11-Introduction to Organic Chemistry
- Organic chemistry is the study of the millions of covalent compounds of the element carbon
- These structurally diverse compounds vary from naturally occurring petroleum fuels to DNA and the molecules in living systems.
- Organic compounds also demonstrate human ingenuity in the vast range of synthetic materials created by chemists. - Many of these compounds are used as drugs, medicines and plastics.
\ ==Why can carbon form rings and very long chains?==
- A carbon atom has 4 electrons in its outer shell, so it forms 4 covalent bonds
- ==Carbon-carbon bonds== are relatively ==strong and non-polar==
![]()
![]()
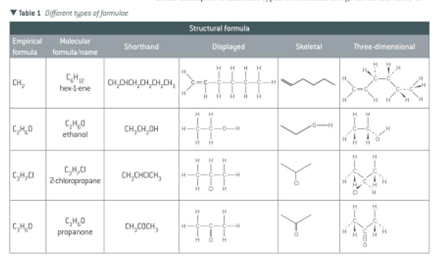
\ Displayed formula
- shows every atom and every bond in the molecule
- shows the arrangement of atoms showing all the bonds and atoms in the molecule

Structural formula
- shows the unique arrangement of atoms in a molecule in a simplified form, without showing all the bonds
![]()
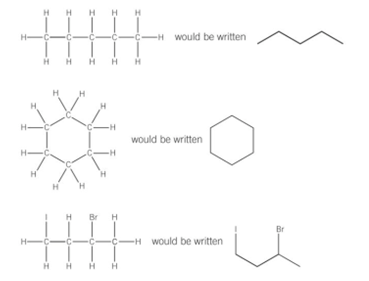
Skeletal formula
- carbon atoms are not drawn at all
- straight lines represent carbon to carbon bonds
- carbon atoms are assumed to be where the bonds meet
- shows that bonds of the carbon skeleton only.
- Hydrogen and carbon atoms are not shown, only functional groups are
Free radicals
- when a covalent bond breaks in a way that one electron goes to each atom that originally formed the bond
- the fragments of the original molecule have an unpaired electron - free radicals
- extremely reactive
\
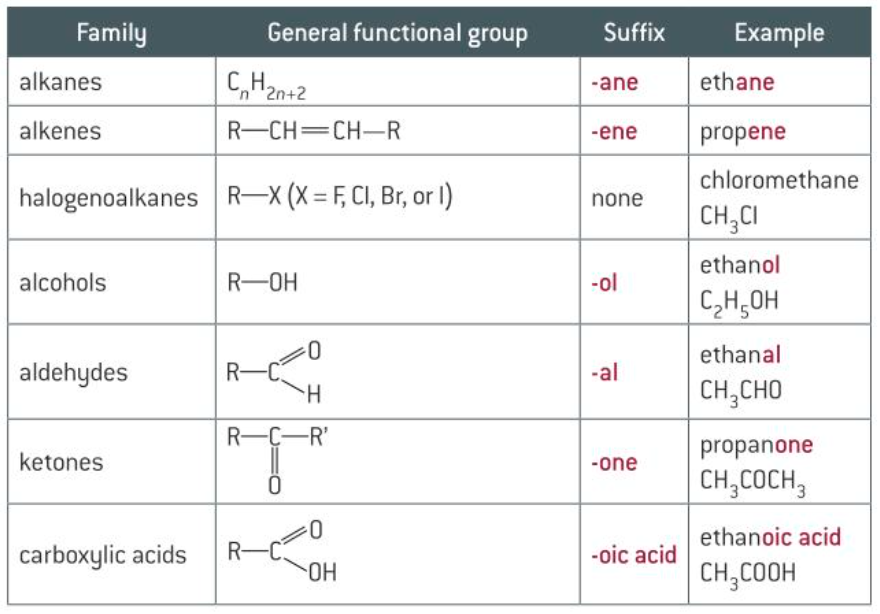
@@Nomenclature - naming organic compounds@@
- The system used for naming compounds was developed by IUPAC
- Systematic names tell us… about the structure of the compounds rather than just the formula
\ Roots
- tells us whether there are any double bonds
- -@@ane@@ means no double bond
- -@@ene@@ means double bond
\ Functional groups
