Thema 1: koolhydraatmetabolisme
Kennis en inzicht verkrijgen in:
o (regulering van) het glucose-transport (GLUT1, 2, 3 en 4),
o de functie van de glycolyse in verschillende celtypen, o de hiërarchie van regelmechanismen in de glycolyse,
o de novo synthese van glucose, i.e. de gluconeogenese (substraten, specifieke enzymen, cellulaire en sub- cellulaire lokalisatie),
o (regulering van) het glycogeenmetabolisme,
o de werking van de pancreas-hormonen [insuline/glucagon] en de bijnier-hormonen [(nor)-adrenaline]
o de secretie van insuline door een β-cel in de pancreas
Na afloop van dit thema kun je:
o Precies de regel-punten en -mechanismen van de glycolyse, gluconeogenese, glycogeen-afbraak en glycogeen-opbouw aangeven,
o Het begrip glucostase uitleggen,
o Globaal uitleggen hoe het (koolhydraat-)metabolisme hormonaal wordt aangestuurd,
o De verschillende functies van glycogeen in spier- en lever-weefsel uitleggen.
HC1: introductie & metabologica
metabole kaart mag je bij tt gebruiken - wat staat er op wat je wel moet weten:
namen vd metabole routes & enzymen
regulering vd paden & enzymen
namen metabole routes
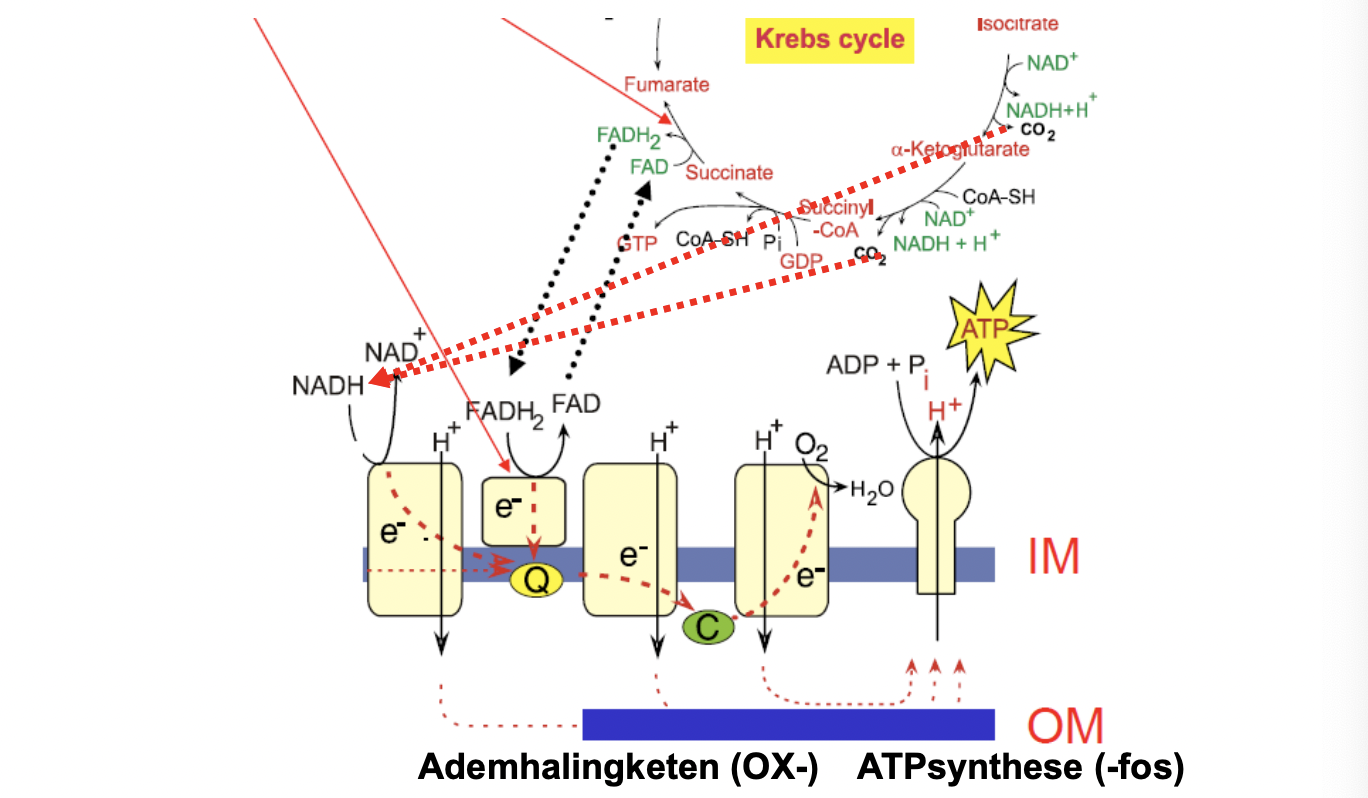
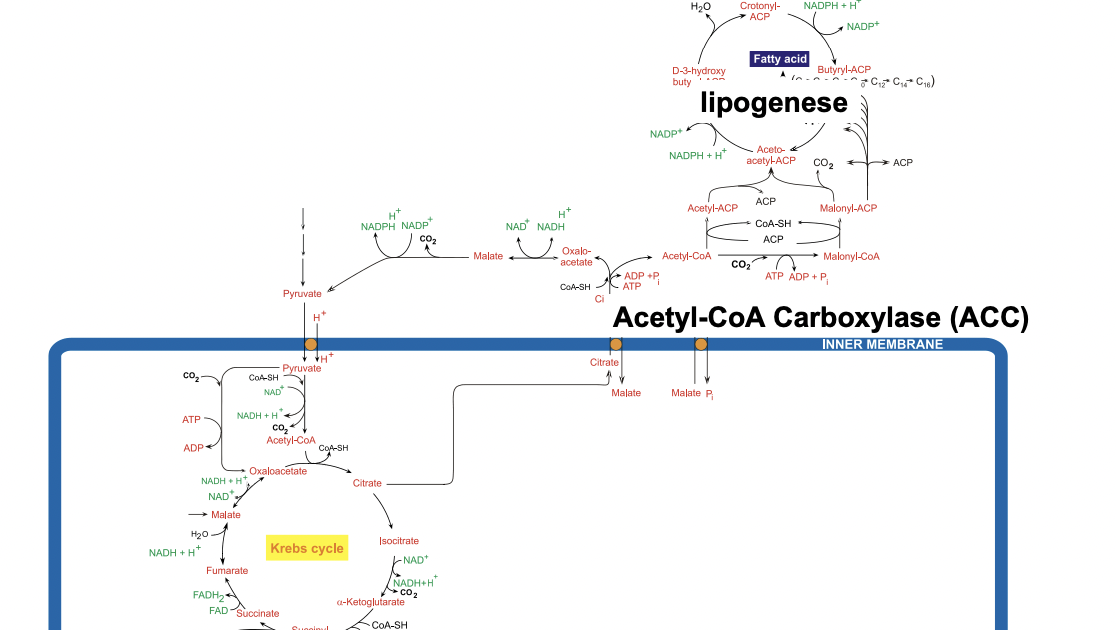
naamgeving enzymen
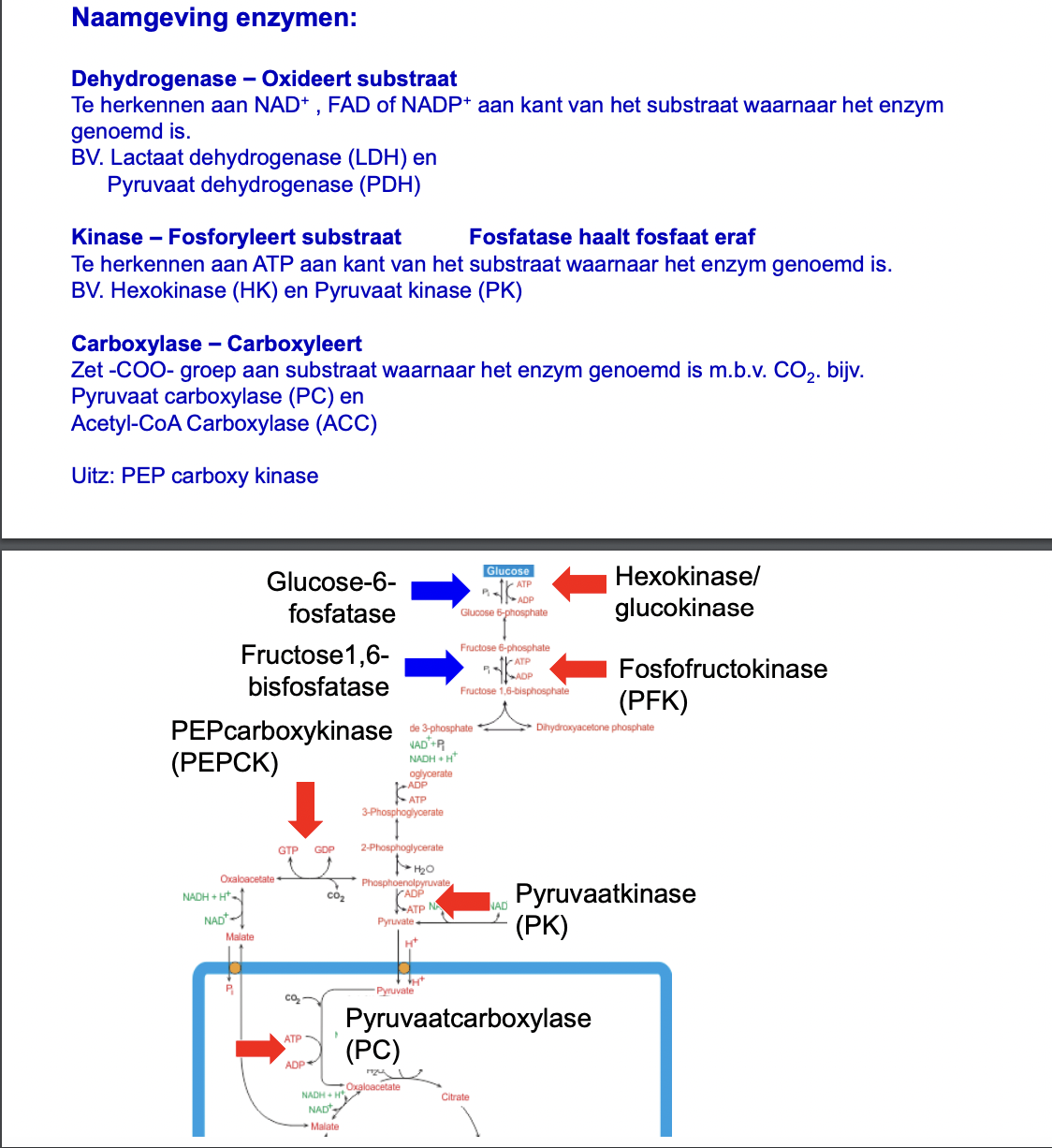
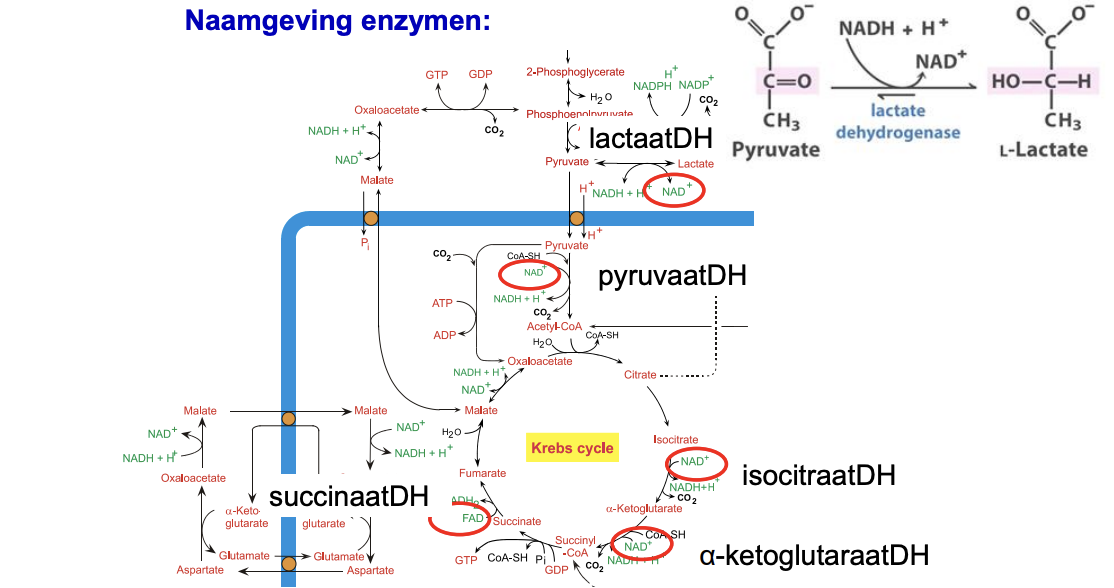
hoofdregels regulering
