Entropy And Energetics
Ionic Bonding Recap
The bigger the charge on an ion the stronger the electrostatic force of attraction
More energy required to overcome these forces so they have a high M.P and B.P
The Smaller the ion the stronger the electrostatic force of attraction between ions
Smaller ions pack together more closely and more energy is required to overcome the stronger forces. Therefore, the M.P and B.P are higher for smaller ions
Theoretical and Experimental lattice Enthalpies
Theoretical lattice enthalpies are calculated assuming a perfectly ionic model
Most the time the positive ion distorts the charge distribution in the negative ion. We say that the positive ion polarises the negative ion
The more polarisation the more covalent character there will be
Below is an image of the perfectly ionic model

The difference between experimental and theoretical lattice enthalpy of formation can tell you how much covalent character an ionic compound has.
The bigger the difference in lattice enthalpy the more polarisation you have the greater the covalent character
Polarisability
Smaller cations are more polarising than larger ones
Smaller cations have a higher charge density as the charge is concentrated in a smaller area. The cation pulls electrons towards itself more readily
Large anions with a large charge are polarised much more easily than smaller, lower charged anions.
This is because the electrons are further away from the nucleus and there is more repulsion between the electrons in the ion. The electrons can be pulled towards the cations.
The more polarisation the more covalent character
Most polarisation occurs when the cation has a high charge but small radii and the anion has a large negative charge and large radii
Electronegativity
Electronegativity is the ability to attract electrons towards itself in a covalent bond
The bigger the difference in the electronegativity (using the Pauling’s scale) the more ionic the compound will be a difference of 0 is purely covalent
Enthalpy Change of Solution
The enthalpy change when 1 mole of an ionic substance is dissolved in the minimum amount of solvent to ensure no further enthalpy change is observed upon further dilution
For a substance to dissolve
Substance bonds must break (endothermic)
New bonds formed between the solvent and substance (exothermic)
Most ionic compounds dissolve in polar solvents like H2O. The H+ is attracted to the negative ions and the O- is attracted to the positive ions.
The water molecules surround the ions in a process called hydration
For this to happen the new bonds formed must be the same strength or greater than those broken.
If not then the substance is very unlikely to dissolve. Soluble substances tend to have exothermic enthalpies of solution for this reason
Enthalpy of Hydration
2 things can affect the enthalpy change of hydration - Charge and Size of the ion
Charge:
Ions with a higher charge attract water molecules more strongly as the electrostatic attraction.
More energy energy is released when the bond is made which means they have a more exothermic enthalpy of hydration
The larger the charge the greater the enthalpy of hydration
Size:
Smaller ions have a higher charge density than larger ions
They can attract water molecules more strongly hence there is a more exothermic enthalpy of formation
The smaller the ion the greater the enthalpy of hydration
Entropy
Entropy is the measure of disorder in a system
It is the number of ways energy can be shared out between particles
The more disorder there is the higher the entropy
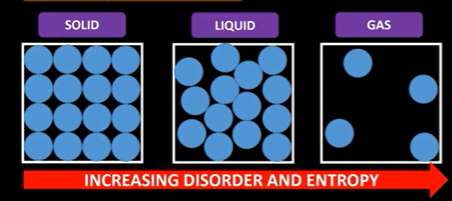
The number of particles also affects entropy change
If a reaction in the same state but more moles are produced then entropy increases. There are more ways energy can be distributed
A reaction can be spontaneous (feasible) even if it is enthalpically unfavourable for example endothermic reactions
A reaction will tend towards more disorder and hence increase entropy
Increasing entropy is energetically favourable and some reactions that are enthalpically unfavourable can still spontaneously react if changes in entropy overcome changes in enthalpy
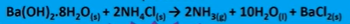
This reaction is endothermic and enthalpically unfavourable (+164 kJmol-1)
3 moles on the LHS and 13 moles on the RHS. Entropically favourable
Starting with 2 solids but making a gas and a liquid. Increased disorder so entropically favourable
Calculating Entropy
Entropy change (delta S) can be calculated between reactants and products.

Under standard entropy so:
1 mole of substance
100kPa
298K
Total entropy change can be calculated using entropy changes of the system and surroundings


Gibbs free Energy
Tells us if a reaction is feasible or not
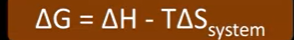
A reaction is feasible if the delta G is negative or zero
Even if the reaction is feasible you may not observe a reaction occurring. May be due to activation energy being too high or the rate of reaction being very slow
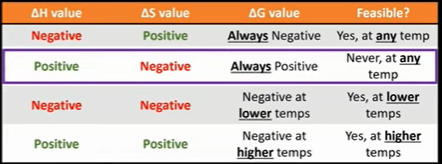
The temperature at which a reaction becomes feasible can be calculated
Just sub 0 in for delta G

Key point to remember is to convert delta H from kJ mol-1 to Jmol-1
Equilibrium constants are large when reversible reactions are feasible
When Delta G is negative and the reaction is theoretically feasible the equilibrium constants are greater than 1
When Delta G is negative and the reaction is not theoretically feasible the equilibrium constant are less than 1
