Ionic bonding
Why do atoms bond
we can work out how many electrons are the in outer shell of atoms by looking at its group ( that is how many ). However, the noble gases have a full outer shell so they are very unreactive. This is because atoms react so that they gain a more stable arrangement of electrons, they want to gain a full outer energy level.
Ionic bonding
Ionic substance will always be compounds and never metals because they must contain a metal and a non-metal.
Atom: - neutral, has an equal number of protons + neutrons
Ions: - formed from n atoms which have lost/gained electrons
- Charged, because the number of electrons is no longer equal to number of protons
- full outer shell
Elements in group 4 rarely from ionic bond
GROUP 1
Group 1 will lose it’s 1 electron in it’s outer shell ( this requires less energy than gaining 7 )
This will make the charge +1 as there is now 1 more proton than electron
GROUP 2
Group 2 will lose the 2 electrons in it’s outer shell ( this requires less energy than gaining 6 )
This will make the charge +2
GROUP 3
Group 3 will lose the 3 electrons in its outer shell
This will make the charge +3
GROUP 5
Group 5 will gain 3 electrons as thats easier than losing 5
This will make the charge -3( 3 more electrons than protons )
GROUP 6
Group 6 will gain 2 electrons
This will make the charge -2
GROUP 7
Group 7 will gain 1 electron
This will make the charge -1
If you need to know the charge of a transition metal this will be given - sometimes in Roman numerals
When writing the formula of an ion, write the element symbol and then write the charge in the top right hand corner.
There are some multi- atom ions that you need to learn:
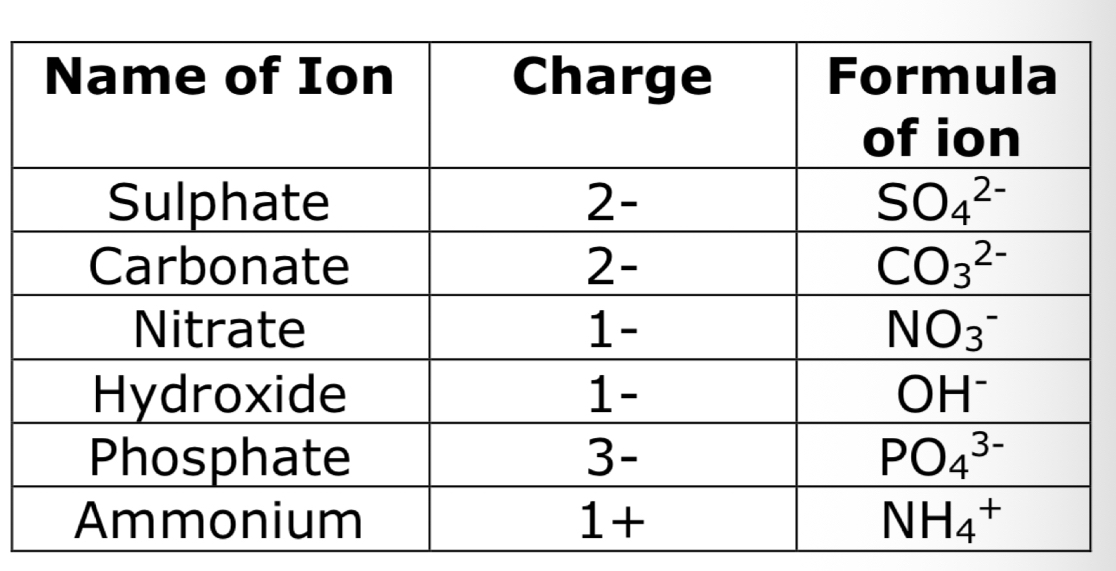
Students sometimes get mixed up between symbols, formulae and equations
Symbol for the element hydrogen - H
What is the formula of a hydrogen molecule - H*2 ( subscript small 2 )
What is the formula of water - H*2O
The formula shows the ratio
Overall, an ionic compound is neutral and the formula must reflect this. To do this, find the charge of each ion and swap them around ( like we did with valency ).
In an ionic compound the metal atoms have lost electrons to become positively charged ions and the nonmetals have gained electrons to become negatively charged. The strong electrostatic forces of attraction between oppositely charged ions ( SEFABOCI )is the ionic bond which holds the ion together.
STRUCTURE
The structure of an ionic compound is described as a giant regular lattice. The strong electrostatic forces of attraction between oppositely charged ions ( SEFABOCI ) act in all directions in the lattice.
Summary
Bond formation - electrons in outer energy level of metal are transferred to non metal
Bond - strong electrostatic forces attraction between oppositely charged ions
Structure - giant regular lattice