2 tegenovergestelde paden lopen niet tegelijk
snelheid glycolyse, krebs, oxfos wordt mede bepaald door ATP behoefte
HC2: regulering koolhydraatmetabolisme
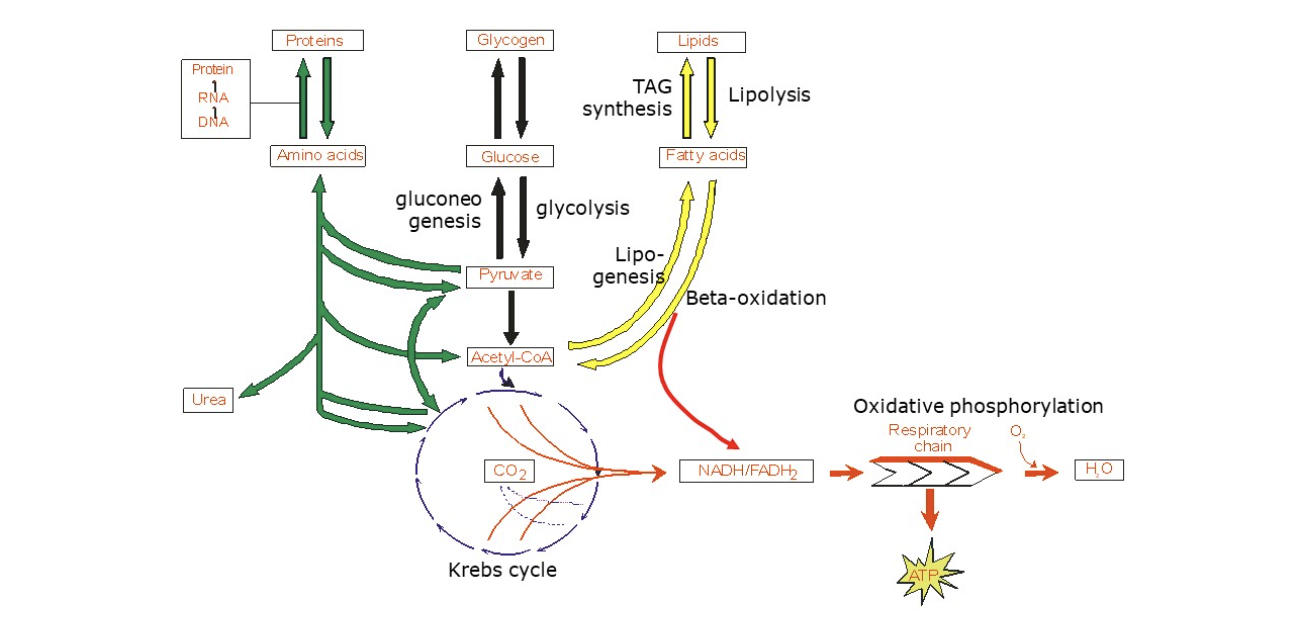
1 overzicht regulering koolhydraat metabolisme
glucose heeft meerdere metabole mogelijkheden:
glycolyse, KC, oxfos → E productie
PPP → NADPH + ribose-5-P (voor antioxidanten, biosynthese lipides & DNA/RNA)
opslag voor slechte tijden als
glycogeen in lever & spier
vet (via acetyl-CoA → FA → TAG)
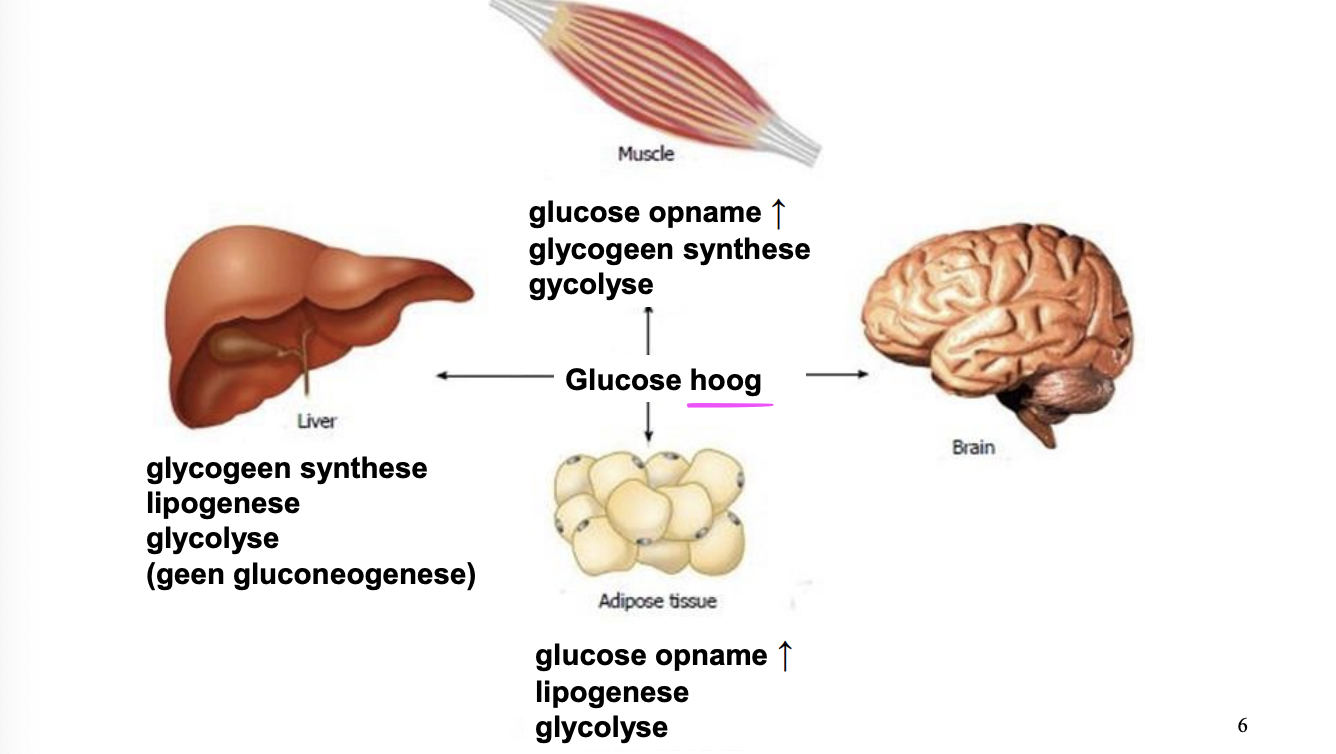
hoge glucose conc → glucose kan worden opgenomen, gebruikt voor energie of opgeslagen - welke optie verschilt per orgaan!