- reactive groups attached to a hydrocarbon chain
- halogenalkenes are named using a prefix rather than a suffix (bromo-, chloro-, fluoro-)
@@Chain and position isomers @@
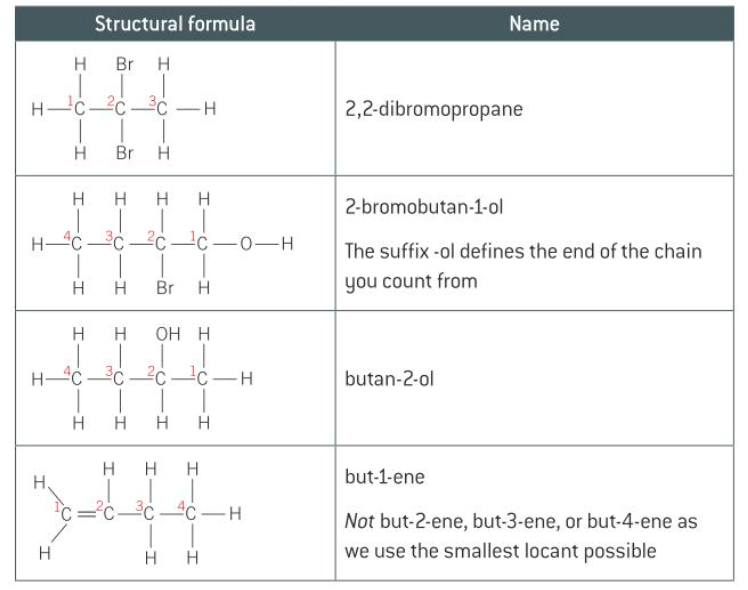
- a number is used to tell us the position of any branching in a chain and the position of any functional group
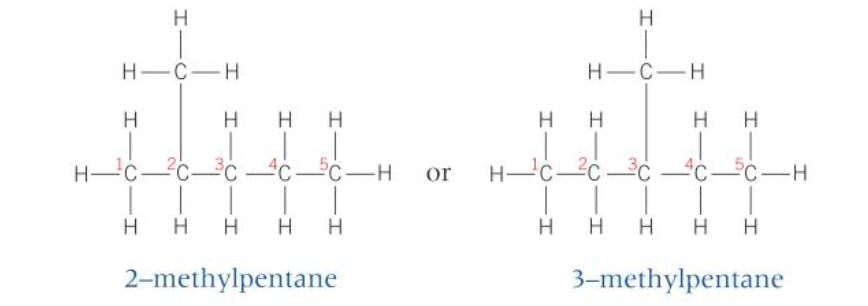
- structural isomers have the same molecular formula but different structural formulae
![]()
![]()
\ @@Homologous series@@
- }}Homologous series is a family of organic compounds with the same functional group but different carbon chain length}}
- members of a homologous series have the same general formula
- successive members of the same homologous series increases by CH2

- the length of the carbon chain of the functional group
- the length of the carbon chain such as boiling and melting point * melting and boiling point increase by a small amount when the number of carbon in the chain increases
- ==reduces== because the
Isomers
- {{Isomers are molecules that have the same molecular formula but whose atoms are arranged differently{{
- There are 2 basic types of isomerism: * ==Structural isomerism== * ==Stereoisomerism==
\ Structural isomerism
- Structural isomerism - have the ==same molecular formula== but ==different structural formula==
- 3 Different Structural isomers: * * *
\ Positional isomerism
- the same functional groups attached to the main chain at different points
- same molecular formula but different position of the functional group
![]()
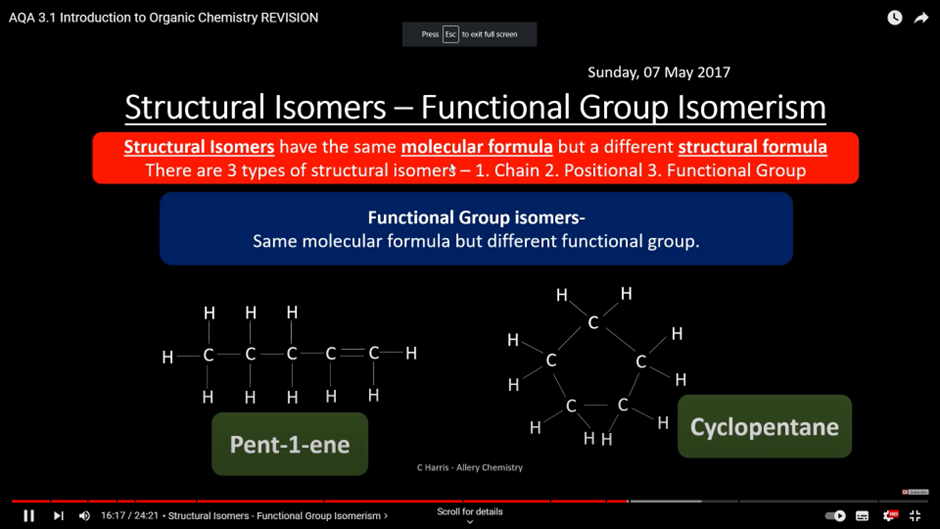
Functional isomerism
- functional groups that are different
- same molecular formula but different functional group
![]()
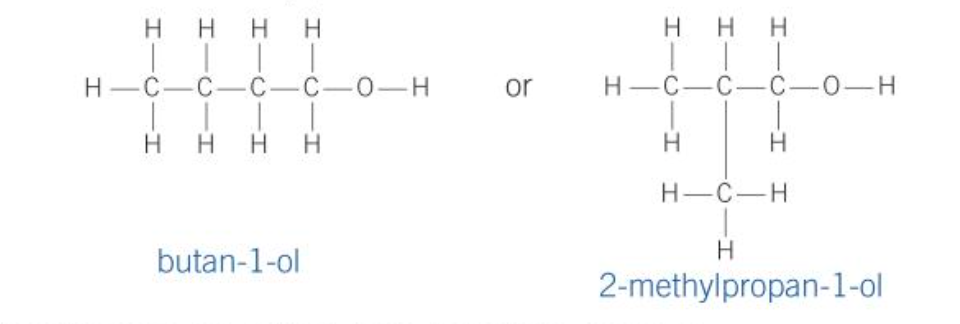
Chain isomerism
- a different arrangement of the hydrocarbon chain, e.g., branching

==Stereoisomerism==
