine, Serotonin, Histamine.
Decrease in: Acetylcholine, GABA.
Narcolepsy characteristics:
Starts with REM rather than NREM sleep.
Associated with Class II antigen of MHC on Chromosome 6.
Fewer hypocretin (orexin)-producing neurons.
Language
Corpus Callosum: for interhemispheric communication.
(R) Cerebral Hemisphere:
NON-DOMINANT or REPRESENTATIONAL hemisphere in most
(R) handed people.
Dominant in facial expression, intonation, body language, spatial tasks.
Left Cerebral Hemisphere:
Usually dominant in language.
Lesions here cause aphasia.
DOMINANT or CATEGORICAL hemisphere in most right-handed people.
Wernicke Aphasia: receptive aphasia – “can say, but can’t understand” (Mnemonic: “Wordy Wernicke”).
Broca Aphasia: expressive aphasia – “can understand, but cannot say” (Mnemonic: “Broken Broca”).
Clinical Correlates
Damage to:
Angular Gyrus in the categorical hemisphere: Anomic Aphasia.
Inferior parietal lobule: unilateral attention and neglect.
Parietal Lobe of the representational hemisphere: Astereognosis (inability to recognize objects by feeling them) and Agnosia.
Mamillary bodies: loss of recent memory.
Cholinergic neurons in the nucleus basalis of Meynert and related areas of the forebrain: loss of recent memory.
Basal Ganglia
The basal ganglia consist of the , globus pallidus, subthalamic nuclei, and substantia nigra.
They modulate thalamic outflow to the motor cortex to plan and execute smooth movements.
The primary function of the basal ganglia is the planning and programming of movement.
Main NT is GABA.
Dopamine is the neurotransmitter between the striatum and substantia nigra.
Subthalamic Nucleus: releases Glutamate → excites globus pallidus, internal segment
Substantia Nigra Parts Reticulata → releases GABA → inhibits thalamus
Substantia Nigra Pars Compacta → releases Dopamine → inhibit the striatum
Striatum → releases ACh → inhibit substantia nigra pars reticulata
Globus Pallidus External Segment → releases GABA → inhibit subthalamic nucleus
Lesions in different parts of the basal ganglia can lead to specific movement disorders:
Globus Pallidus: inability to maintain posture.
Subthalamic Nucleus: wild, flailing movements (hemiballismus).
Striatum: quick, continuous, uncontrollable movements (e.g., in Huntington disease).
Substantia nigra: tremors, cogwheel rigidity, reduced voluntary movement (akinesia), and postural problems (Parkinson's disease).
Motor Cortex
It consists of two main parts:
Premotor Cortex and Supplementary Motor Cortex (BA 6): These areas generate the movement plan, which is then transferred to the primary motor cortex for execution.
Supplementary motor cortex: rehearses complex motor sequences
Primary Motor Cortex (BA 4): This area executes the movement plan, sending signals to the brainstem and spinal cord, where lower motor neurons initiate voluntary movements.
Epileptic events in the primary motor cortex can cause Jacksonian seizures (focal partial seizures).
Clinical Correlations
Brown-Sequard Syndrome
Caused by functional hemisection of the spinal cord
(+) contralateral loss of pain and temperature sensation beginning 1-2 segments below the lesion
(+) ipsilateral weakness and spasticity in certain muscles groups
Amyotrophic Lateral Sclerosis (ALS) or Lou Gehrig Disease
(+) Degeneration & loss of motor neurons in the motor cortex, spinal cord, brain stem & corticospinal tract
Does NOT usually affect sensation
May present with UMN or LMN SSx depending on location
Patients with Transected Spinal Cords
Have negative nitrogen balance as they catabolize large amounts of body protein because they are paralyzed below the level of transection
Trinucleotide Repeat Diseases
Include Fragile X, Spinocerebellar ataxia type 3, Huntington Disease, Friedreich Ataxia

- if umabot ka dito, goodluck sa exam!, Drie :)