HW #24 - Kognity
Bond Breaking: energy is absorbed, endothermic, + delta H
Bond Forming: energy is released, exothermic, - delta H
exothermic reactions have more stable products
endothermic reactions have less stable products compared to the reactants
chemical species with higher energy are less stable than those with lower energy
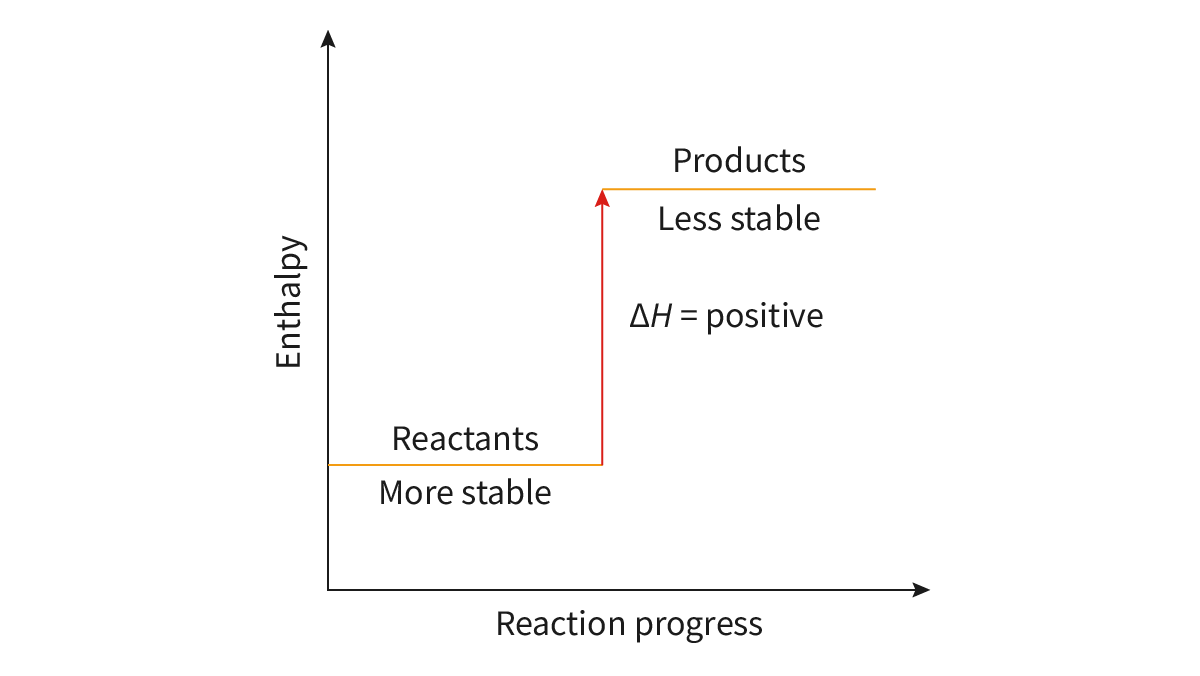
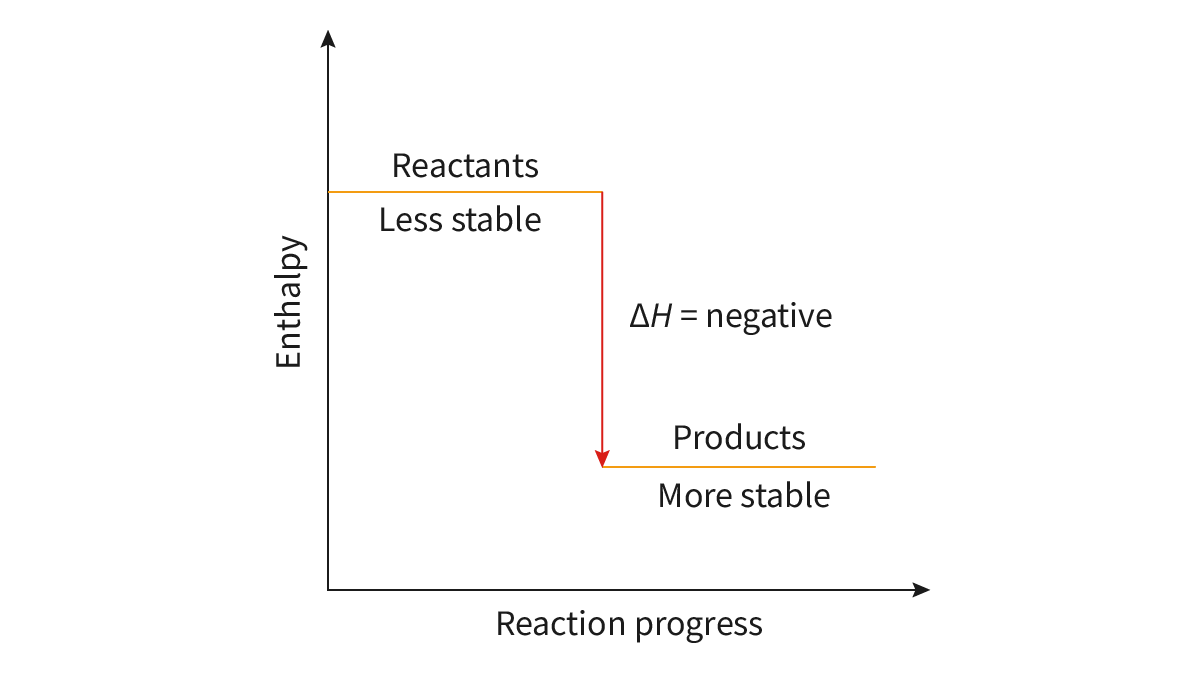
Bond Enthalpy (H): the energy required to break one mole of chemical bonds in the gaseous state
Bond Dissociation Energy (E): both of these terms are used interchangeably & used to quantify the energy required to break a bond
Bond enthalpy values are always positive because they refer to bonds being broken (bond breaking is endothermic)
C-H bonds differ in different molecules ——> bond enthalpies are called average bond enthalpy: The enthalpy change when one mole of bonds are broken in the gaseous state averaged for the same bond in similar compounds
Calculate the Enthalpy of a reaction by ——> difference in bond energy between the bonds broken and the bonds formed:
