chemistry notes im crying as we speak
Chapter 1 - Intro to Chem
1.1 Intro to Chemistry
chemistry - study of composition matter and the changes they undergo.
Two Worlds of Chemistry
a chemist is someone who studies chemistry
they use two worlds of chemistry: macroscopic and microscopic
macroscopic - substances that can be physically seen, touched, measured
microscopic - referring to small particles that make matter
*chemists must observe matter and do experiments macroscopically world and then use their observations to make generalizations and propose microscopic explanations
Fields of Chemistry
types of chemistry
pure chemistry - emphasis on theory and how matter changes in a chemical process
applied chemistry - directed to a specific goal or application irl
there are 5 main disciplines of chemistry:
biochemistry - pure chemistry that study chemical processes of living things
organic chemistry - study of any chemicals that contain carbon (deadass almost everything bc carbon is abundant and can make many chemicals) ; both pure and applied chem
inorganic chem - study of any chemical/substances with no carbon ; both pure and applied chem
physical chem - study of macroscopic and atomic properties and phenomena in chemical systems (ex: rates of reactions, energy transfers, structure of at a molecular level)
analytical chemistry - more on the composition of matter with a focus on separating, identifying, and quantifying chemical samples
1.2 Scientific Method
scientific method - a logical, systematic, approach to observing and investigating natural phenomena.
scientific law - explanation of summary of results of many many experiments with NO EXCEPTIONS
SCIENTIFIC METHOD STEPS
Observation and Question - questions can be specific or general
Making A Hypothesis - a hypothesis or a testable conjecture that proposes an answer (if/then format)
Research - research topic of interest
Design Experiment - gather materials and write procedure;
need experimental variables such as:
independent variables - variables that are being changed
dependent variables - variables being observed/measured
control group - experiences the same conditions as the experimental group without being tested on by a different factor/variableExperiment - test the hypothesis; you can make revisions along the way
Record Data and Interpret when finished
Conclusions/Revision - revise the hypothesis if the results are inconsistent with the hypothesis. further experiments for revision can be made to theories too. conclusions can use deductive or inductive reasoning (mostly the latter, its very fundamental to the scientific method)
deductive reasoning - a specific conclusion based on facts, and theories. conclusion is only correct if everything else is correct
inductive reasoning - a general conclusion based on observations of patterns, relationshipsDeveloping Theories - theory is an explanation repeatedly tested and confirmed by multiple researchers and gained wide acceptance. this could only happen if similar results were from more and more experiments. theory details can be adjusted as more discoveries are made.
Chapter 2 - Matter
2.1 Properties of Matter
matter - anything with mass that takes up space
Properties of Matter
chemical properties - describes a chemical’s ability to undergo chemical changes. can be seen by changing its chemical composition
Examples: reactivity, flammability, ability to oxidize/rust, toxicity
physical properties - properties of a material/chemical that can be observed without changing it's chemical composition
Intensive Properties - properties that depend on the type of matter
Examples: color, odor, melting point, boiling point, hardness, texture, density
Extensive Properties - properties that depend on the amount of matter
Examples: volume, mass
States of Matter
solid - matter with definite shape and definite volume. it cant be compressed and can expand slightly when heated. molecules are bonded, packed and structured and have the lowest molecular kinetic energy.
liquid- matter with only definite volume. it cant be compressed and expands and can expand slightly when heated. molecules in liquids have a bendy structure. liquids are the result of solids whose temperatures are more than their melting point
gas - matter with no definite shape and volume. it can be compressed and expands greatly when heated. particles are very far apart from each other and have a high molecular kinetic energy. It is considered a vapor if its material is a solid or liquid at room temp (ex: water).
plasma - ionized gas made with positively and negatively charged ions. has the highest molecular kinetic energy of all states of matter because it only exists in very high temperatures
Matter Transitions:
solid —> liquid = melting solid —> gas = sublimation
liquid —> solid = freezing liquid —> gas = evaporation
gas —> liquid = condensation gas —> solid = deposition
Q&A
Explain why water is considered a pure substance, while milk is not.
A: Pure substances are made with a uniform composition, meaning water is only made of one type of molecule while milk contains proteins, lipids, and water.
List at least three physical properties of silver.
A: silver is a grey metallic in color, with a density of 10.49 g/cm³, and a boiling point of 1235 K
Give an example of an extensive property and an intensive property of an iron nail. Explain.
A: an extensive property of an iron nail is mass. this property depends on the amount of iron in the nail, so if you cut the nail in half, the mass would be reduced.
an intensive property is density. density doesn’t change no matter how much of the nail you have. whether you have a full nail or just a part of it, the density of iron remains the same.
