Nuclear Chemistry - Chem Honors
Nuclear stability
changes occur in atom’s nucleus in nuclear reactions
strong nuclear force holds protons and neutrons together in nucleus
some mass of nucleons converted to energy (E = mc2) - mass defect
in larger atoms, nucleus spontaneously breaks apart releasing energy and particles - called radioactivity
nuclear symbol
lower # = atomic number; upper # = mass number
Ex. 238U or uranium-238 or U-238
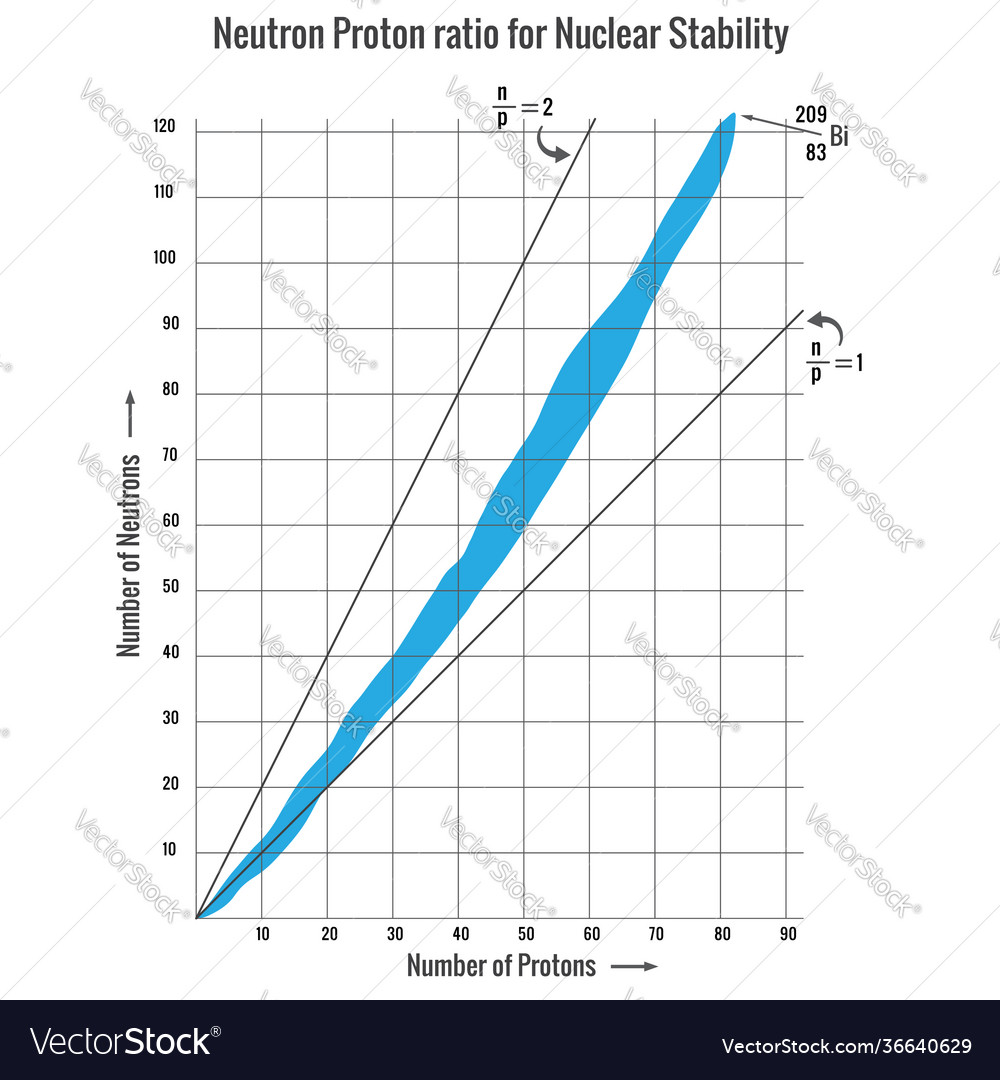
Ration of protons to neutrons
anything over Bi is unstable
Why are some atoms stable?
