Organic 2
Key Words
Homologous series
Family of compounds with similar chemical reactivity
Show trends in physical properties
Each member differs by CH₂
Functional group
Part of a molecule (not C and H only)
Determines chemical reactivity
Anaerobic
Occurs without oxygen
Catalyst
Speeds up a reaction without being used up
Provides an alternative pathway with lower activation energy
Monomer
Small molecule that joins to form a polymer
Polymer
Long-chain molecule made from many monomers joined together
Alcohols
General formula of alcohols: CₙH₂ₙ₊₁OH
A functional group is a group of atoms (other than C or H) that gives a compound its characteristic reactions
The functional group present in all alcohols is the hydroxyl group (–OH)
Alcohol Homologous Series
Members differ by CH₂
Similar chemical reactions
Gradual trends in physical properties (e.g. boiling point)
Examples of Alcohols
Alcohol | No. of C atoms | Molecular formula | Structural formula | Displayed formula |
|---|---|---|---|---|
methanol | 1 | CH₃OH | CH₃–OH | 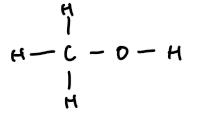 |
ethanol | 2 | C₂H₅OH | CH₃–CH₂–OH | 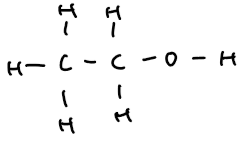 |
propan-1-ol | 3 | C₃H₇OH | CH₃–CH₂–CH₂–OH | 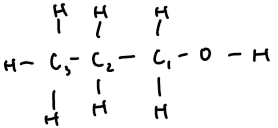 |
propan-2-ol | 3 | C₃H₇OH | CH₃–CH(OH)–CH₃ | 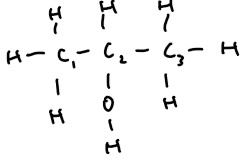 |
butan-1-ol | 4 | C₄H₉OH | CH₃–CH₂–CH₂–CH₂–OH | 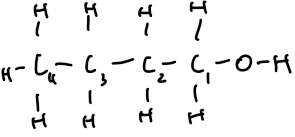 |
butan-2-ol | 4 | C₄H₉OH | CH₃–CH(OH)–CH₂–CH₃ | 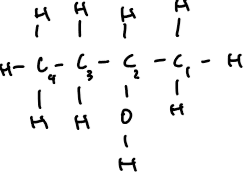 |
Alcohols with the same molecular formula but different structures are structural isomers
Ethanol
Preparation Of Ethanol (Two Methods)
1. Fermentation Of Sugars