al deze processen worden geregeld door de hormonen insuline & glucagon
bij hoge glucose conc in bloed: insuline (uit beta cellen)
bij lage glucose conc in bloed: glucagon (uit alpha cellen)
2 insuline afgifte
3 glucose tranporters (GLUT)
4 regulering glycogeenmetabolisme
5 regulering glycoluse & gluconeogenese (F-2,6-BP)
e-module: Glucostase
LOs
effects of insulin & glucagon on a cellular level (1)
relationships between organs regarding blood glucose levels and the roles of insulin & glucagon (2)
effects of rising & dropping glucose levels and reactions of different organs (2)
pathophysiology of glucostatic disorders diabetes mellitus & insulinoma (3)
0. introductie
0.1 properties insulin & glucagon
insulin & glucagon are peptide hormones with antagonistic functions - produced by cells in islets of Langerhans in endocrine pancreas
after meal → blood glucose rises → insulin (produced in β-cells) released into bloodstream → lowers blood glucose by bringing it into tissues
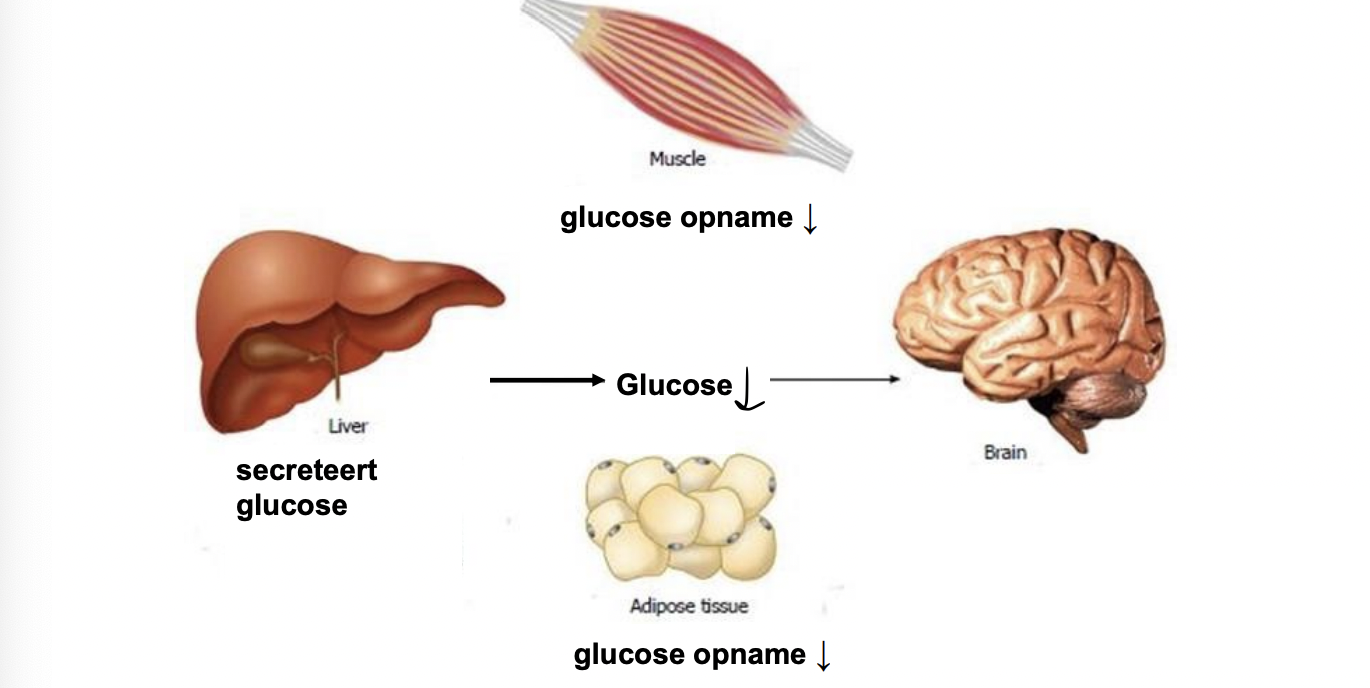
after fasting/exercise → blood glucose drops → glucagon (produced in α-cells) released → increases blood glucose by bringing it from liver into blood
insulin is an anabolic hormone = its effect is a net tissue buildup & energy supply formation
high insulin in blood signals there’s high energy (glucose or TAG) →
surplus E must be stored as glycogen fat or protein
insulin has receptors in multiple organs so E can be stored
E store breakdown must be stopped
glucagon is a catabolic hormone = its effect is a net tissue breakdown & energy supply use
high glucagon signals energy shortage (vnl glucose)
brings glucose into bloodstream by
breaking down E supplies
producing new glucose from dif precursors
peptide hormones so → receptors on outside of cell - binding causes signaling cascade
insulin binds → activation protein/lipid kinase cascade → dephos of target p
