Electron Transport Chain Lecture
Overview of Cell Respiration
Unit Focus: Completion of the topic on cell respiration.
Lab Due Date: Photosynthesis labs and reports need to be submitted.
Lab packets must be turned into the designated tray.
Notebooks must be left in the back cabinet by eighth period; late submissions may incur penalties.
Instructor offers to check notebooks after notes to facilitate completion before the weekend.
Introduction to Cell Respiration
General Concept: Cell respiration processes the energy stored in glucose.
Initial Stage - Glycolysis:
Occurs in the cytosol.
Process of breaking down glucose to produce two pyruvate molecules.
Requirement: Oxygen is essential for pyruvate to enter the mitochondria.
Pyruvate and Mitochondria
Entry Requirement: Pyruvate cannot enter without oxygen, termed as the "cool friend" for accessing the mitochondria (often referred to as "club mitochondria").
Krebs Cycle
Process: After glycolysis, pyruvate proceeds to the Krebs cycle.
Carbon Molecule Removal: Releases carbons, generating reduced electron carriers: NADH and FADH₂.
ATP Yield:
From Glycolysis:
Gross Yield: 4 ATP (2 used; net yield of 2 ATP).
From Krebs Cycle:
Net ATP yield is 2 from one pyruvate; can be counted variably as 4 from one glucose molecule.
Energy Demand During Cellular Activities
ATP Usage:
High energy demand in muscle cells, e.g., 10,000,000 ATPs utilized every second in active muscle.
In 30 seconds of running, approximately 300 million ATPs are utilized by a single muscle.
Total energy demands per day can reach billions or trillions of ATP, necessitating efficient ATP production beyond glycolysis and Krebs Cycle yields.
Electron Transport Chain (ETC)
Final Stage of Cell Respiration: Occurs in the inner mitochondrial membrane.
Characterized as a pivotal series for electron transport down a gradient.
ATP Generation: 34 ATP produced from one glucose molecule in the presence of oxygen.
Composition of Mitochondria
Structure: Double membrane system in mitochondria.
Outer membrane: Acts as a barrier for entry; permits oxygen.
Inner membrane: Contains folds (cristae) which increase surface area for reactions.
Intermembrane Space: Fluid-filled space between membranes.
Electron Carriers
Overview of NADH and FADH₂ from glycolysis, pyruvate oxidation, and Krebs cycle:
NADH Production: 4 from glycolysis.
Link Reaction: 2 NADHs generated from two pyruvate molecules.
Krebs Cycle: 6 NADHs and 2 FADHs from one glucose molecule.
Total: 8 NADHs and 2 FADHs per glucose.
Mechanism Inside the Mitochondrial Matrix
Hydrogen Ion Dynamics:
NADH drops off electrons and releases hydrogen ions, causing them to be expelled from the VIP area (mitochondrial matrix).
Electron Dynamics: Electrons follow the transport chain down toward oxygen, facilitating redox reactions.
Proton Gradient Charting: Establish a gradient against which the protons aim to rebound, promoting the formation of ATP through ATP synthase.
Oxidative Phosphorylation
Important Process:
Definition: Adding a phosphate group ( ext{P}) to ADP to convert it into ATP.
Electron transport and the resultant hydrogens create a concentration gradient, which fosters ATP generation as protons flow through ATP synthase.
Chemiosmosis
Definition:
Movement of ions across a selectively permeable membrane, specifically protons in this case.
This movement generates a proton motive force which facilitates ATP synthesis.
Summary of ATP From Cell Respiration
Overall Yield Calculation:
Glycolysis: 2 ATP
Krebs Cycle: 2 ATP
Electron Transport Chain: 34 ATP
Total Approximation: Between 36-40 ATP from one glucose molecule, averaging about 38 ATP per cellular respiration event.
Implications of Oxygen Abundance
Oxygen’s Role: Critical for the functioning of aerobic respiration; acts as the final electron acceptor.
Consequences of Absence:
If oxygen is absent, the following occurs:
Pyruvate cannot enter the mitochondria and energy production halts.
Cells would deplete energy stores leading to cell disorder and death.
Learning Insights
Key Processes of Cell Respiration:
Glycolysis occurs in the cytosol, yielding minimal ATP.
Krebs looks after further processing within the mitochondria alongside the Electron Transport Chain, showcasing maximum efficiency of ATP production via oxidative phosphorylation.
