Chemistry of Hydrocarbons and Combustion
Hydrocarbons
A hydrocarbon is defined as a compound consisting only of hydrogen and carbon atoms.
Crude Oil
Crude oil is characterized as a mixture of various hydrocarbons.
Fractional Distillation
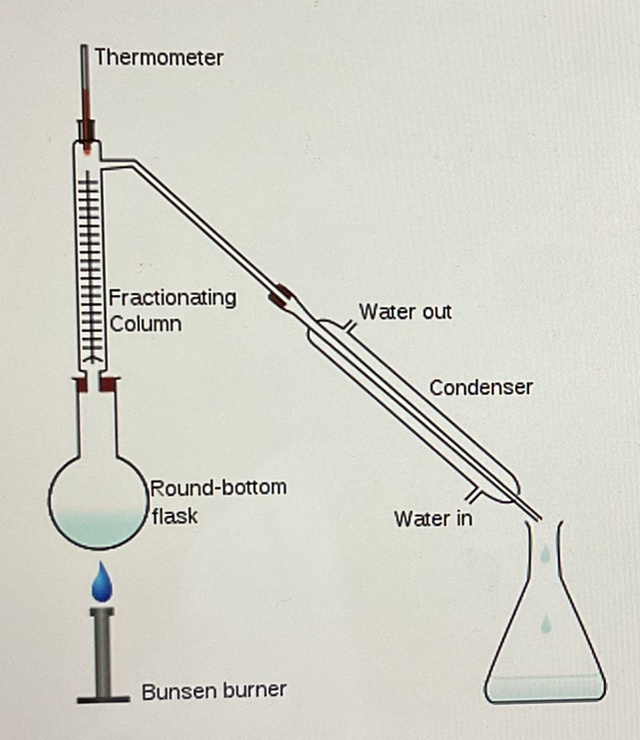
Fractional distillation is an industrial process that separates crude oil into different fractions based on their boiling points.
The fraction with the lowest boiling point is separated first
The process involves the following steps:
Crude oil is heated, vaporized, and transformed mostly into gas.
The vapor enters a fractionating column that has a temperature gradient, being hot at the bottom and cooler at the top.
The vapors rise up the column and begin to cool, condensing at different heights within the column, as each substance condenses at its specific boiling or condensing point.
Molecules with longer carbon chains, which have higher boiling points, condense near the bottom of the column, while those with shorter chains condense near the top.
Each fraction collected contains molecules with similar boiling points.
Main Fractions of Crude Oil
The primary fractions obtained from crude oil and their uses are:
Refinery Gases: Used in domestic heating and cooking.
Gasoline (Petrol): Used as a fuel in cars.
Kerosene: Used as a fuel in aircraft.
Diesel: Used as a fuel in some cars and larger vehicles like trains.
Fuel Oil: Employed as a fuel for large ships and in some power stations.
Bitumen: Used for surfacing roads and roofs.
Trend in Physical Properties
The main fractions exhibit trends in color, boiling point, and viscosity:
As the size of the hydrocarbon molecules increases (higher number of carbon atoms), the color tends to darken, the boiling point increases, and the viscosity increases.
Fuels and Combustion
A fuel is defined as a substance that, when burned, releases heat energy.
Combustion reactions are crucial in understanding fuel behavior:
Complete Combustion: Occurs when hydrocarbons react with sufficient oxygen, producing carbon dioxide and water:
Hydrocarbon + Oxygen → Carbon dioxide + water canIncomplete Combustion: Happens when oxygen supply is limited, resulting in carbon monoxide (a toxic byproduct) and possibly soot: Hydrocarbon + insufficient Oxygen → (toxic) Carbon Monoxide + Water + [less produced: Carbon dioxide + Carbon (soot)]
Carbon Monoxide Poisoning
Carbon monoxide (CO) is poisonous because it can bind to hemoglobin in red blood cells, impeding their ability to transport oxygen. This can lead to symptoms of fainting, coma, or even death due to reduced oxygen availability in the bloodstream.
Environmental Impact of Combustion
Combustion of fossil fuels can produce harmful environmental effects:
Sulfur Dioxide Emission: Some hydrocarbons contain impurities, such as sulfur. When these fuels are burned, sulfur reacts with oxygen to form sulfur dioxide (SO2).
Formation of Acid Rain: Sulfur dioxide can react with water vapor in the atmosphere, creating dilute sulfuric acid, thus increasing rainwater acidity.
Rainwater is naturally slightly acidic due to CO2 mixing with it, forming carbonic acid.
Nitrogen Oxides Impact: Similar to sulfur oxides, nitrogen oxides can react with water vapor forming nitric acid, further adding to rainwater acidity.
Reactions in Car Engines:
In car engines, high temperatures facilitate reactions between nitrogen and oxygen in the air, leading to the formation of nitrogen oxides (NO and NO2).
Consequences of Acid Rain
Acid rain has detrimental effects, including:
Killing aquatic life in lakes and rivers.
Damaging plant leaves and reducing their ability to absorb minerals.
Erosion of statues and buildings due to corrosive effects.
Global Warming and Fossil Fuels
The combustion of fossil fuels contributes to an increase in carbon dioxide, a greenhouse gas associated with global warming.
Higher levels of greenhouse gases in the atmosphere correlate with rising global temperatures, leading to climate change with potentially catastrophic effects.
Increased Frequency and Intensity of Extreme Weather Events: Climate change results in more frequent and severe hurricanes, droughts, and heatwaves, resulting in significant damage to infrastructure and loss of life.
Melting Ice Caps and Rising Sea Levels: The polar ice caps and glaciers are melting, contributing to rising sea levels that threaten coastal cities and ecosystems.
Ocean Acidification: Increased CO2 levels lead to higher ocean acidity, impacting marine life, particularly coral reefs and shellfish, which are vital to marine ecosystems and human economies.
Disruption of Ecosystems: Altered weather patterns and temperatures can disrupt habitats, leading to the extinction of species unable to adapt, and negatively impacting biodiversity.