glucagon binds (G-p coupled) → production cAMP → activates PKA → phos target p
both secreted on basal level → both always present in blood
→ speak of insulin/glucagon ration ipv presence/absence
blood glu high →insulin high = INS/GCN-ratio high
glucagon high = INS/GCN-ratio low
0.2 metabolism
insulin & glucagon also have opposite effects on metabolism
insulin → glycolyis & formation glycogen and fat
glucagon → gluconeogenesis & breakdown glycogen and fat
catabolism
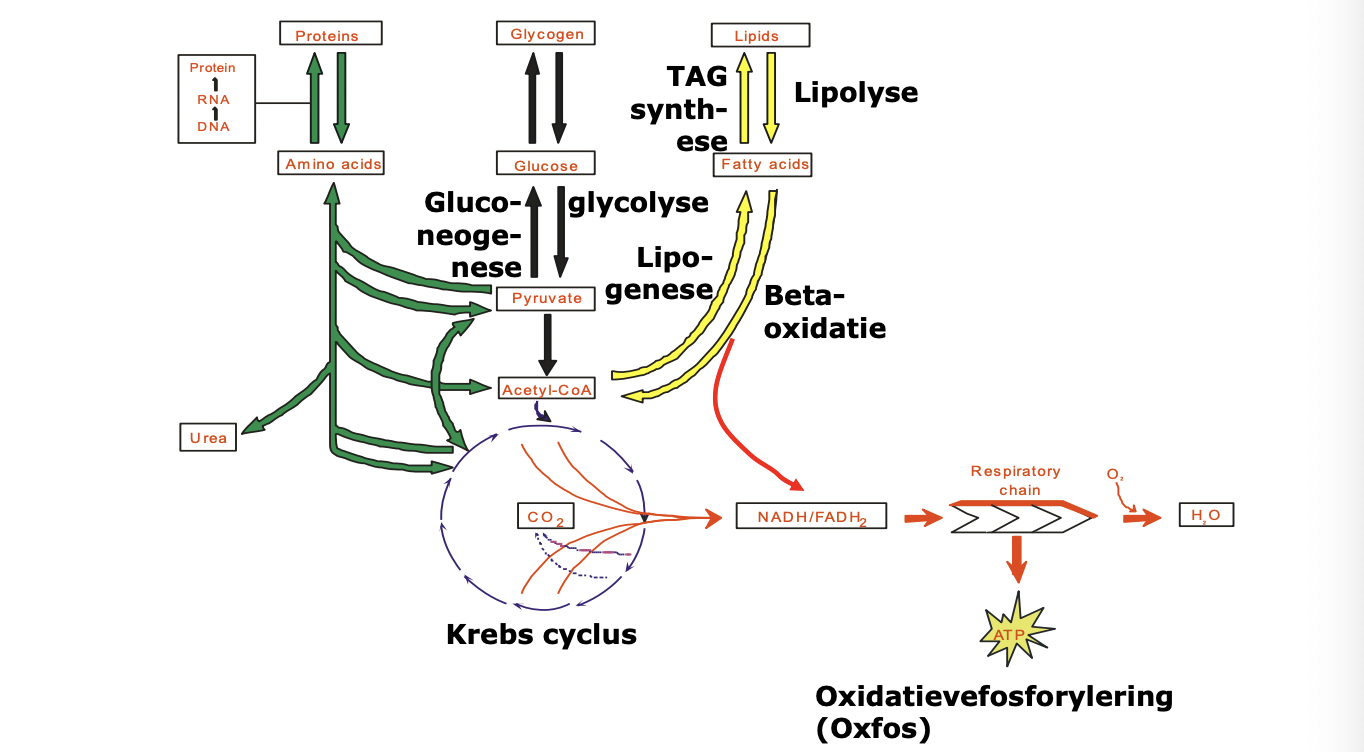
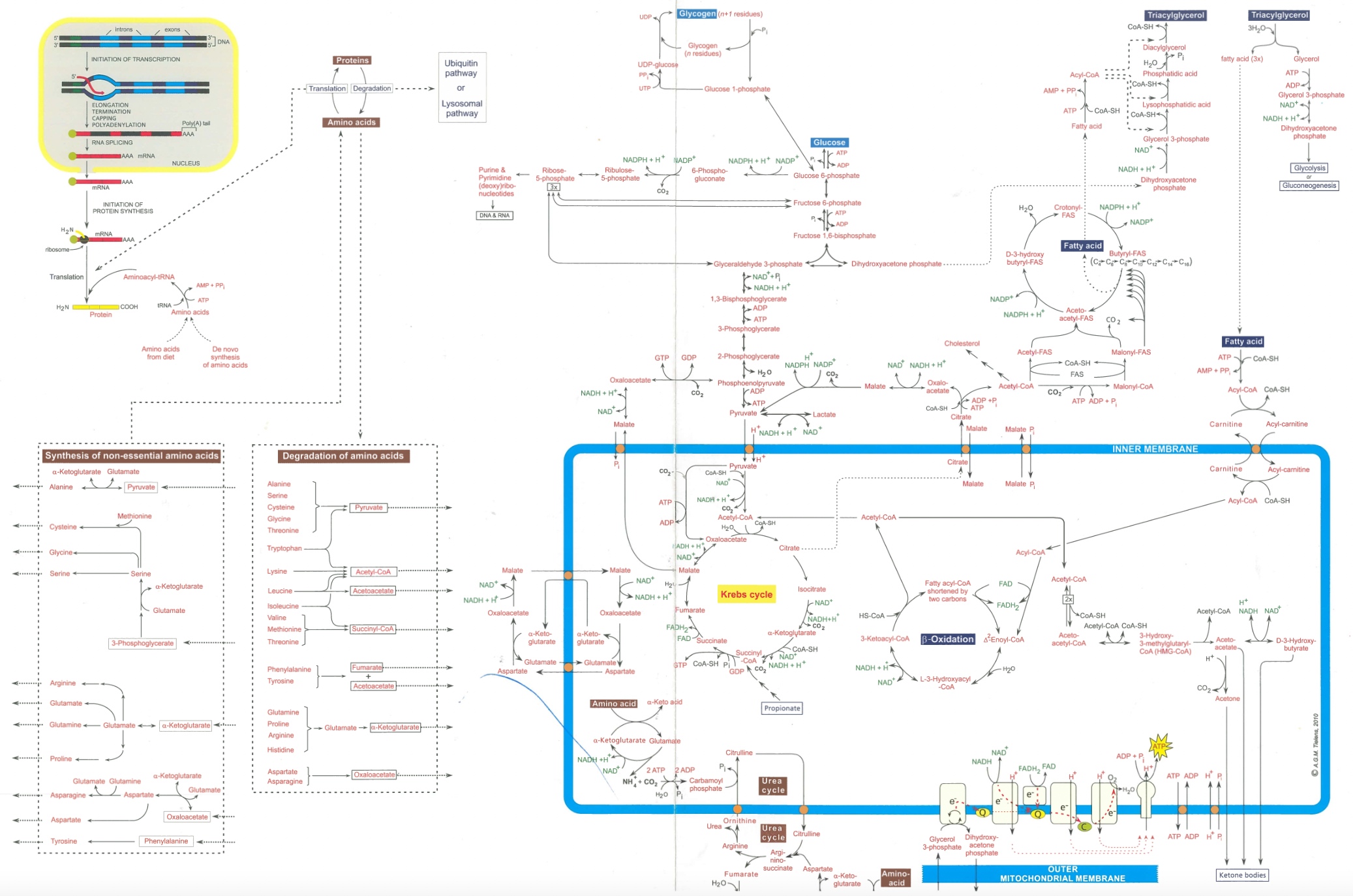
cell can make E (ATP) via dif routes - from glucose, FA or AA
breakdown of glucose (glycolysis)
glucose in cytoplasm → 2x pyruvate + 2x ATP
pyruvate transported to mitochondria and irreversibly converted by pyruvate dehydrogenase (PDH)→ acetyl-CoA
breakdown of FA (β-oxidation)
FA transported from cytoplasm to mitochondria and via β-oxidation broken down into multiple acetyl-CoA
in Kreb’s cycle 1x acetyl-CoA broken down into 2x CO2 + e- acceptors: 3x NAD+ & 1x FAD (can both accept 2 e-)
they give their e- away in e- transport chain to build up H+ gradient - used for ATP formation
anabolism
formation of glucose (gluconeogenesis)
(in mitochondrium) pyruvate → oxaloacetate
oxaloacetate transported to cytosol (via malate) and converted into phosphoenolpyruvate (PEP)
PEP then follows glycolysis backwards til → glucose (in liver) or glucose 6-phosphate (in other cells)
formation of FA (lipogenesis) & TAG synthesis
acetyl-CoA transported from mitochondrium (via citrate) to cytosol and converted → malonyl-CoA
both acetyl-CoA & malonyl-CoA put onto fatty acid synthase (FAS)
multiple cycles til chain is 16C long, then released from FAS
the FA can then be connected to a glycerol phosphate and w 2 more FA form triacylglycerol (TAG)
buildup and break down of glycogen
once activated via phosphorylation, glucose can go into different pathways
1) glycolysis
