2.8 Mass Spectroscopy
Mass Spectroscopy
Definitions:
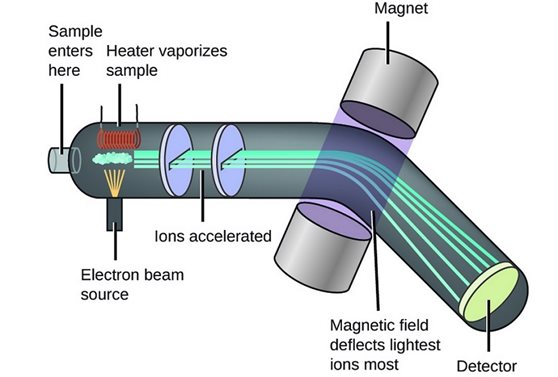
Vaporisation: The samples of particles are heated until they are a gas state.
Ionisation: Particles are bombarded with a stream of electrons, electrons are knocked out of sample particles.
Acceleration: An electric field accelerated the positive ions at high speed.
Deflection: Different ions are deflected by the magnetic field depending on mass/charge
Detection: The beam of ions are passed through the machine, only ions with 1+ charge will make it through the machine.
Uses of mass spectral data:
Identifying molecular mass of unknown compounds.
Gain info about structure of chemical properties.
Summary:
Positive ions are formed when atoms or molecules are bombarded with electrons, knocking out an electron to form M⁺.
Ions are separated based on their mass-to-charge ratio in the magnetic field, with lighter or more highly charged ions deflected more.
The mass spectrum generated shows the m/z ratio and relative abundance of the ions, providing information about the sample's composition.
Mass Spectroscopy of Organic Compounds:
Mass spectrometry of organic compounds provides the Mr from the molecular ion peak.
M → [M]+ + e-
Fragmentation: Molecular ions fragments form due to covalent bonds breaking:
[M]+ → X+ + Y•
Fragmentation Patterns:
The pattern of smaller peaks corresponds to fragment ions.
These fragments can provide information about the structure of the molecule.
The more stable the ion, the greater the peak intensity.
Example of ethanol:
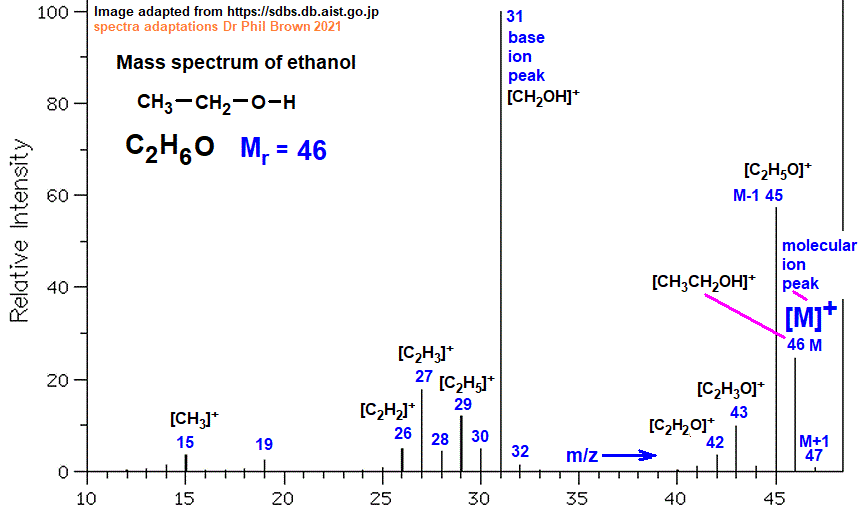
Interpreting the Mass Spectrum
Molecular Ion Peak (M⁺):
The peak with the highest m/z value (mass/charge) represents the molecular ion, giving the relative molecular mass (Mr) of the compound.
Base Peak:
The tallest peak in the spectrum is the base peak. It corresponds to the most abundant ion and is often a fragment ion. -(broken up from whole molecule)
Isotopic Peaks:
Peaks with slightly higher m/z values than the molecular ion arise due to isotopes (e.g., 13C^{13}C13C or 2H^2H2H). (not needed for exam).
Ethanol (C2H5OHC_2H_5OHC2H5OH)
Molecular ion peak (M+) at m/z = 46, representing the molecular ion [CH3CH2OH]+
Fragmentation may produce peaks such as:
[CH3]+ at m/z = 15
[CH3CH2]+ at m/z = 29
[CH2OH]+ at m/z = 31
[CH3CH2O]+ at m/z = 45