ΔH = Σ(bond enthalpy (BE) of bonds broken) – Σ(BE of bonds formed)
OR
ΔH = Σ(reactant bond enthalpies) – Σ(product bond enthalpies)
the net enthalpy of a chemical reaction is independent of the path taken
enthalpy is a state function: thermodynamic value is independent of the path taken to reach that specific value
Hess’s Law (published by Germain Henri Hess in 1840): states that the total enthalpy change in a chemical reaction is independent of the route by which the chemical reaction takes place, as long as the initial and final conditions are the same
an application of the law of conservation of energy since both state that energy is conserved
Energy cycles: useful tool when applying Hess’s law in determining the enthalpy of a reaction ——> show separate pathways from reactants to products
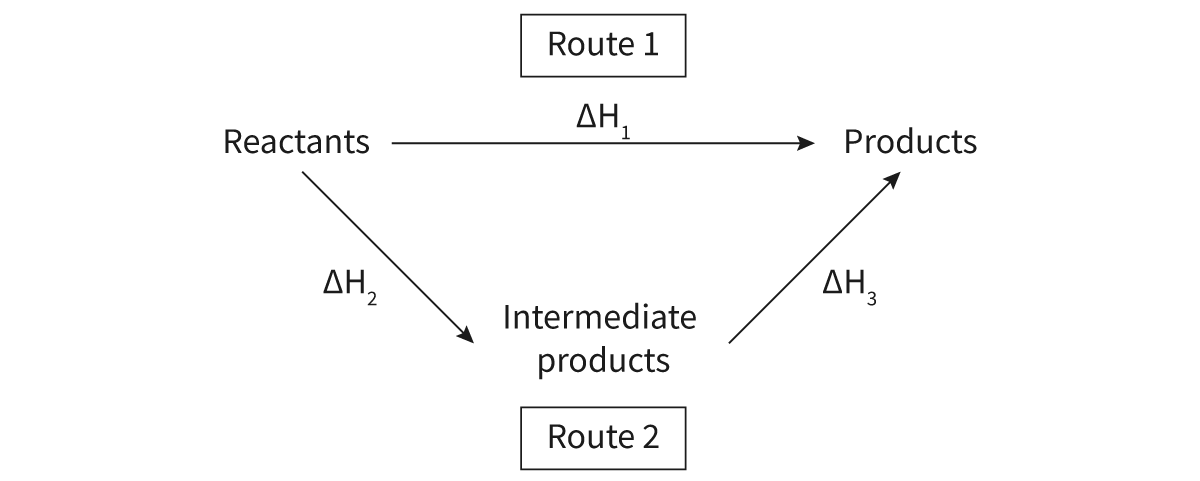
Calculating the enthalpy of reaction using Hess’s law involves manipulating the different pathway equations and cancelling out those atoms or molecules that occur on both sides of the reaction arrow