2) pentose phosphate pathway: glucose → ribose-5-P
used to make nucleotides (for DNA & RNA) & NADPH (reducing power for biosynthesis)
3) glucose-6-P can be built into glycogen for storage
glucose-6-P → glucose-1-P
glycogen synthase adds glucose-1-P to the growing chain
branching enzymes cause the branch formation in the glycogen molecule → fast break down & buildup glycogen
glycogen is broken down into glucose-1-P by glycogen phosphorylase
branches broken by debranching enzymes (bc gly phosphorylase can’t)
→ some free glucose released - needs to be activated into glucose-6-P before it can be broken down via glycolysis
glucose-1-P can then be converted → glucose-6-P
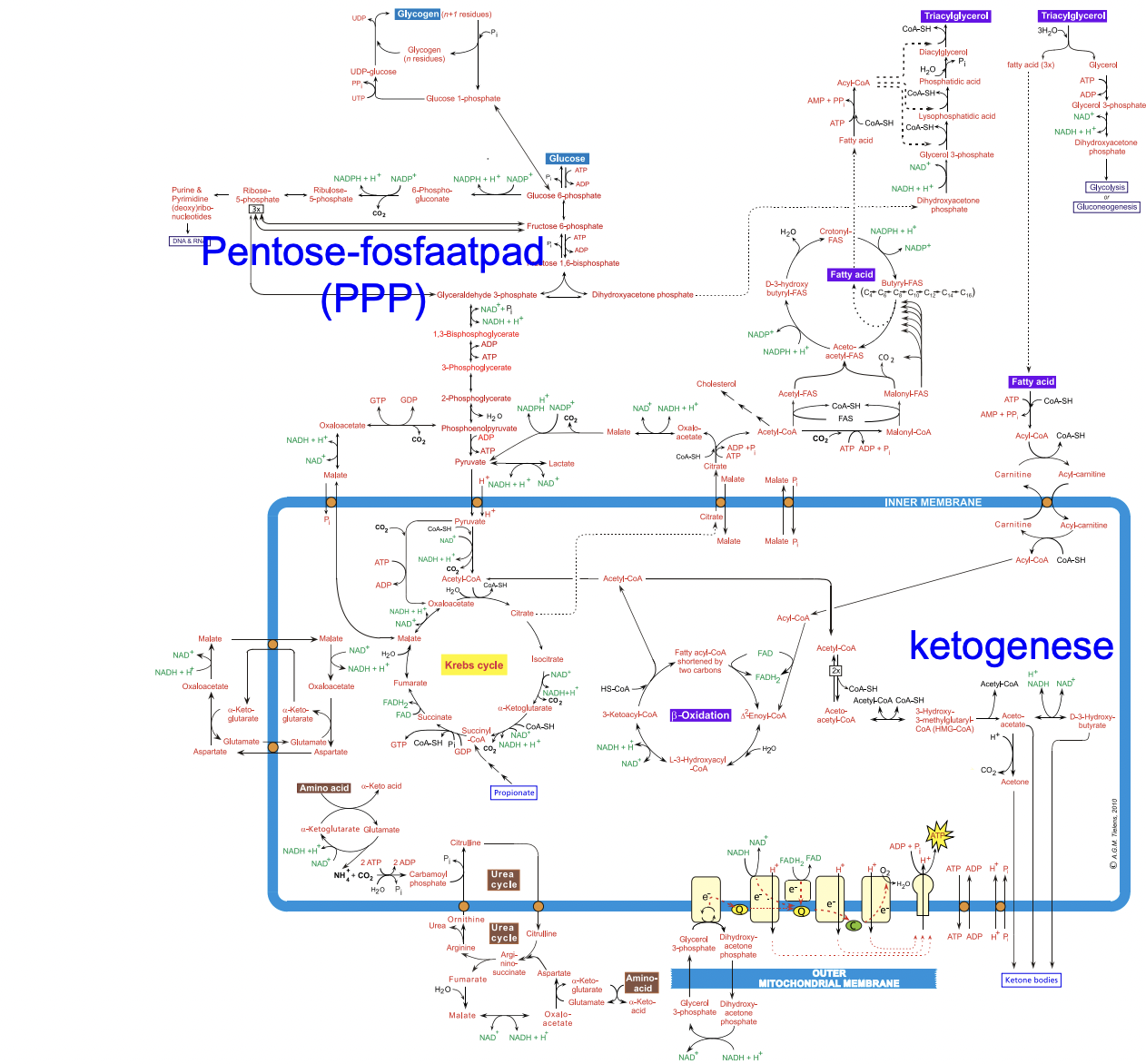
0.3 metabolic map
0.4 summary
rising blood glucose → insulin released → lowers blood glucose
by stimulating glycolysis & synthesis of glycogen, FA, p
has inhibitory effect on lipolysis in adipocytes
dropping blood glucose → glucagon released → raises blood glucose
by stimulating breakdown TAG and glycogen & stimulates gluconeogenesis
has inhibitory effects on glycolysis in liver
1. glucostasis on cellular level
1.1 introduction: cell types & GLUT
every organ has a different role in maintaining stable blood glucose levels
endocrine pancreas: β-cells secrete insulin, α-cells secrete glucagon
liver: store glycogen (oiv insulin) or release glu into blood (oiv glucagon and adrenaline)
muscle: store and use glycogen (oiv insulin) - no glucagon rec
adipocytes: take in glucose (oiv insulin) & convert to glycerol-3-P to store FA by making TAG. TAG breakdown (oiv glucagon) → FA release as alt E source so brain can use glucose
neurons: brain only uses glucose as E source
exceptional circ (bv prolonged fasting) → partial switch to ketone bodies as E source - uptake depends on conc in blood NOT hormonally regulated
use of glucose (or ketone bodies) NOT hormonally regulated
glucose taken into cell by GLUT - there’s dif isotypes
