Chemistry and physics
A chemical reaction is the interaction of substances to make new substances
Example 1: A+B → C+D
A plus B reacts to form C plus D.
The substances that react are called reactants (left of arrow). The substances that are made are called products (right of arrow).
A chemical change happens when a chemical reaction makes new chemical substances. A physical change happens when no new chemical substances are made.
H20 - the small “2” shows there are two atoms of hydrogen. No number shows there is one atom of oxygen. Two capital letters means there are two elements.
Hydrogen = 2 atoms
Oxygen = 1 atom.
We can also include "big numbers" at the start of molecules as we have seen before - 2H20 The big "2" shows there are two molecules.
Hydrogen = 2x2 = 4 atoms
Oxygen = 2×1 = 2 atoms
We can also write a chemical formula from a diagram of a chemical Structure.
A substance can be named from its chemical formula:
NaF = sodium and flourine - sodium flouride
LizS = lithium and sulfur - lithium sulfide
Carbonate : CO2
Sulfate : SO4
Nitrate : NO3
Hydroxide : OH
A + B → C + D
“A and B react to form C and D”
Example 1
“Carbon and Oxygen react to form carbon dioxide”
Carbon + Oxygen → Carbon dioxide
Example 2
Methane is produced from a reaction between Carbon and Hydrogen
Carbon + Hydrogen → Methane
Numbers written in standard form must:
start with a number between 1 and 10
be followed by a ‘x10’ raised to a power
A number between 1 and 10 (excluding 10) - a x10b - any number (can be a negative)
Prefixes:
tera - T - x10(12)
giga - G - x10(9)
mega - M - x10(6)
kilo - k - x10(3)
centi - c - x10(-2)
milli - m - x10(-3)
micro - µ - x10(-6)
nano - n - x10(-9)
Power is the rate at which energy is transferred.
1 watt = 1 joule per second
scooter - 250 W motor
The scooter transfers 250 joules per second
Power = energy transferred. time
Efficiency tells you the proportion of energy which is usefully transferred.
Efficiency = useful energy output. total energy input
There are NO units for efficiency
Work done is the amount of energy transferred by a force.
Work done is measured in joules.
work done = force x distance
W = F x s
Gravitational potential energy is the energy an object has due to its height above the ground.
gravitational potential energy = mass x gravitational field strength x change in height
E(p) = m x g x h
Kinetic energy is the energy an object has due to its speed
kinetic energy = 0.5 x mass x speed(2)
E(k) = 0.5 x m x v(2)
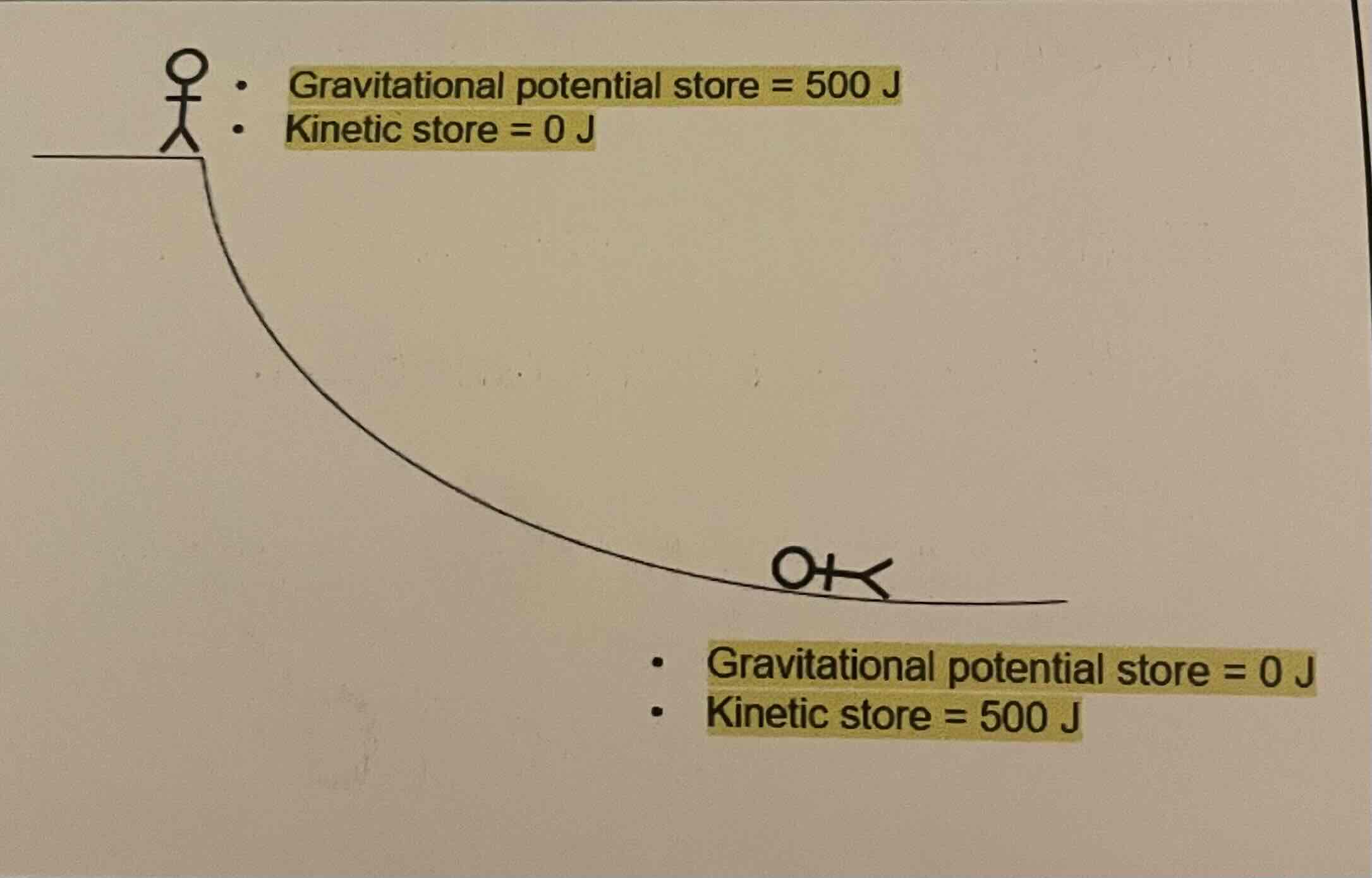
Often in physics we ignore frictional forces such as air resistance.
This means that we don’t need to worry about energy lost to the surroundings.
We assume that all energy moves from one store to another without any loss.
All energy in the GPE store moves to the KE store.
Anomalies are results which do not fit the pattern.
Random errors are made by the person carrying out the task
Systematic errors are a series of measurements that differ from the true value by the same amount each time
Zero errors occur when the equipment doesn’t reset to zero properly