Revisiting importance of electron carriers and how they enable the critical processes necessary for energy yield in cellular contexts.
Highlighting the complete workflow from glucose to ATP, underscoring energy flow dynamics in a biological context.
Instructor Closing Remarks
Questions and clarifications offered to ensure understanding of complex processes discussed in cell respiration, emphasizing the GBP in mitochondria and likelihood of mistakes that can happen when oxygen isn’t available.
Offered a recap and prepared students for an upcoming quiz stating importance of retention of the above core concepts, especially oxidative phosphorylation and chemiosmosis as pivotal to understanding cellular respiration mechanisms.
Overview of Cell Respiration
Introduction to cellular respiration, the metabolic pathway that converts biochemical energy from nutrients into ATP, releasing waste products.
Unit Focus: Completion of the topic on cell respiration.
Lab Due Date: Photosynthesis labs and reports need to be submitted.
Lab packets must be turned into the designated tray.
Notebooks must be left in the back cabinet by eighth period; late submissions may incur penalties.
Instructor offers to check notebooks after notes to facilitate completion before the weekend.
Introduction to Cell Respiration
General Concept: Cell respiration is the catabolic process that breaks down food molecules (like glucose) to release stored chemical energy, which is then captured in the form of adenosine triphosphate (ATP). This process is fundamental for sustaining life.
Overall Equation: The simplified equation for the complete oxidation of glucose is:
Initial Stage - Glycolysis:
Location: Occurs in the cytosol of the cell.
Process: Glycolysis ("sugar splitting") is a 10-step metabolic pathway that breaks down one molecule of glucose (a 6-carbon sugar) into two molecules of pyruvate (a 3-carbon compound).
Phases:
Energy Investment Phase: The cell invests 2 molecules of ATP to phosphorylate glucose and an intermediate. This destabilizes the glucose molecule.
Energy Payoff Phase: This phase produces 4 molecules of ATP (via substrate-level phosphorylation) and 2 molecules of NADH (an electron carrier) per glucose molecule.
Net Yield from Glycolysis: 2 ATP and 2 NADH.
Requirement for Aerobic Respiration: Oxygen is crucial for the subsequent steps, allowing pyruvate to enter the mitochondria. Without oxygen, pyruvate follows anaerobic pathways.
Diagram Suggestion: A diagram illustrating the 10 steps of glycolysis, showing glucose, key intermediates, ATP consumption/production, and NADH production would be highly beneficial here.
Pyruvate Oxidation (Link Reaction) and Mitochondria
Entry Requirement: Pyruvate, the product of glycolysis, must enter the mitochondria for further oxidation in aerobic respiration. This entry is contingent on the availability of oxygen.
Process: Once inside the mitochondrial matrix, each pyruvate molecule undergoes oxidative decarboxylation or the link reaction.
Conversion to Acetyl-CoA: A multienzyme complex catalyzes three key reactions:
A carboxyl group is removed from pyruvate, releasing it as a molecule of carbon dioxide ().
The remaining two-carbon fragment is oxidized, and the electrons transferred to to form NADH (an electron carrier).
The oxidized two-carbon fragment (acetate) attaches to coenzyme A, forming acetyl-CoA.
Yield per Glucose: From one glucose molecule (which yields two pyruvates), the link reaction produces 2 acetyl-CoA molecules, 2 NADH molecules, and 2 molecules.
"Cool Friend" Analogy: The analogy of oxygen being a "cool friend" for accessing "club mitochondria" highlights its absolute necessity for pyruvate's entry and subsequent processing. Without oxygen, the mitochondrial pathways cannot proceed, and pyruvate is shunted to fermentation.
Krebs Cycle (Citric Acid Cycle)
Process: After the link reaction, the acetyl-CoA molecules enter the Krebs Cycle (also known as the Citric Acid Cycle or TCA cycle), which takes place in the mitochondrial matrix. This cycle completes the breakdown of glucose by oxidizing acetyl-CoA.
Entry of Acetyl-CoA: The two-carbon acetyl group from acetyl-CoA combines with a four-carbon molecule (oxaloacetate) to form a six-carbon molecule (citrate).
Cyclical Nature: Through a series of eight enzyme-catalyzed steps, citrate is progressively broken down, ultimately regenerating oxaloacetate, thus completing the cycle.