GLUT1: standard GLUT, normal affinity, in every cell.
ensures basal & continuous uptake → cell always enough E
GLUT2: low affinity → uptake only if (relatively) high blood glucose
in pancreatic β-cells (measure blood glucose here to adjust their insulin secretion) & liver cells
GLUT3: high affinity → uptake even is (relatively) low blood glucose - daarom in brain cells (bc glucose only E source)
GLUT4: normal affinity, only muscle & adipocytes. insulin-regulated!
high insulin → incr GLUT4 in memb → cell takes in more glu
insulin conc dcr → GLUT4 taken up by cell (wait in vesicle til insulin rec triggered again)
1.2 pancreas
pancreas is blood glucose meter of the body → hormone release adjustment
pancreatic β-cells have GLUT2 → can incr insulin (if blood glu high)
insulin secretion is directly regulated by blood glucose level
glucose enters cell → metabolized via glycolysis → ATP
incr ATP/ADP ratio → K+ channel closes
dcr K+ efflux → change memb potential → opens Ca2+ channel
Ca2+ influx → granules w insulin fuse to plasma memb → insulin into blood
1.3 muscle
insulin (minorly) & adrenaline affect glucose metabolism in muscle cells
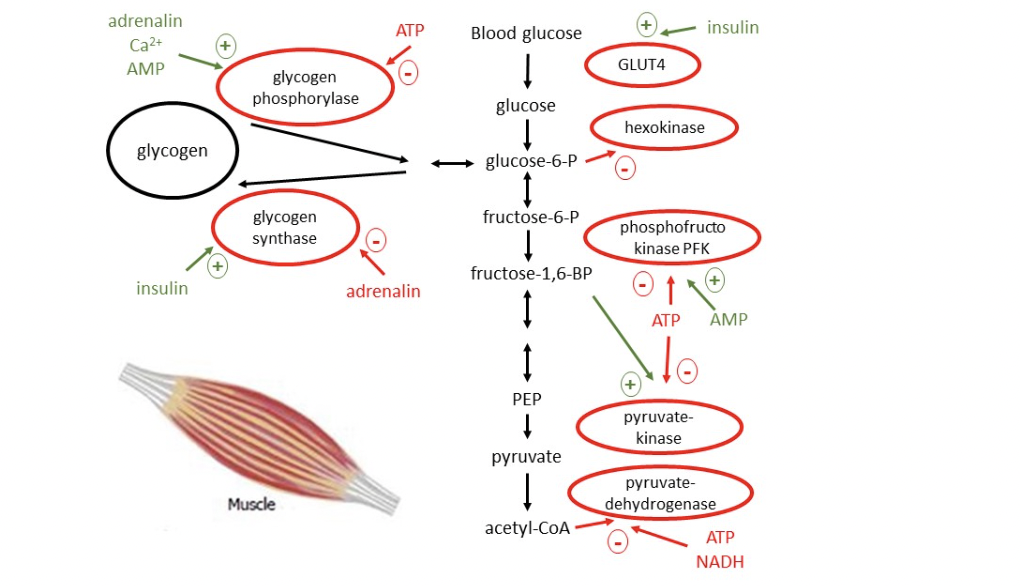
1.3.1 glycolysis & gluconeogenesis
barely any enzymes for gluconeogenesis in muscle → almost no gluconeogenesis
glycolysis regulated to meet ATP needs - 3 control sites:
main 1) phosphofructokinase (PFK): turns fructose-6-P → fructose 1,6-bisP
ATP inhibits PFK, AMP reverses inhibition → adjusts speed to E consumption
2) hexokinase: glucose → glucose-6-P (1st step glycolysis)
PFK inhibition → glucose-6-P accumulates & inhibits hexokinase → dcr glucose into glycolysis
3) pyruvate kinase (PK): dephos of PEP → pyruvate (last step)
ATP inhibits PK
1.3.2 pyruvate dehydrogenase (PDH)
PDH converts pyruvate → acetyl-CoA, links glycolysis to Krebs cycle
important bc acetyl-CoA can’t be converted back into glucose!
