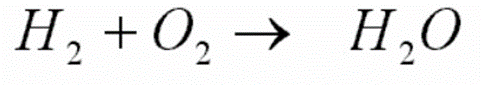Science: What you need to know for your exam…
Energy:
10 different forms of energy: kinetic and potential - kinetic = sound, mechanical, light, radiant, electric, thermal. Potential = nuclear, mechanical also, gravitational, elastic, chemical.
Difference between energy transfer and energy transformation: Energy transfer is the transferring of energy into different objects. Energy transformation is the converting of energy into different forms.
Be able to draw or describe a Sankey diagram.
Rectangle with the total energy supplied first, then an approximate accurate size for the arrow of useful energy, and the arrow going down for wasted energy.
Calculate the percentage efficiency equation:
Useful/Total energy x 100
Describe the particle model of matter: The particle model of matter is a theory that states:
all matter is made up of particles too small to see with the human eye
they are constantly moving
there is nothing in the space between particles
Describe the difference between solids, liquids and gases in terms of the particle model of matter:
Solids are tightly packed together and usually form a regular pattern. There is little space between particles.
Liquids are less packed together but their particles are usually still touching. They don’t form a regular pattern.
Gases are not packed together and randomly moves with much space between particles. They don’t form a regular pattern.
Exothermic vs endothermic:
Exothermic chemical reactions release heat so the surrounding areas are hot.
Endothermic chemical reactions absorb heat so the surrounding areas are cooler.
Difference between conduction, convection and radiation. Conduction occurs with two objects or materials that directly come in contact with each other. Convection occurs with heating from fluids (liquids and gases) where the colder, denser particles sink to the ground and the hotter particles expand and rise to the top in a continuous cycle until all the fluid is heated. Radiation occurs when electromagnetic waves heat objects without direct contact.
Example of conduction, convection and radiation.
Conduction: touching a cold ice cream.
Convection: a kettle boiling water.
Radiation: feeling the sun’s heat while outside.
Difference between independent variable, dependent variable and controlled variables.
Independent variable: the variable that is changed in an experiment. Eg. if you were measuring the difference in bean plants’ growth due to different amounts of sunlight, the different amounts of sunlight would be independent variable.
Dependent variable: the variable being measured. Eg. in the same example, the dependent variable would be the plants’ growth.
Controlled variable: the variable that stays the same. May be multiple of them. Eg. in the same example, the controlled variable/s would include the same bean plant being used, and same amount of water given to each bean plant.
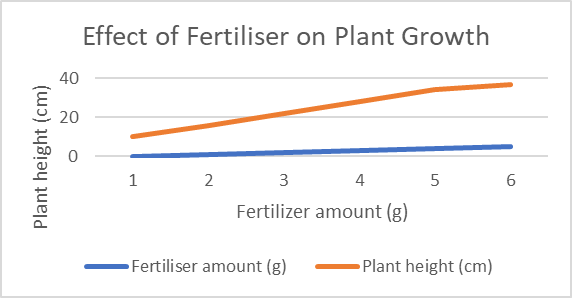
What does this graph depict?
This graph depicts the different heights of plants in different fertiliser amounts.
Create a hypothesis (if, then, because) for this:
If the amount of fertiliser increases, then the plant height will increase and the plant will flourish, because fertiliser provides essential nutrients that can boost a plant’s growth. However, if too much fertiliser is added, then the plant’s growth will be stunted and it will decrease in height, because there is an imbalance in nutrients and the roots will be damaged.
Difference between physical and chemical change:
A physical change is a reversible change where there is no change in chemical structure and only the appearance looks different. It could be melted, evaporated, cooled, etc. Eg. dissolving salt in water. A chemical change is an irreversible change where the chemical structure changes from rearranged particles and a new substance is created. It could change colour, give off odour, etc. Eg. wood burning.
List the first 20 elements and symbols in the periodic table.
H-Hydrogen, He-Helium, Li-Lithium, Be-Beryllium, B-Boron, C-Carbon, N-Nitrogen, O-Oxygen, F-Fluorine, Ne-Neon, Na-Sodium, Mg-Magnesium, Al-Aluminium, Si-Silicon, P-Phosphorus, S-Sulfur, Cl-Chlorine, Ar-Argon, K-Potassium, Ca-Calcium.
Difference between groups and periods in periodic table.
There are 18 groups, they are the vertical ones.
There are 7 periods, they are the horizontal ones.
Examples of metals and non-metals (2 will do):
Metals: Could include: Iron, Copper.
Non-metals: Could include: Hydrogen, Oxygen.
Examples of:
Metalloids: could include silicon, boron.
Noble gases: could include helium, neon.
Halogens: could include fluorine, chlorine.
Transition metals: could include gold, silver.
Alkali metals: could include lithium, potassium.
Alkali earth metals: could include beryllium, magnesium.
Main properties of specific groups:
Alkali metals (group 1): very reactive, they have only one electron.
Alkaline earth metals (group 2): more dense and slightly less reactive than alkali metals. Highly metallic.
Halogens (group 17): Very reactive, low melting and boiling points.
Noble gases (group 18): Inert so they do not easily form compounds.
Identify the reactants and products in each chemical word equation:
Reactants = H2 and O2, product = H2O
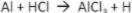
Reactants = Al and HCI, products = AlCl3 and H