Compare the arrangements of particles in the three states of matter.
A: in solids, particles are tightly packed and arranged in a fixed, orderly pattern. they vibrate but stay in place, giving solids a definite shape and volume.
in liquids, particles are close together but not in a fixed arrangement. they can move and slide past each other, allowing liquids to take the shape of their container but maintain a definite volume.
in gases, particles are far apart and move freely in all directions. They have no fixed shape or volume, so they expand to fill the entire space of their container.
The odor of gasoline is very strong and distinctive. Can gasoline in the gaseous state be referred to as a vapor? Explain.
A: well yes because it is a liquid at room temperature. vapor is a gaseous state of a material that is a solid or liquid at room temperature.
Describe how the sharpening of a pencil is a different type of physical change than dissolving some sugar in water.
A: sharpening a pencil changes its shape, but the materials (wood, graphite) stay the same.
dissolving sugar in water spreads the sugar throughout, but the sugar itself doesn’t change and can be recovered later. both are physical changes, just different ways of rearranging matter.
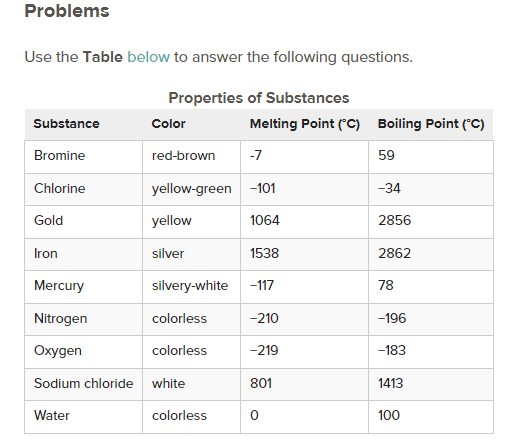
What is the physical state of each of the following substances at room temperature?
a. oxygen - gas
b. iron - solid
c. mercury - liquid
What is the name of the physical change that occurs in each of the following instances?
Oxygen is cooled from −150°C to −200°C. - condensation
Iron is cooled from 1600°C to 1500°C - no phase change temperature decrease
Mercury is heated from −130°C to -100°C. - melting
Which colorless substance is a solid at −50°C?
A: water
Which substance is a liquid at 1000°C?
A: sodium chloride
Which substance condenses from a gas to a liquid as the temperature is cooled from 20°C to −50°C?
A: chlorine
`
2.2 Classifying Matter
matter can be classified into two groups
Pure Substances
pure substances are matter with a uniform and definite composition
elements - the simplest form of matter with a unique set of properties
compounds - are made by more than one element and when broken down, its pieces can have different properties than the original compound.
- example: water is made by two gaseous elements but combined makes a liquid
chemical formula is an expression that shows the elements in a compound and the relative proportions of those elements: (example: H20)
Mixtures
physical blend of two or more components each of which retains its own identity
homogenous mixtures - mixtures with a uniform and definite composition throughout
heterogeneous mixtures - mixtures with no uniform or definite composition throughout and their parts retain their own identities
examples:
- colloids
- suspensions
- emulsions
Separation of Mixtures
Chromatography - is used to separate ink or dyes
Filtration - is used to separate mixtures by size
Distillation - separates mixtures by boiling point
Crystallization - solid crystals form from a dissolved
solution
Q&A
Why can’t a mixture be classified as a pure substance?
A: mixtures are made of one or more components while substances are made of one universal composition
Classify each of the following as a homogeneous or heterogeneous mixture.
salad - heterogenous
iced tea - homogeneous
soil - heterogeneous
cooking oil - homogeneous
When is filtration used to separate a mixture? When is distillation used?
A: Filtration is used to separate solids from mixtures and distillation is used to separate mixtures based on boiling points
What is wrong with the following explanation of an experiment? “When it was heated, the sample of carbon decomposed.
Identify the following as either a chemical symbol or a chemical formula.
CO - formula
Co - symbol
W - symbol
Be - symbol
HI - formula
Describe a way to separate a mixture of fine sand and table salt without having to directly sort tiny crystals using tweezers.