PDH regulated in 2 main ways
1) allosteric interaction of acetyl-CoA & NADH
PDH is inhibited by its products: acetyl-CoA, NADH & ATP
when high conc, they form a signal that E needs are met and no more glucose has to be broken down
2) phosphorylation: PDH kinase phosphorylates PDH which inactivates it
muscle cell at rest: ATP/ADP ratio high → directly inhibits PDH + stims PDH kinase →phosphorylates PDH so inactive PDH
muscle cell contraction: ATP/ADP ratio drops (ATP used) & glycogen/glucose converted to pyruvate → ADP + pyruvate inhibit PDH kinase → stimulate PDH
also phosphatase (stimulated by Ca2+ which also initiates contraction) is activated to dephosphorylate PDH
1.3.3 glycogen metabolism
when muscle cell needs ATP → glycogen phosphorylase activation, so glycogen can be broken down → glucose-6-P → ATP
contraction → incr ATP → incr AMP → stim glycogen phosphorylase
before AMP can stimulate it, it has to be activated by phosphorylase kinase which must be activated in 2 ways:
Ca2+ binds to camodulin subunits of phosphorylase kinase
phosphorylation of it by PKA
PKA is stimulated by adrenalin
when insulin binds to muscle cell it has 2 main effects
1) more GLUT4 put into membrane → more glucose in
2) activation of protein phosphatase (PP1)
inactivates phosphorylase kinase & glycogen phosphorylase → inhibition glycogen breakdown
activates glycogen synthase → stims glycogen synthesis
adrenaline has a similar double-sides effect by activating PKA which:
1) activates glycogen phosphorylase → stims glycogen breakdown
2) inactivates glycogen synthase → inhibits glycogen breakdown
1.4 liver
ATP & Ca2+ fluctuates less than in muscles cells so less of an effect
instead dif molecules with reg effects on the various enzymes
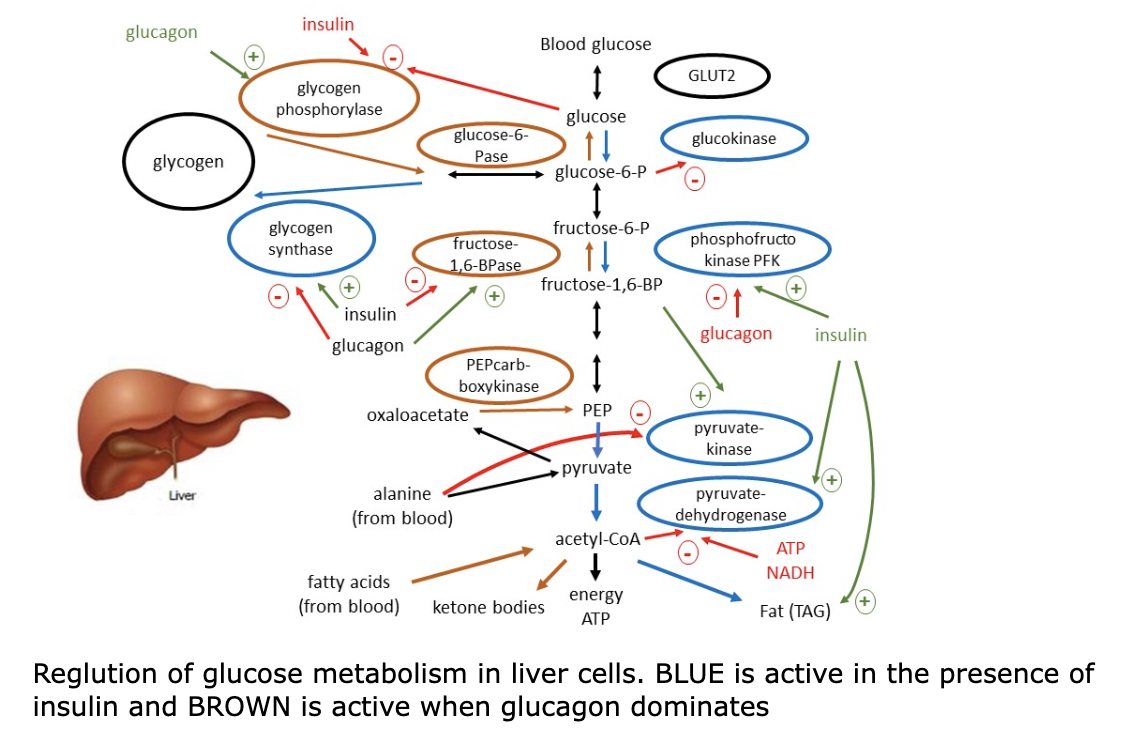
1.4.1 glycolysis
glycolysis speed hormonally regulated (as response to blood glucose) by insulin & glucagon at 2? regulation points:
main 1) phosphofructokinase (PFK): turns fructose-6-P → fructose 1,6-bisP
ATP & citrate inhibit PFK
2) hexokinase: glucose → glucose-6-P (1st step glycolysis)
PFK inhibition → glucose-6-P accumulates & inhibits hexokinase → dcr glucose into glycolysis
3) pyruvate kinase (PK): dephos of PEP → pyruvate (last step)
ATP & alanine inhibit PK
1.4.2 gluconeogenesis
1.4.3 pyruvate dehydrogenase
1.4.4 glycogen metabolism
1.4.5 ketogenesis
1.5 adipose tissue
1.6 questions
Kennis en inzicht verkrijgen in:
o (regulering van) het glucose-transport (GLUT1, 2, 3 en 4),
o de functie van de glycolyse in verschillende celtypen, o de hiërarchie van regelmechanismen in de glycolyse,
o de novo synthese van glucose, i.e. de gluconeogenese (substraten, specifieke enzymen, cellulaire en sub- cellulaire lokalisatie),
o (regulering van) het glycogeenmetabolisme,
o de werking van de pancreas-hormonen [insuline/glucagon] en de bijnier-hormonen [(nor)-adrenaline]
o de secretie van insuline door een β-cel in de pancreas
Na afloop van dit thema kun je:
o Precies de regel-punten en -mechanismen van de glycolyse, gluconeogenese, glycogeen-afbraak en glycogeen-opbouw aangeven,
o Het begrip glucostase uitleggen,
o Globaal uitleggen hoe het (koolhydraat-)metabolisme hormonaal wordt aangestuurd,
o De verschillende functies van glycogeen in spier- en lever-weefsel uitleggen.