3.15: Hess's Law
Hess’s Law: The enthalpy change of each reaction in a series, adds up to the enthalpy change of the overall reaction.
When solving for enthalpy changes, if trying to find the enthalpy of a reactant, but the next step shows the reactant in the products side, reverse the sign of the enthalpy, and vice-versa. e.g.
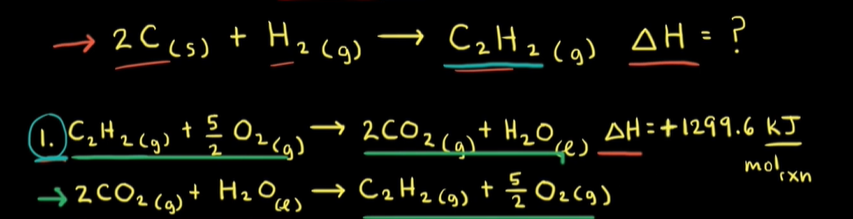
Then, reactants or products need to be scaled according to the number of moles they have.
Finally, add all of the values together