Carried out by yeast in the absence of oxygen (anaerobic)
An exothermic reaction that provides energy for the yeast
Yeast occurs naturally on fruit skins, e.g. grapes
Ethanol concentration is limited to about 15% because yeast is killed by ethanol
Fractional distillation is used to produce more concentrated ethanol
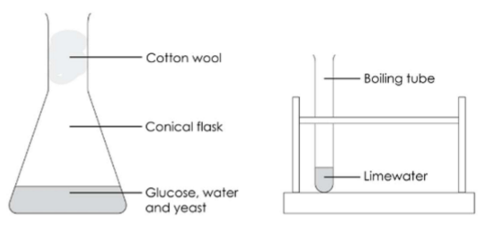
Equation
Glucose → ethanol + carbon dioxide
C₆H₁₂O₆ → 2C₂H₅OH + 2CO₂
Conditions
Warm temperature (about 30–40 °C)
No oxygen present
Yeast as the catalyst
Usually a batch process, though continuous-flow is possible using immobilized yeast
Exothermic reaction, the reaction is a source of energy for yeast in the absence of oxygen
Yeast is found naturally in the skin of many ripening fruits like grapes
The yeast dies when the concentration of ethanol reaches approx. 15%
The mixture is fractionally distilled to obtain higher concentrations of ethanol
Addition Of Steam To Ethene – Main Industrial Process For Ethanol
Equation
Ethene + steam → ethanol
C₂H₄(g) + H₂O(g) → C₂H₅OH(l)
Conditions
High temperature (about 300 °C)
High pressure (about 60–70 atm)
Phosphoric acid catalyst on a solid support
Continuous process with recycling of unreacted ethene
Only a small proportion of ethene reacts each pass
Ethanol is cooled, condensed, and collected
Unreacted ethene is recycled, improving efficiency
Comparison Of The Two Methods
Fermentation
Uses renewable resources such as sugar cane or sugar beet
Batch process, so less efficient (stop–start)
Slow rate, taking several days
Produces impure ethanol that needs fractional distillation
Low temperature and atmospheric pressure
Lower energy costs and safer operating conditions