Carbon Molecule Removal: During the cycle, two carbon atoms are released as molecules per acetyl-CoA molecule. This means a total of four molecules are released per glucose molecule from the two acetyl-CoA inputs.
Generation of Reduced Electron Carriers: The primary energy-carrying products of the Krebs cycle are the reduced electron carriers:
NADH: 3 molecules per acetyl-CoA (6 per glucose).
FADH₂: 1 molecule per acetyl-CoA (2 per glucose).
ATP Yield:
Directly, one molecule of GTP (guanosine triphosphate) is produced per acetyl-CoA, which is equivalent to 1 ATP per acetyl-CoA (2 per glucose). This is also via substrate-level phosphorylation.
Summary for two acetyl-CoA (per glucose): 4 , 6 NADH, 2 FADH₂, 2 ATP (or GTP).
Diagram Suggestion: A detailed diagram of the Krebs cycle, showing the entry of acetyl-CoA, intermediates, release of , and the specific points of NADH, FADH₂, and ATP/GTP generation, would be highly informative.
Energy Demand During Cellular Activities
ATP Usage: Cellular activities, especially those requiring kinetic energy, demand immense amounts of ATP.
High energy demand in muscle cells, e.g., ATPs utilized every second in active muscle.
In seconds of running, approximately million ATPs are utilized by a single muscle cell.
Total energy demands per day can reach billions or trillions of ATP, necessitating highly efficient ATP production beyond the minimal yields of glycolysis and the Krebs Cycle. This highlights the crucial role of the Electron Transport Chain.
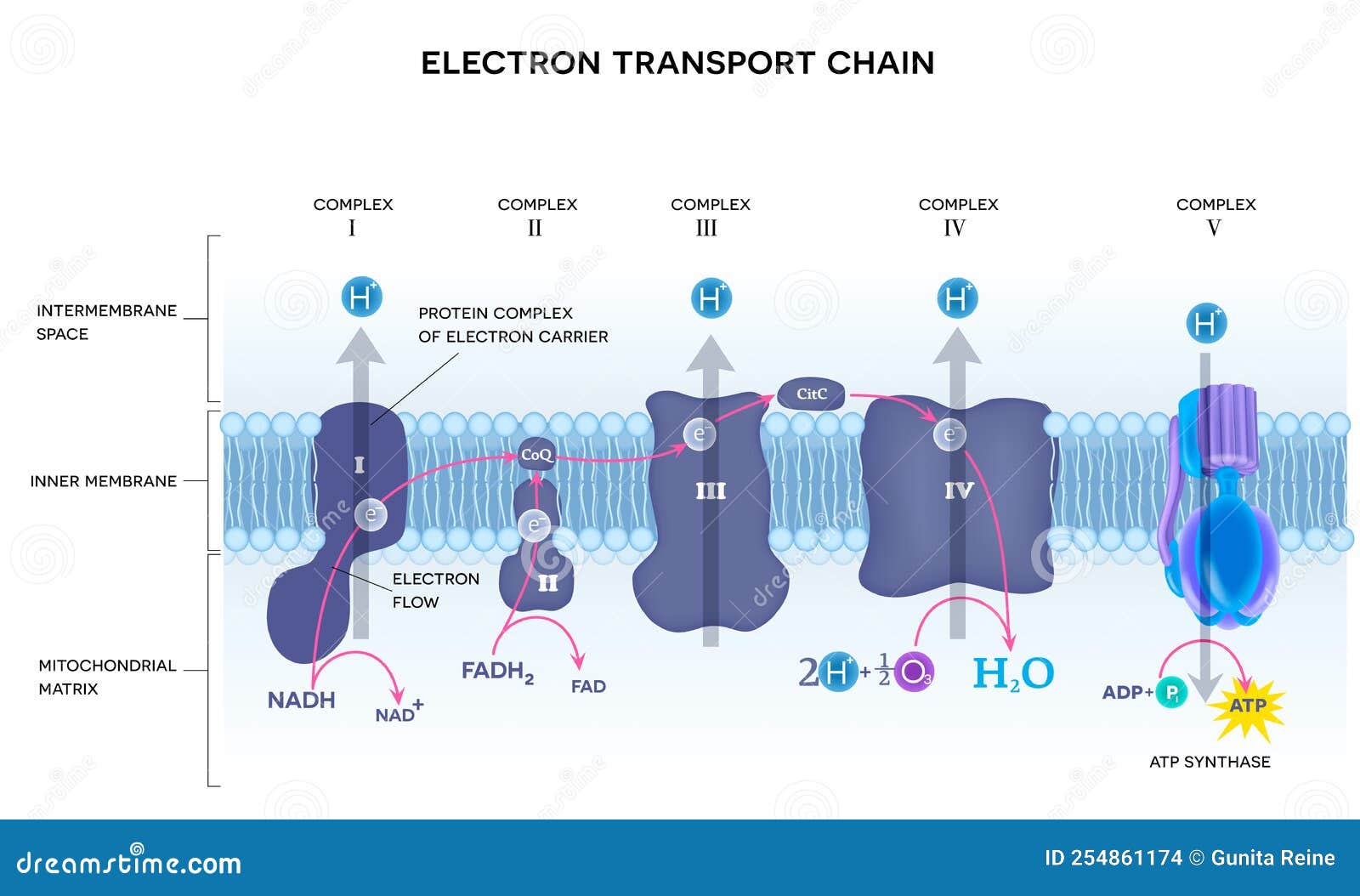
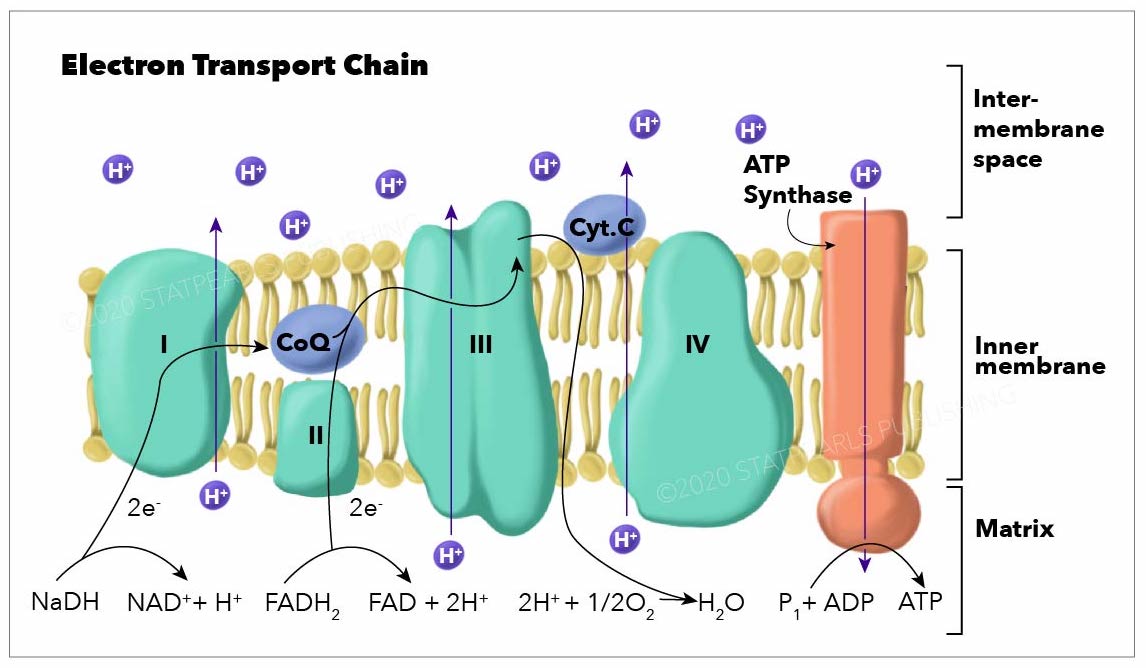
Electron Transport Chain (ETC)
Final Stage of Cell Respiration: The ETC is the third and most productive stage of aerobic respiration, occurring in the inner mitochondrial membrane. It is where the vast majority of ATP is generated.
Nature of the ETC: Characterized as a pivotal series of protein complexes embedded in the inner mitochondrial membrane, through which electrons are transported down an high-energy gradient.
Electron Flow:
Electron Donors: NADH and FADH₂ (generated in glycolysis, pyruvate oxidation, and the Krebs cycle) deliver their high-energy electrons to the ETC.
Complex I (NADH dehydrogenase): Accepts electrons from NADH, passing them down the chain.
