BIOLOGY 9
1st Quarter Examination Notes
INORGANIC COMPOUNDS - WATER
WATER
Molecule that supports all life.
Life on Earth began in water and evolved for 3 billion years before spreading onto land.
Three quarters of Earth’s surface is covered by water.
The only common substance on earth to exist in the natural environment in all three physical states of matter.
POLAR COVALENT BONDS IN WATER MOLECULE
Oxygen is more Electronegative than Hydrogen, making electrons spend more time with Oxygen. These are Polar Covalent Bonds.
Polar Molecule : The unequal sharing of Electrons and waters v-like shape.
OPPOSITES ATTRACTS : The partially positive (+) H attracts partially negative (-) O, and two molecules are held together by a Hydrogen Bond.
When in Liquid form, its hydrogen bonds are very fragile, each only about 1/20 as strong as a covalent bond.
Electronegativity : Attraction of an atom for the Electrons of a Covalent bond
Why can the Central Water Molecule hydrogen bond to four other water molecules but not three or five?
Because of it’s two polar covalent bonds, a water has four regions of partial charge.
It has 2 positive Regions (The H atoms)
and 2 negative Regions (the O atoms)
Water is an Excellent Solvent
The Polarity of water molecule and the ability to form Hydrogen bonds is what makes it an excellent solvent. (Meaning that many substances can dissolve in it.)
Hydrogen Bonds are much more stronger than Ionic bonds between 2 ions, so the solid dissolves while water molecules tug the ions apart and surround each one.
When a substance like NaCl dissolves, its component ion disperses uniformly among the molecules of a liquid and becomes a solute.
NaCl is called salt because aside from H+ and OH-, it releases other ions when it dissolves in water.
FOUR EMERGENT PROPERTIES
Cohesion of Water Molecules
Cohesion - A Phenomenon in where the Hydrogen Bonds hold the substance together.
Adhesion - The act of clinging of one substance to another.
Water Transport in plants are because of Cohesion and Adhesion.
Surface Tension - The measure of how difficult it is to stretch or break the surface of a liquid.
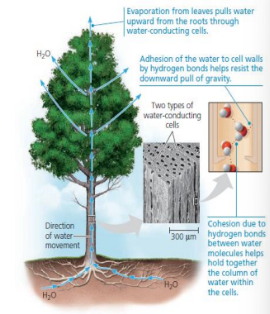
Moderation of Temperature by Water
Water is able to moderate air temperature through absorbing heat from air that is warmer then releasing the stored heat through air that is cooler.
Water is a Heat Bank because it can absorb/release large amounts of heat with only a slight change in its own temperature.
Temperature and Heat
Anything that moves has Kinetic Energy. (Kinetic Energy : The Energy of Motion.)
Thermal Energy : The kinetic Energy associated with the random movement of atoms or molecules.
Thermal Energy of a body of matter reflects the total kinetic energy, thus depends on the matter’s volume.
Temperature represents the average kinetic energy of molecules in a body of matter.
Heat : The Thermal Energy in transfer from one body of matter to another.
Calorie : The amount of heat it takes to raise the temp. of 1g of water by 1 degree Celsius. Also the amount of heat that 1g of water releases when it cools by 1 degree Celsius.
Kilocalorie (kcal) : 1000 Cal is the quantity of heat required to raise the temp. of 1kg of water by 1 degree Celsius.
Joule (J) : One Joule = 0.239 cal ; One calorie = 4.184 J
Water’s high specific heat
Specific Heat : The Specific Heat of a substance is defined as the amount of heat that must be absorbed or lost for 1g of that substance to change its temp. by 1 degree Celsius.
Water has a high specific heat; Ethyl Alcohol in alcoholic beverages has a specific heat of 0.6 cal.
What is its Relevance to life on Earth?
Large Body of water can absorb and store huge amounts of heat from the sun in the daytime and during summer while only warming up a few degrees. During the winter or nighttime, the cooling water can warm the air. This capability can moderate air temps. in costal areas.