Hydration Of Ethene
Uses finite resources from crude oil
Continuous flow process, more efficient
Fast reaction rate
Produces ethanol as the only product
Requires high temperature and high pressure
Higher energy costs and greater safety considerations
Uses Of Ethanol
Alcoholic beverages such as wine, beer, and spirits
Solvent in cosmetics, toiletries, inks, and the chemical industry
Alternative fuel; E10 fuel contains 10% ethanol
Reactions Of Ethanol
Combustion (Oxidation)
Ethanol burns in oxygen with a clean, almost invisible flame
Equation:
C₂H₅OH(l) + 3O₂(g) → 2CO₂(g) + 3H₂O(g)
Oxidation In Air To Form Ethanoic Acid
Occurs when wine turns into vinegar
Ethanol is slowly oxidised by oxygen in the air
Equation:
C₂H₅OH(aq) + O₂(g) → CH₃COOH(aq) + H₂O(l)
Oxidation Using Potassium Dichromate(VI)
Uses potassium dichromate(VI), K₂Cr₂O₇, and sulfuric acid
Used in industrial chemistry
Orange Cr₂O₇²⁻ ions are reduced to green Cr³⁺ ions
This is a redox reaction
[O] represents oxygen from the oxidising agent
Equation:
C₂H₅OH + 2[O] → CH₃COOH + H₂O
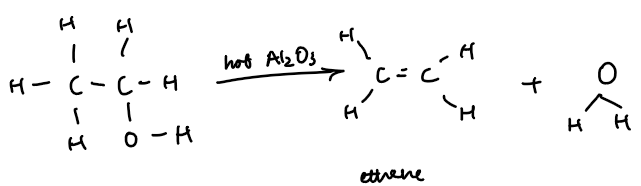
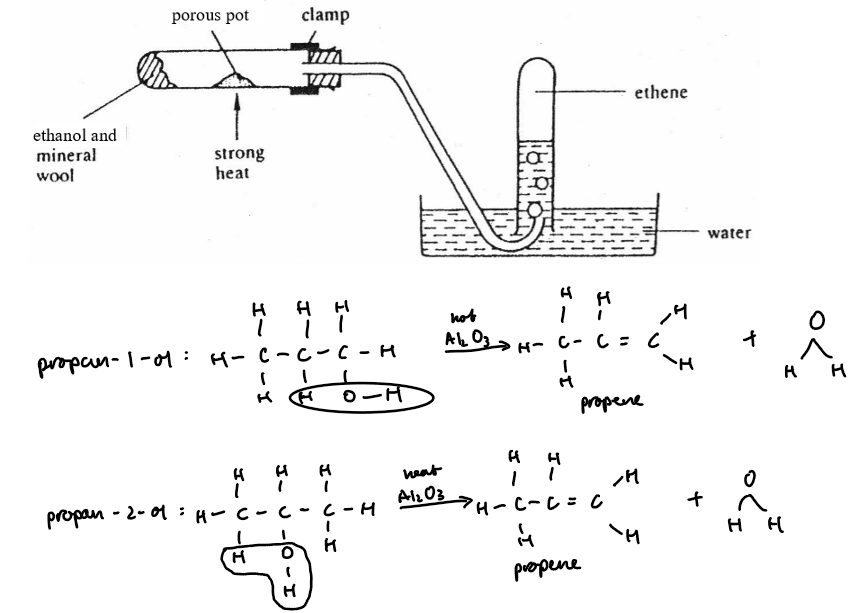
Dehydration (Removal Of Water)
Ethanol is dehydrated to form ethene, an alkene
Ethanol vapour is passed over hot aluminium oxide catalyst
Ethene gas is collected, often over water
Equation:
C₂H₅OH(l) → C₂H₄(g) + H₂O(l)
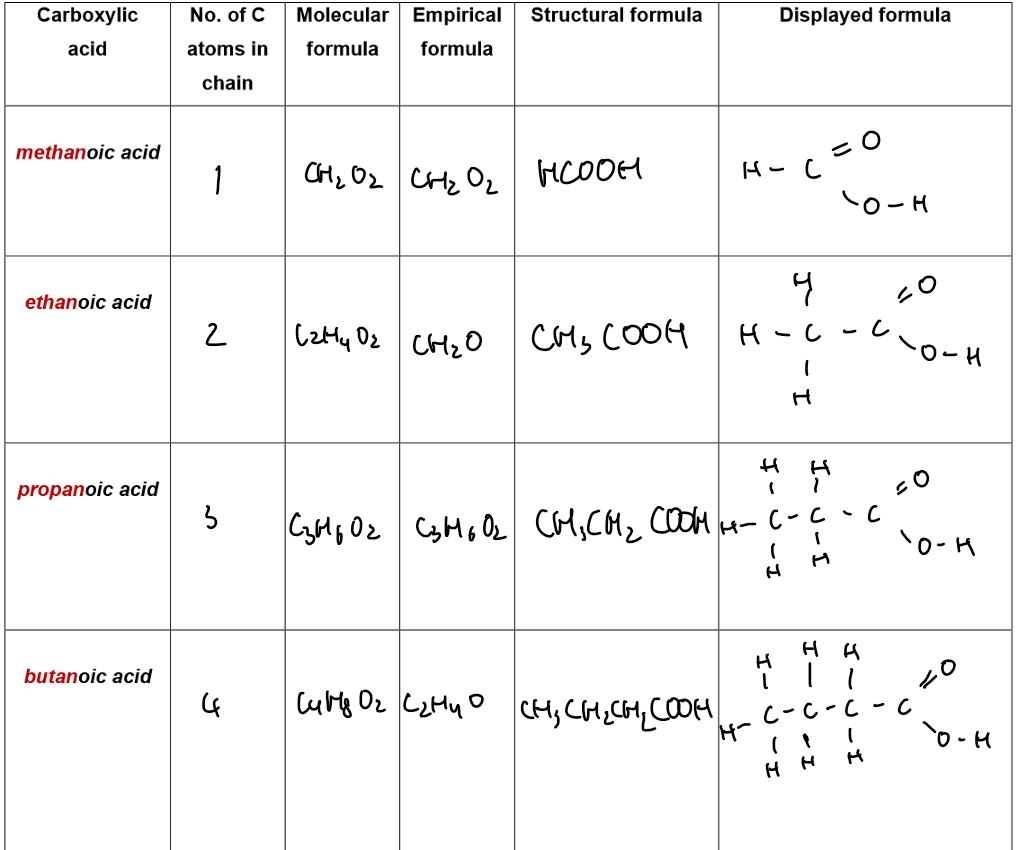
Carboxylic Acids
General formula (linear carboxylic acids): CₙH₂ₙO₂
Functional group: –COOH
Example: Vinegar is an aqueous solution of ethanoic acid.
Reactions of Carboxylic Acids
Carboxylic acids are weak acids as they are only partially ionised to form H⁺ ions in water.
