Rates of reaction
To be able to compare rates of reaction obtained from different techniques the experimental data is converted into concentration.
Take the following reaction for example
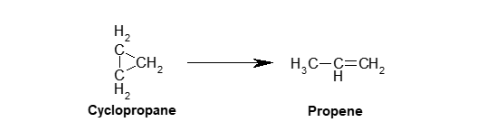
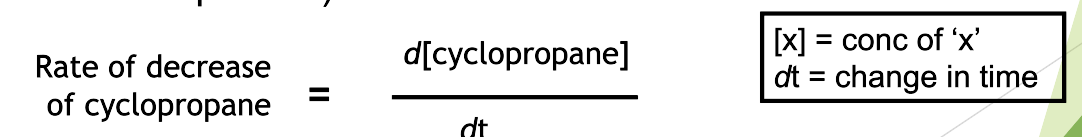
Rate of reaction can be regarded as the change in concentration (of a reactant or product) with unit time
Effect of concentration on rate
How can we find the rate of the reaction?
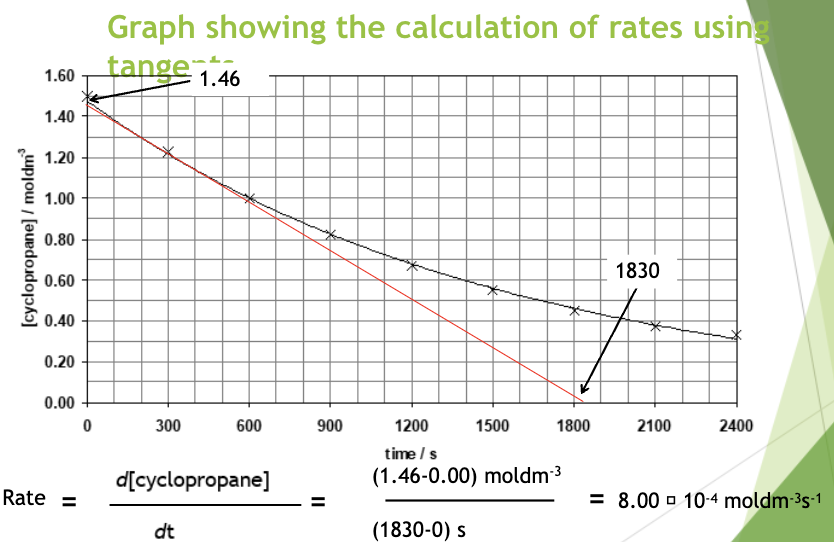
By drawing a tangent to the curve at various points we can calculate the rate of reaction at that specific point
Remember:-
Tangents should only touch the curve at 1 point
The tangent should be of a reasonable length, it can be useful to extend it to both axes but this isn’t strictly necessary
The angle between the tangent and the point above and below the curve should be the same
Dependence of rate on concentration
How does the rate depend on concentration?
To find out a graph is plotted of rate versus concentration
Concentration / moldm-3 | Rate / moldm-3s-1 |
1.20 | 8.00 × 10-4 |
0.90 | 6.00 × 10-4 |
0.60 | 4.00 × 10-4 |
By looking at the gradient we can see that if the concentration is halved then the rate is also halved
The rate is directly proportional to the concentration of cyclopropane
rate x [cyclopropane]
Instead of saying rate ∝[cyclopropane] we can introduce a proportionality constant - k is called a rate constant.
rate = k[cyclopropane]
This is called the rate equation
Rate equations
The exact rate equation depends on the exact reaction that is being investigated
The rate equation can only be determined empirically ie by experimentation
Rate constant
From our cyclopropane results we can see that the rate constant is 6.7 × 104s-1
NOTE the units of rate constants depend on the exact rate equation