Tends to stabilize ocean temperatures.
The water that covers most of earth keeps temp. fluctuations on land and water within limits that permit life.
Organisms are made primarily of water, they are better able to resist changes in their own temp.
Evaporative Cooling
Vaporization / Evaporation : Transformation from liquid to gas.
The speediest molecules can escape into the air, even at low temp.
Evaporation occurs at any temperature.
If a liquid is heated, the average kinetic energy of molecules increases and the liquid evaporates more rapidly.
Heat of Vaporization
Is the Quantity of heat a liquid must absorb for 1g of it to be converted from liquid to gas.
Has high heat of Vaporization, relative to most other liquids.
About 580 Cal of heat is needed to evaporate 1g of water at 25 degrees Celsius.
Another Emergent property resulting from the strength of its hydrogen bonds. (which must be broken before the molecules can exit from the liquid through water vapor.)
The high amount of energy required to vaporize water has a wide range of effects.
It helps moderate Earth’s Climate.
Accounts for the Severity of steam burns.
Evaporative Cooling
As liquid evaporates, the surface of the liquid that remains behind cools down.
Occurs because of the “hottest” molecules, those with greatest kinetic energy are most likely to leave as gas.
Contributes to the stability of temp. in lakes and ponds
Provides mechanism that prevents terrestrial organisms from overheating.
Floating Ice on Liquid Water
Ice floats on liquid water because water is less dense in a solid form. Water also expands when solidified. This is caused by Hydrogen Bonding.
At temperatures above 4 degrees Celsius, water expands as it warms and contracting as it cools. At temps 4 to 0 degrees Celsius, Water begins to freeze because more and more of its molecules are moving slowly so it can break hydrogen bonds. at 0 degree Celsius, molecules become locked into a crystalline lattice.
Water reaches its greatest density at 4 degree Celsius.
This factor is important because if ice sank, then eventually all ponds, lakes and even oceans would freeze, making life impossible on Earth.
ACID AND BASES
ACIDIC AND BASIC CONDITIONS AFFECT LIVING ORGANISMS
Hydrogen ion - Proton with a charge of 1+
Hydroxide Ion - Water molecule that has lost a proton (now with a charge of 1-)
The Proton Binds to the Other water molecule, turning it into a hydronium ion. (H3O+)
A reversable reaction that reaches a state of dynamic equilibrium (balanced) when water molecules dissociate (separate) at the same time rate they are being reformed from H+ and OH-.
Changes in the concentrations of the substances H+ and OH- can affect a cells proteins and other complex molecules.
Their concentrations are equal in water (pure) but adding different types of solutes disrupts this balance.
pH scale : is used to measure the acidity/basicity of a substance/solution. (also means the potential of hydrogen).
pH 7 : neutral
pH <7: acid
pH >7: base/basic
What would cause an aqueous solution to have an imbalance in H+ and OH- concentrations?
Addition of other substance
When acids dissolve in water, they donate additional H+ to the solution.
ACID
Substance that increases the hydrogen ion concentration of a solution.
Ex. HCl → H+ + Cl-
When HCl is added to water, hydrogen ions dissociate from chloride ions (to lessen Acidity)
BASE
Substance that leaves the concentration of hydrogen ion in a solution.
Some reduce H+ concentration by accepting hydrogen ions and indirectly by dissociating to form hydroxide ions.
Neutral : Substance that is neither an acid nor base
Strong Acid : Substance that Ionizes (Completely) in an aqueous solution.
That is because the bond between the hydrogen ion (H+) and the rest of the acid molecule is very weak.
Ex. Hydrochloric Acid
Strong Base : Substance that dissociate into its ions in a aqueous solution, producing a high concentration of hydroxide ions (OH-)
Weak base : Substance that only partially dissociate into its ions in an aqueous solution.