A: u could filter out the sand using a special filter because the sand grains are smaller than the salt crystals
Name the elements found in each of the following compounds.
ammonia (NH3) - nitrogen, hydrogen
saltpeter (KNO3) - potassium, nitrogen, oxygen
glucose (C6H12O6) - carbon, hydrogen, oxygen
millerite (NiS) - nickel, sulfur
Identify each of the following items as an element, compound, or mixture. If it is a mixture, further classify it as homogeneous or heterogeneous.
gasoline - mixture
calcium - element
carbon dioxide - compound
blood - mixture
2.3 Changes in Matter
Physical Changes
a measurable or visible change without changes to the composition of matter
is usually reversible
Examples: cut, boil, melt, split, crack, bend
Chemical Changes
happens one or more substances turned into a product with one or more different compositions; also known as a chemical reaction
is usually irreversible
Examples: burnt, fermented, rust, decomposed
there’s always a transfer of energy in a chemical reaction.
a chemical equation can be used to describe a chemical reaction:
formula is generally: reactant + reactant —> product
some visual clues that indicate that a chemical reaction (likely but not necessarily occurring)
A change of color occurs during the reaction.
A gas is produced during the reaction.
A solid product, called a precipitate, is produced in the reaction.
A visible transfer of energy occurs in the form of light as a result of the reaction
Q&A
What is the main difference between a physical change and a chemical change?
A: A physical change are observable and measurable and reversible but it doesn’t change the chemical components of the material. A chemical change are usually permanent and it changes a material’s chemical structure.
How must the chemical properties of a given substance be observed?
A: The chemical properties of a given substance describes it’s ability to go under go a chemical change which we try to chemically change the substance in order to see
Give an example of a situation where a color change occurs, but not because of a chemical reaction.
A: a chameleon’s skin changing color, it expands the different pigments in order to change but there’s rlly no chemical process involved
Classify the following as physical properties or chemical properties of the element nickel.
It is gray in color. - physical
It melts at 1455°C. - physical
It reacts with sulfuric acid. - chemical
It is a conductor of electricity. - chemical
It forms a compound with oxygen. - chemical
Combustion, also called burning, is a reaction with oxygen gas. When propane is combusted, carbon dioxide and water are formed. Identify the reactants and the products of this combustion reaction.
A: reactants are oxygen gas and propane, while products are carbon dioxide and water
Classify each of the following as a chemical change or a physical change.
Sugar dissolves in water. - physical change
A peach rots. - chemical change
Icicles melt in the warm sunlight. - physical change
A baking cake rises in the oven. - chemical change
A leaf changes its color in the fall. - chemical change
Food coloring is added to a glass of water. - chemical change
Describe clues that you might observe during the following situations that would indicate that a chemical reaction is likely occurring.
