Half Life and Activity
Definition of Half-Life:
The half-life of a radioactive substance is the time required for half of the radioactive atoms in a sample to decay into a different element or isotope.
For any given radioactive isotope, the half-life is a constant value that does not change with the amount of substance present or environmental conditions.
Equation(s):
N = No e-λt
With:
(N) = Undecayed atoms remaining
(No) = Original amount of undecayed atoms
(λ) = Decay constant
(t) = Duration decay is measured (s)
N = No / 2x
(x) = number of Half Lives passed
Definition of Activity:
Activity refers to the rate at which a radioactive substance undergoes decay.
It is measured in decays per second, commonly expressed in units such as becquerels (Bq), where 1 Bq is equal to one decay per second.
Activity is directly proportional to the number of radioactive atoms present in a sample; as the number of undecayed atoms decreases over time, the activity of the substance also diminishes.
Equation(s):
A = Ao e-λt
With:
(A) = number of decays detected per second
(Ao) = Original amount of undecayed atoms
(λ) = Decay constant
(t) = Duration decay is measured (s)
A = Ao / 2x
(x) = number of Half Lives passed
Equations Linking Activity and Half Life
A = λN
A = ΔN / Δt
∴
ΔN / Δt = - λN
Exponential Decay Graphs
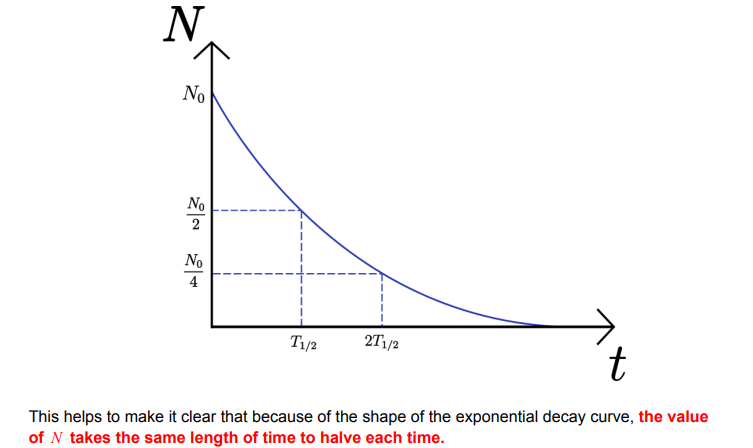
Derivation of λ = ln2 / T½
1. The Law of Radioactive Decay
The number of undecayed nuclei, N, at any time t is given by:
N (t) = N0 e−λt
2. Definition of Half-Life
The half-life (T½ ) is the time required for half of the radioactive nuclei to decay:
N (T½) = N0 / 2
3. Substituting Half-Life
Using the decay law:
N0 / 2 = N0 e−λT½
Cancel N0 (assuming N0 ≠ 0):
1 / 2 = e−λT½
4. Solve for λT½
Take the natural logarithm (ln) on both sides:
ln(1 / 2)= −λ T½
Simplify ln (1 / 2) using logarithmic properties ( ln ( a / b ) = ln ( a ) − ln ( b ) ):
ln (1 / 2) = ln (1) − ln (2) = 0 −ln (2) = − ln (2)
So:
− ln (2) = − λT½
Cancel the negatives:
λT½ = ln (2)
Finally, solve for λ:
λ = ln (2) / T½