- Stereoisomerism - where two or more compounds have the same structural formula. They differ in the arrangement of the atoms in space
- There are @@2 types of stereoisomerism:@@ * ==E-Z isomerism== * ==Optical isomerism==
\ E-Z isomerism
- tells us about the position of substituents at either side of a carbon-carbon double bond
- two substituents can either be in the @@same side of the bond (Z isomer)@@ or on %%opposite sides (E isomer)%%
- substituted groups joined by a single bond can rotate around the single bond
- there is no rotation around a double bond
- have @@different chemical and physical properties@@
- e.g. different reaction rates for the same reaction (chemical property) or different melting/boiling points (physical property)
- \


How can I tell if the compound is stereo or structural?
- Is there is @@no restricted bond rotation around C-C bond, (no double bond)@@ so it is @@structural isomerism@@
- structural isomerism = no restricted bond rotation = no double bond
- stereo isomerism = a restricted bond rotation = double bond
\ How to deduce isomers?
- Step 1: Draw the structural formula of the compound
![]()
- Step 2: Determine whether it is a stereo or structural isomer * There is no restricted bond rotation around the C-C bond, so it is structural isomerism
- Step 3: Determine whether it is a functional group, chain or positional isomerism * ==Functional group?== No, as there are no functional groups * ==Positional?== No, as there are no functional groups which can be positioned on different carbon atoms * ==Chain?== Yes!
\n 
How to deduce isomers? Example 2
- Step 1: Draw the possible structural formula of the compound
![]()
- Step 2: Determine whether it is a stereo or structural isomer
The compound has to be ==unsaturated== for it to have molecular formula C2H2Cl2 ; Due to the ==double bond== there is ==restricted rotation about the C-C bond;== This compound will therefore display ==geometrical isomerism==
\
![]()