Complex II (Succinate dehydrogenase): Accepts electrons from FADH₂, specifically from the succinate-fumarate conversion in the Krebs cycle.
Ubiquinone (Q) and Cytochrome c: Mobile electron carriers shuttle electrons between the fixed complexes.
Complex III (Cytochrome reductase) and Complex IV (Cytochrome oxidase): Further transport electrons, progressively lowering their energy level.
Proton Pumping: As electrons move through Complexes I, III, and IV, the energy released is used to pump hydrogen ions ( protons) from the mitochondrial matrix into the intermembrane space. This creates a high concentration of protons in the intermembrane space.
Oxygen's Role: Molecular oxygen () is the final electron acceptor at the end of the ETC (at Complex IV). It combines with electrons and protons to form water (). This continuous removal of electrons is essential for the entire chain to function.
ATP Generation: The electrochemical gradient of protons (the proton-motive force) drives the synthesis of ATP through a process called chemiosmosis, facilitated by ATP synthase.
Yield: Approximately ATP produced from one glucose molecule in the presence of oxygen, primarily through oxidative phosphorylation.
Diagram Suggestion: A diagram showing the inner mitochondrial membrane with Complexes I, II, III, IV, ubiquinone, cytochrome c, and ATP synthase. It should illustrate the flow of electrons, the pumping of protons into the intermembrane space, and the formation of water by oxygen.
Composition of Mitochondria
Structure: Mitochondria possess a unique double membrane system, crucial for its function in cellular respiration.
Outer Membrane: Smooth and permeable to small molecules, acting as a general barrier and defining the organelle's shape.
Inner Membrane: Highly folded into structures called cristae.
Significance of Cristae: These folds significantly increase the surface area available for the embedding of the electron transport chain complexes and ATP synthase molecules, thereby maximizing ATP production.
Permeability: Less permeable than the outer membrane, controlling the passage of substances into the mitochondrial matrix.
Intermembrane Space: The fluid-filled compartment located between the outer and inner mitochondrial membranes. This is where protons accumulate due to the activity of the ETC, establishing the proton gradient.
Mitochondrial Matrix: The innermost compartment, enclosed by the inner membrane. It contains enzymes for the Krebs cycle and pyruvate oxidation, as well as mitochondrial DNA and ribosomes.
Electron Carriers
Overview of the crucial roles of NADH and FADH₂ as high-energy electron shuttles, generated during glycolysis, pyruvate oxidation, and the Krebs cycle.
Detailed Production per Glucose Molecule:
Glycolysis: 2 NADH molecules are produced.
Link Reaction (Pyruvate Oxidation): 2 NADH molecules are generated (one per pyruvate).
Krebs Cycle: 6 NADH molecules (3 per acetyl-CoA) and 2 FADH₂ molecules (1 per acetyl-CoA) are yielded.
Total per Glucose: NADH and FADH₂. These carriers will then donate their electrons to the Electron Transport Chain.
Mechanism Inside the Mitochondrial Matrix: Proton Gradient and ATP Synthesis
Hydrogen Ion Dynamics:
NADH and FADH₂ deliver their electrons to the ETC complexes.
As electrons are passed down the chain, the energy released is harnessed to actively pump protons ( ions) from the mitochondrial matrix across the inner mitochondrial membrane into the intermembrane space.
Result: This creates a significant concentration gradient of protons, with a much higher concentration in the intermembrane space compared to the matrix. This gradient is often referred to as a proton-motive force.
Electron Dynamics: Electrons follow the transport chain down toward oxygen, which acts as the ultimate electron acceptor, facilitating a series of redox reactions. Each transfer releases a small amount of energy.
Proton Gradient as Energy Source: The established proton gradient represents a form of potential energy. Protons naturally tend to diffuse back down their concentration gradient into the mitochondrial matrix.
Oxidative Phosphorylation
Important Process: This is the process that couples the electron transport chain with ATP synthesis.
Definition: Oxidative phosphorylation is the metabolic pathway in which cells use enzymes to oxidize nutrients, releasing chemical energy (in the form of electrons), which is used to make adenosine triphosphate (ATP). This involves adding a phosphate group () to ADP to convert it into ATP, driven by the energy from the proton gradient rather than direct enzymes (as in substrate-level phosphorylation).