Ex. Ammonia
NH3 + H+ → NH4+ (double arrow* indicate that the binding and release of hydrogen ions are reversable)
Weak Acid : Acids that reversibly release and accept back hydrogen ions (H+)
Ex. Carbonic Acid
H2CO3 → HCO3- + H+ (*double arrow*)
pH SCALE
In any aqueous solution at 25 degrees Celsius, the product if the H+ and OH- concentrations is constant at 10-14
Brackets indicate molar concentrations
an acid not only adds hydrogen ions to a solution, but also removes hydroxide ions because of the tendency for H+ to combine with OH-, forming water.
a base increases OH- concentrations but also reduces the H+ concentrations by the forming of water.
Buffers : substances that allows biological fluids to maintain a constant pH despite the addition of acids or bases
Accepts hydrogen ions from the solution when they are in excess and donating hydrogen ions to the solution when they are depleted.
weak acid and corresponding base.
Ex. H2CO3 → HCO3- + H+
Acidification : A threat to our ocean
when CO2 dissolves in seawater, it reacts with water to form Carbonic Acid, lowering the pH of the Ocean.
H2O + CO2 → H2CO3
How?
: As seawater acidifies, the extra H+ combine with the Carbonic Acid to form bicarbonate ions, reducing carbonate ion concentration. In short, acidification kills the corals.
pH decreases as H+ concentration increases.
Each pH unit represents a tenfold difference in H+ and OH- concentration
Ex. pH of 3 and 6 is 1000 times more acidic (10 × 10 × 10)
ORGANIC COMPOUNDS
FOUR MOLECULES OF LIFE :
Proteins, carbohydrates, lipids, and nucleic acid
The most important large molecules found in all living things.
Carbohydrates, Proteins, and Nucleic acid are huge and therefore called macromolecules.
Large biological molecules exhibit unique emergent properties arising from the orderly arrangement of their atoms.
POLYMERS
A long molecule consisting of many similar or identical building blocks linked by covalent bonds. (Like a train consists of a chain of cars.)
Large Carbohydrates, proteins, and nucleic acids are polymers.
from Greek polys = many and meros = part
The repeating units tat act as the building blocks of a polymer are called monomers which are smaller molecules. (From the Greek monos = single).
THE SYNTHESIS AND BREAKDOWN OF POLYMERS
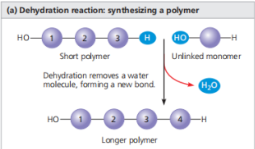
Enzymes - Specialized macromolecules that speed up chemical reactions.
Dehydration Reaction - A reaction in which two molecules are covalently bonded to each other with the loss of a water molecule. The picture above illustrates how the Dehydration reaction happens when connecting monomers.
Polymers are disassembled to monomers through the process of hydrolysis
Hydrolysis : Opposite of Dehydration ; addition of water molecule to break a bond in the polymer.
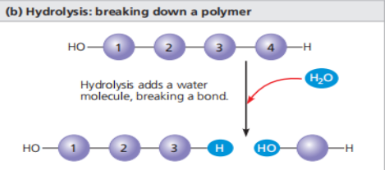
Example of hydrolysis - digestion. The organic material i our food is in the form of polymers, which is too large to enter our cells. In the digestive tract, enzymes attack the polymers, which speeds up hydrolysis. Then the released monomers are then absorbed into the bloodstream for distribution to all cells in the body. Then the cells use the dehydration reactions to assemble the monomers into a new different polymers that perform specific functions required by the cell. ( can also be involved in the formation and breakdown of molecules that are not polymers, like some lipids.)
CARBOHYDRATES
Include sugars and polymers of sugars.
Simplest carbohydrates = monosaccharides / simple sugars.
these are monomers from which more complex carbohydrates are built.
Disaccharides = double sugars, which has two monosaccharides joined by a covalent bond.
Polysaccharides = Carbohydrates macromolecules, which is composed of many sugar building blocks.