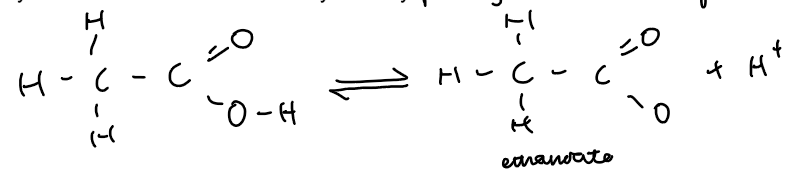
They undergo the same reactions as mineral acids (e.g., HCl) but react more slowly.
(a) Reaction with reactive metals (e.g., magnesium)
Carboxylic acid + metal → salt + hydrogen gas
Example: 2CH₃COOH + Mg → (CH₃COO)₂Mg + H₂
(b) Reaction with a base (e.g., sodium hydroxide)
Carboxylic acid + base → salt + water
Example: CH₃COOH + NaOH → CH₃COONa + H₂O
(c) Reaction with a carbonate (e.g., calcium carbonate)
Carboxylic acid + carbonate → salt + water + carbon dioxide
Example: 2CH₃COOH + CaCO₃ → (CH₃COO)₂Ca + H₂O + CO₂
Reactions with Alcohols to Form Esters
Heating a carboxylic acid with an alcohol in the presence of concentrated sulphuric acid produces an ester and water:
All of these reactions are reversible
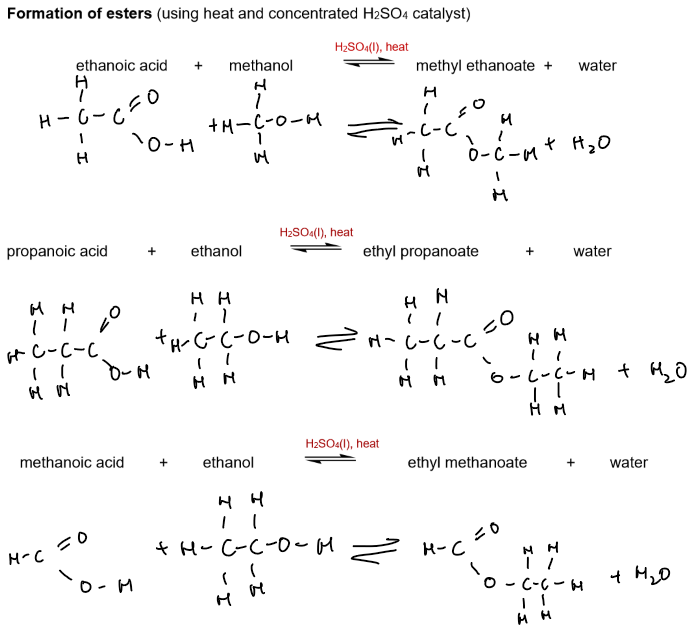
carboxylic acid + alcohol → ester + water
Ethanoic acid + ethanol → ethyl ethanoate + water
CH₃COOH + CH₃CH₂OH → CH₃COOCH₂CH₃ + H₂O
Displayed formula example:
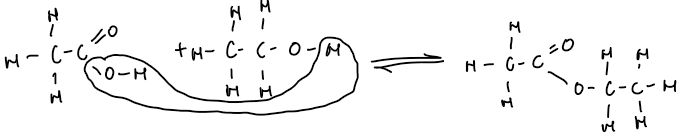
Reaction is called esterification.
Also a condensation reaction because water is formed when two molecules join.
Esters
Functional group: RCOOR′ (R/R′ = alkyl group, e.g., –CH₃)
Naming:
First part from the alcohol: methanol → methyl
Second part from the carboxylic acid (carboxylate anion): ethanoic acid → ethanoate
Example: CH₃COOCH₃ = methyl ethanoate
Formation of esters (heat + concentrated H₂SO₄ catalyst)
Uses of esters
Volatile compounds with distinctive smells
Used as:
Solvents
Food flavourings
Perfumes
Condensation Polymerisation
A condensation polymer forms when two types of monomer react, with the loss of a small molecule (e.g., water or HCl).