every element has one or more isotopes (isotope is element with dif atomic mass but same atomic number - difference in neutrons)
most isotopes are stable and not naturally radioactive
some are unstable
smaller atoms (elements under element 15) have ration of 1:1 neutrons to protons - stable
larger atoms have slightly higher ratio of neutrons to protons of 1.5:1
radioactive decay occurs when nucleus is unstable and starts to break apart releasing energy
Radioactive decay
spontaneous disintegration (decomposing/breaking down) of nucleus into slightly lighter and more stable nucleus
nuclear particles and/or electromagnetic radiation caused from reaction
transmutation - change in identity of an element as a result of a change in number of protons
Ex. 23890U → 23490U + 42He - shows conservation of mass and charge
no stable isotopes of elements with atomic number above Pb-82
bismuth and larger are all unstable radioactive isotopes
decay of these naturally unstable elements is called natural radioactivity
Alpha decay α
emission of an alpha particle by the nucleus
least penetrating power
equivalent to a helium nucleus - 42He
Ex. 21084Po → 20682Pb + 42He
Beta decay β-
emission of an electron by the nucleus
medium penetrating power
equivalent to an electron 0-1e
Ex. 146C → 147N + 0-1e
7 and -1 = 6 so conservation of charge
neutron decays into proton and electron which is why you gain a proton from C to N
Gamma emission γ
emission of high energy electromagnetic waves
high penetration power
no mass and no charge 00γ
Positron emission β+
emission of a positively charged electron
lower penetrating power
positively charged electron 0+1e
Ex. 3819K → 3818Ar + 0+1e
opposite of neutron decaying into proton and electron
Electron capture
capture of inner orbital electron by nucleus
electron combines with proton to form neutron
Ex. 10647Ag + 0-1e → 10646Pd
Rays experiment (Rutherford)
alpha particles will be attracted to negative plate
beta particles will be attracted to positive plate
gamma rays will not be affected by the charged field
Radioactive decay series
radioactive decay of naturally occurring radioisotopes of heavy elements often initiate chain of successive decay reactions
decay series ultimately leads to formation of a stable isotope
Half life
time it takes for 1/2 of atoms in a sample of radioactive material to decay (transmute) into another element
shorter the half life, more unstable the element
half life helps determine how long a sample of a useful isotope will be available
Ex. medical scans
determine how long a sample of an undesirable isotope must be stored before it decays to a low radiation level
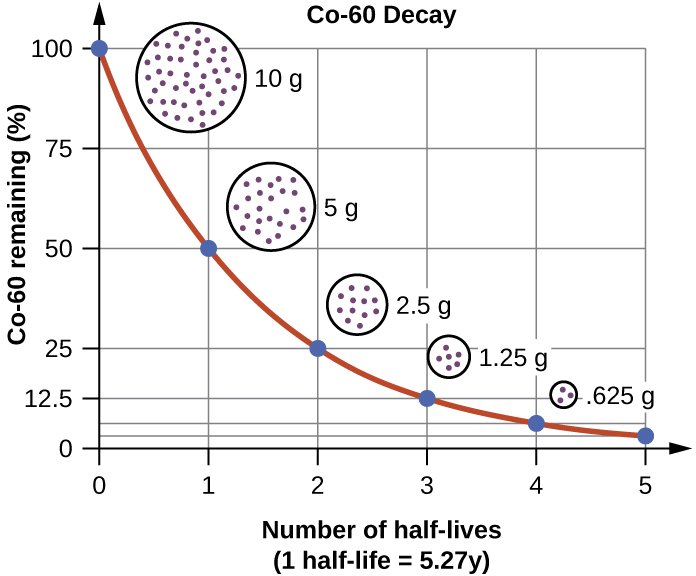
Ex. Co-60
start with 10g of Co-60
after 5.27 years there will be 5 g
after 10.54 years there will be 2.5g
after 15.81 years there will be 1.25g
 Example of a 10g substance with a 6 year 1/2 life
Example of a 10g substance with a 6 year 1/2 life
# of 1/2 lives elapsed | Fraction remaining | % remaining | ½ life (6 years for example) | Sample remaining (10g for example) |
0 | 1 | 100 | 0 | 10g |
1 | 1/2 | 50 | 6 | 5g |
2 | 1/4 | 25 | 12 | 2.5g |
3 | 1/8 | 12.5 | 18 | 1.25g |
4 | 1/16 | 6.25 | 24 | 0.625g |
5 | 1/32 | 3.125 | 30 | 0.3125g |
Examples
problem: half life of I-131 is 8 days. what mass remains unchanged from a 400g sample after 32 days?
solution:
0 days | 400g |
|---|---|
8 days | 200g |
16 days | 100g |
24 days | 50g |
32 days | 25g |
problem: half life of 131I is 8 days and 4g remain unchanged after 48 days. what was the mass of original sample and how many half lives have passed?
solution:
0 days | 256g |
|---|---|
8 days | 128g |
16 days | 64g |
25 days | 32g |
32 days | 16g |
40 days | 8g |
48 days | 4g |
problem: after 36 minutes, 5g remains from original sample of 40g. calculate half life of nuclide
solution:
0 min | 40g |
|---|---|
12 min | 20g |
24 min | 10g |
36 min | 5g |
problem: what fraction of K-42 remains after 24.8 hours
solution:
0 hours | 1 fraction |
|---|---|
12.36 hours | 1/2 fraction |
24.72 | 1/4 fraction |