Mechanism: The electron transport and the resultant pumping of hydrogen ions create a powerful electrochemical gradient. This gradient provides the energy for ATP synthase.
Chemiosmosis
Definition: Chemiosmosis is the movement of ions across a selectively permeable membrane, down their electrochemical gradient. In cellular respiration, this specifically refers to the flow of protons ( ions).
Role in ATP Synthesis:
The high concentration of protons in the intermembrane space creates a strong proton-motive force.
Protons can only re-enter the mitochondrial matrix by passing through a transmembrane protein complex called ATP synthase.
As protons flow through ATP synthase, the energy of their movement causes a conformational change in the enzyme, which catalyzes the phosphorylation of ADP to ATP (ADP + ATP). This process is analogous to a turbine being turned by flowing water.
Diagram Suggestion: A diagram illustrating ATP synthase embedded in the inner mitochondrial membrane, showing protons flowing through it and driving the synthesis of ATP from ADP and inorganic phosphate.
Summary of ATP From Cell Respiration
Overall ATP Yield Calculation (Approximate):
Glycolysis: 2 ATP (net)
Krebs Cycle: 2 ATP (directly from GTP)
Electron Transport Chain / Oxidative Phosphorylation: Approximately 28-34 ATP. (This range accounts for variations due to shuttle systems for cytosolic NADH).
Total Approximate Yield: Between ATP from one glucose molecule. The frequently cited average is about ATP per cellular respiration event for eukaryotes. This significantly higher yield demonstrates the efficiency of aerobic respiration.
Implications of Oxygen Abundance (and Absence)
Oxygen’s Critical Role: Oxygen is absolutely critical for the functioning of aerobic respiration. It acts as the final electron acceptor in the Electron Transport Chain, pulling electrons down the chain and allowing the continuous flow of electrons and proton pumping.
Consequences of Absence: Fermentation: If oxygen is absent or insufficient (anaerobic conditions), the ETC cannot operate because there is no final electron acceptor.
Accumulation of Electron Carriers: NADH and FADH₂ cannot unload their electrons, leading to a buildup of reduced carriers and a depletion of oxidized carriers ( and ).
Halt of Aerobic Pathways: Without and , the Krebs cycle and pyruvate oxidation stop.
Fermentation as an Alternative: To regenerate (essential for glycolysis to continue), cells resort to fermentation.
Lactic Acid Fermentation: In muscle cells and some bacteria, pyruvate is converted to lactic acid, regenerating from NADH. This allows glycolysis to continue, producing a small amount of ATP (2 net ATP per glucose).
Alcoholic Fermentation: In yeast and some bacteria, pyruvate is converted to ethanol and carbon dioxide (), also regenerating from NADH.
Energy Production Halts for Aerobic Pathways: In the absence of oxygen, the vast majority of ATP production (from the ETC) ceases, severely limiting the cell's energy supply.
Cellular Impact: Cells would rapidly deplete their energy stores, leading to cell dysfunction and ultimately death, unless they can switch to anaerobic metabolism or oxygen is restored.
Learning Insights
Key Processes of Cell Respiration:
Glycolysis: The initial breakdown of glucose, occurring in the cytosol and yielding a small amount of ATP and NADH.
Pyruvate Oxidation and Krebs Cycle: Further oxidation of glucose derivatives within the mitochondrial matrix, producing , ATP, NADH, and FADH₂.
Electron Transport Chain and Oxidative Phosphorylation: The main ATP-producing stage within the inner mitochondrial membrane, leveraging electron carriers to establish a proton gradient that drives ATP synthase.
Interconnectedness: Emphasizing the complete workflow from glucose to ATP, underscoring energy flow dynamics in a biological context and the intricate coordination between different metabolic pathways.
Electron Carriers (NADH, FADH₂): Their crucial role in transferring high-energy electrons from catabolic pathways to the ETC, thereby enabling the maximal efficiency of ATP production.
Instructor Closing Remarks
Questions and clarifications offered to ensure understanding of complex processes discussed in cell respiration, emphasizing the overall energy balance (GBP - perhaps referring to the "Great Big Picture" or "Grand Balance of Production") in mitochondria and likelihood of mistakes that can happen when oxygen isn’t available.
Offered a recap and prepared students for an upcoming quiz stating importance of retention of the above core concepts, especially oxidative phosphorylation and chemiosmosis as pivotal to understanding cellular respiration mechanisms.