A log burns in the fireplace. - a log is combusting, I cant rlly unburn a log can I?
A mixture of baking soda and vinegar fizzes. - there’s a noticeable reaction
Chapter 3 - Measurements
3.1 International Systems of Units
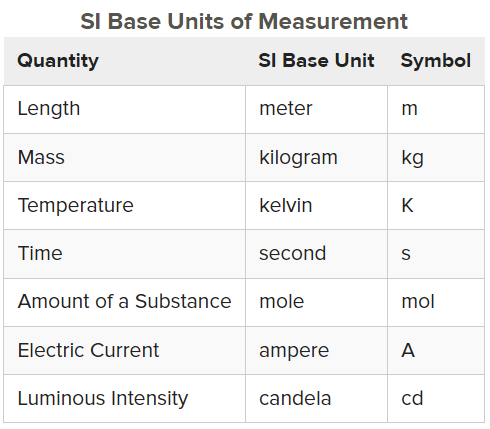
International System of Units
a system of measurement based on the metric system (powers of 10)
energy - the ability to do work or an action
unit: joule (J)
1 joule is equal to a kilogram times a meter squared divided by a second squared (kg•m2/s2). A common non-SI unit of energy that is often used is the calorie (cal), which is equal to 4.184 J
temperature - average amount of kinetic energy of particles in matter
weight - a measure of force that is equal to the gravitational pull on an object
liter -
symbol: L
equals to the volume of something that is 1000cm3
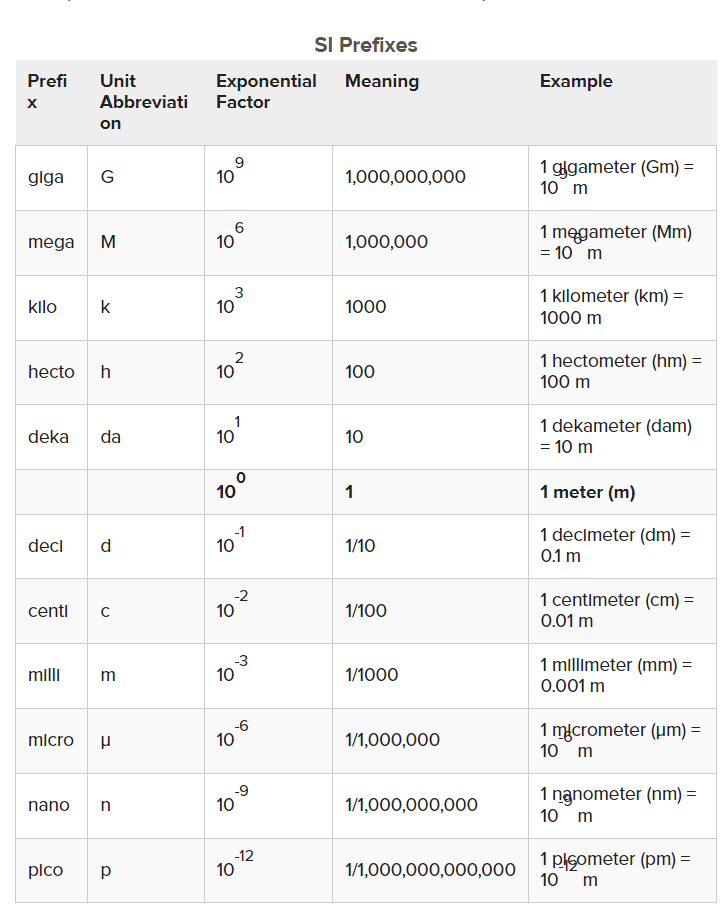
Scientific Prefixes
3.2 Conversion Factors
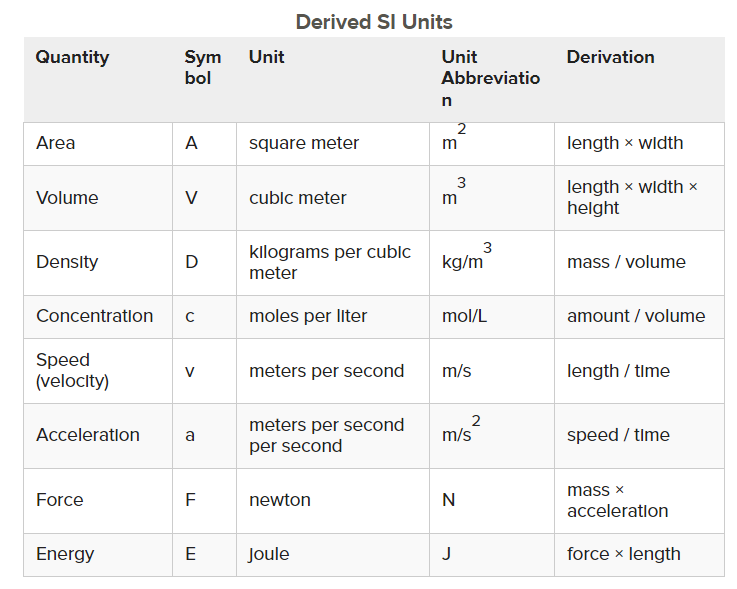
Derived Units
a unit as a result of a mathematical combo of SI base units
Conversion Factors and Dimensional Analysis
conversion factors - ratios of equivalent measurements
dimensional analysis - a technique that uses the units (dimensions) of the measurement to correctly solve problems
dimensional analysis is helpful in converting one unit in the metric system to another
3.3 Uncertainty and Sig. Figs
Significant Figure Rules
significant figures - a measurement consists of all the certain digits in that measurement plus one uncertain or estimated digit
on-zero digits - all non-zero digits are significant.
zeros between non-zero digits - zeros between two non-zero digits are significant.
trailing zeros - trailing zeros in the decimal portion of a number are significant.
leading zeros - leading zeros are not significant. they are placeholders and can be omitted.
example: the number 0.003 has one significant figure, and could be written as 3 x 10-3
When adding/subtracting:
the answer should have the same number of decimal places as the number with the fewest decimal places
When multiplying/dividing:
the answer should have the same number of significant figures as the number with the fewest significant figures
Rounding Sig Figs
1) if the first digit you remove is 4 or less, drop it and all the following digits
example: 2.4271 becomes 2.4 when rounded off to two significant figures because the first dropped digit (a 2) is 4 or less.
2) if the first digit removed is 5 or greater, round up by adding 1 to the last digit kept
Accuracy vs Precision
accuracy
a measure of how close a measurement is to the correct or accepted value of the quantity being measured.
precision
a measure of how close a series of measurements are to one another.
precise measurements are highly reproducible, even if the measurements are not near the correct value.
Percent Error
an individual measurement may be accurate or inaccurate, depending on how close it is to the true value
accuracy - the true or correct value based on general agreement with a reliable reference
experimental value - value that was collected/recorded from the experiment
error
difference between experimental and accepted value
formula: (error = experimental value - accepted value)
percent error
absolute value of the error turned into a percent
formula: (percent error = [experimental value - accepted value] / accepted value x 100% )