Addition followed by elimination is called condensation.
Monomers have different functional groups.
Polyesters are used in textiles and clothing.
Some polyesters are biodegradable (can be broken down in the environment).
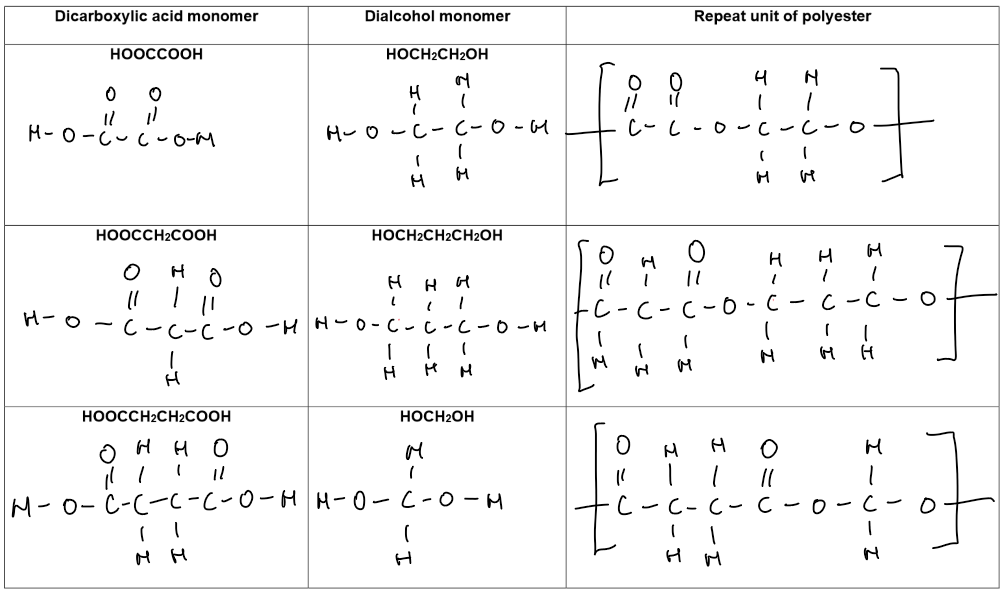
Polyesters (contain –COO– link)
Monomers:
Type | Functional Group | Example |
|---|---|---|
Dicarboxylic acid | –COOH at both ends | Ethanedioic acid |
Dialcohol (diol) | –OH at both ends | Ethane-1,2-diol |
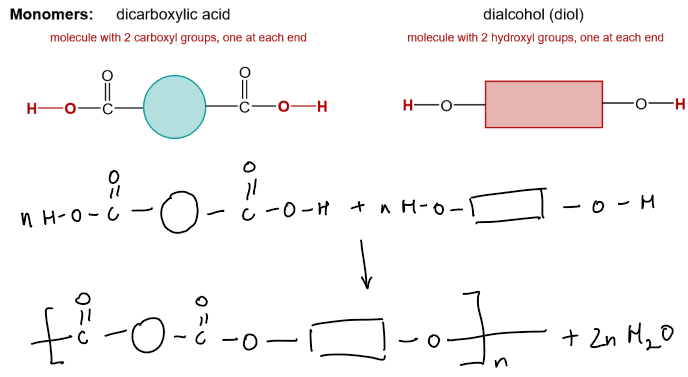
The –COOH group of the acid reacts with the –OH group of the alcohol to form an ester link –COO–.
Repeating this reaction forms a long polymer chain.
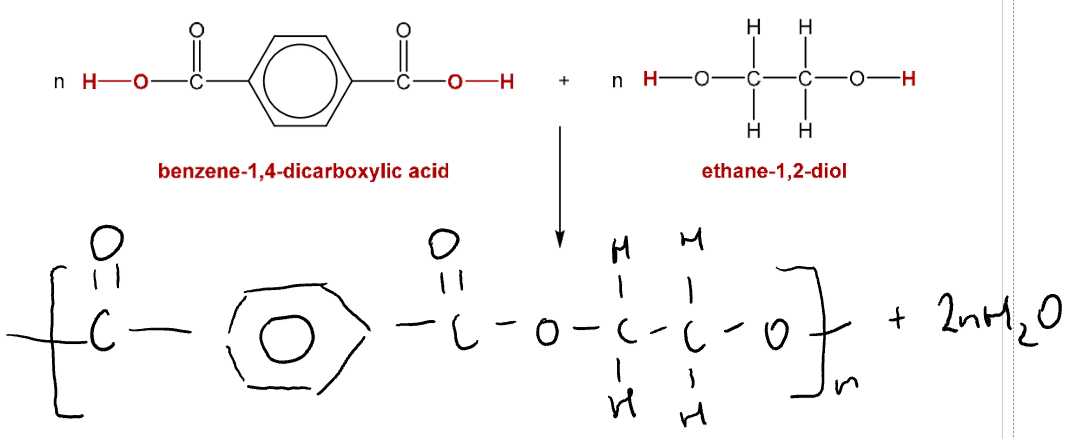
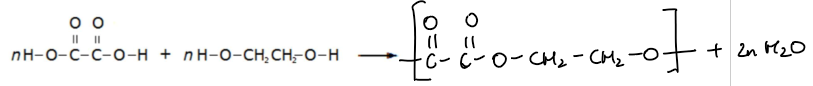
Example Reaction: Condensation polymerisation between ethanedioic acid and ethane-1,2-diol
Word equation:
Ethanedioic acid + Ethane-1,2-diol → Polyester + Water
Terylene
Made from ethane-1,2-diol + benzene-1,4-dicarboxylic acid
Water is released, ester linkages formed
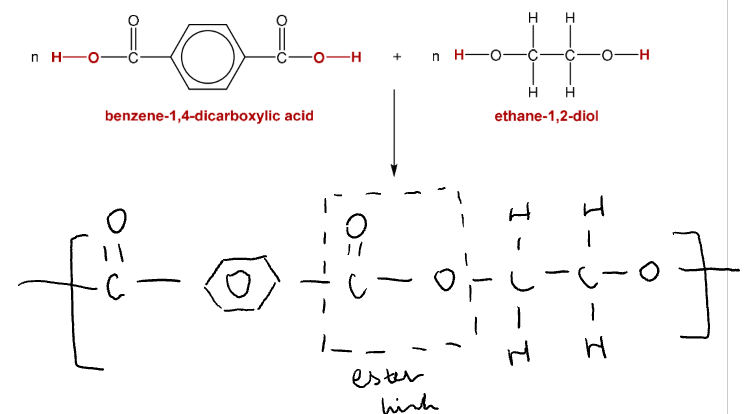
Properties:
Strong fibres, retain permanent creases (useful for trousers)
Low melting point due to weak intermolecular forces between chains
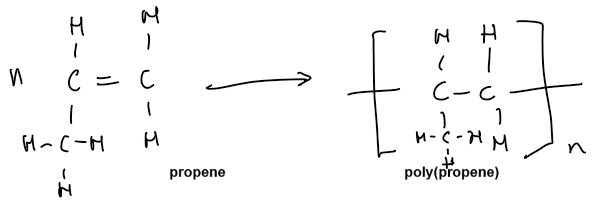
COMPARISON BETWEEN ADDITION AND CONDENSATION POLYMERS
ADDITION POLYMERISATION | CONDENSATION POLYMERISATION |
One type of monomer | 2 types of monomers |
Monomer is an alkene with a C=C double bond | Monomers are a dicarboxylic acid and a diol |
One product, the addition polymer | 2 products, the condensation polymer and water |
Backbone of repeat unit is made up of only C | Repeat unit has an ester OR amide linkage |
Addition polymerisation:
Condensation polymerisation: