Chemistry: Ionic Bonding
Background Information:
Before understanding ionic compounds & bondings, one has to understand some of the simple trends that take place in the periodic table and also the different types of atoms.
\ 1: We must understand what are metals and non-metals–in a chemical sense–and how do their properties affect the way they bond with other atoms. First, let’s discuss metals. Metals are the atoms located on the %%left side of the periodic table%%. Metals tend to be solid at room temperature, and they are also good conductors of heat & electricity. However, what’s more relevant to know is that ^^metals like to give away their electrons^^. Once they give away their electrons, they become positively charged ions–which are known cations. On the other hand, we have non-metals which are located on the %%right side of the periodic table%%–however, there also exists some metalloids that are located in the right side of the periodic table. Non-metals are mostly gases at room temperature, and they are poor conductors of electricity. Unlike metals, non-metals like to ^^gain/seize electrons^^. Once non-metals receive electrons, they become negatively charged ions–which are known as anions.
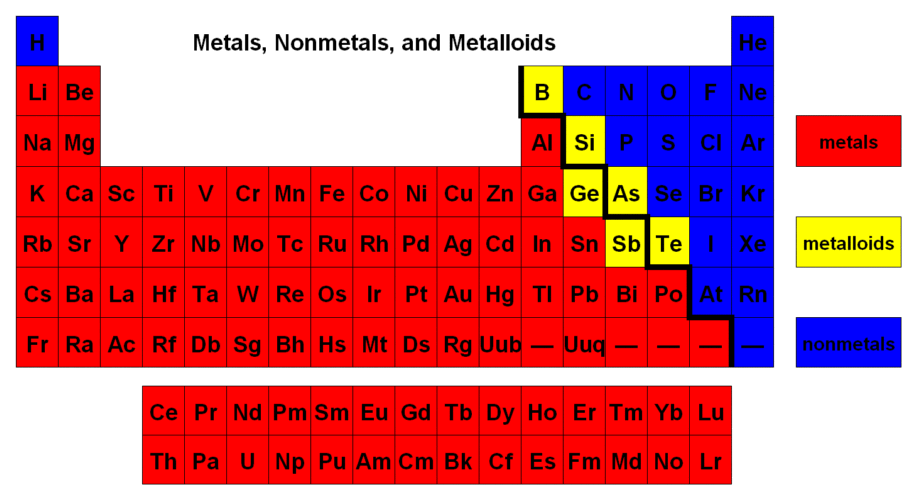
2: We mentioned that metals like to give away electrons, and non-metals like to seize electrons. This happens due to a periodic trend called electronegativity. Electronegativity is simply the tendency of an atom to seize/attract electrons in a chemical bond–you can think of it as “electron-greediness”.

Ionic Bonds & Compounds
Now that we have covered background information, it’s time to talk about ionic bonds & compounds.
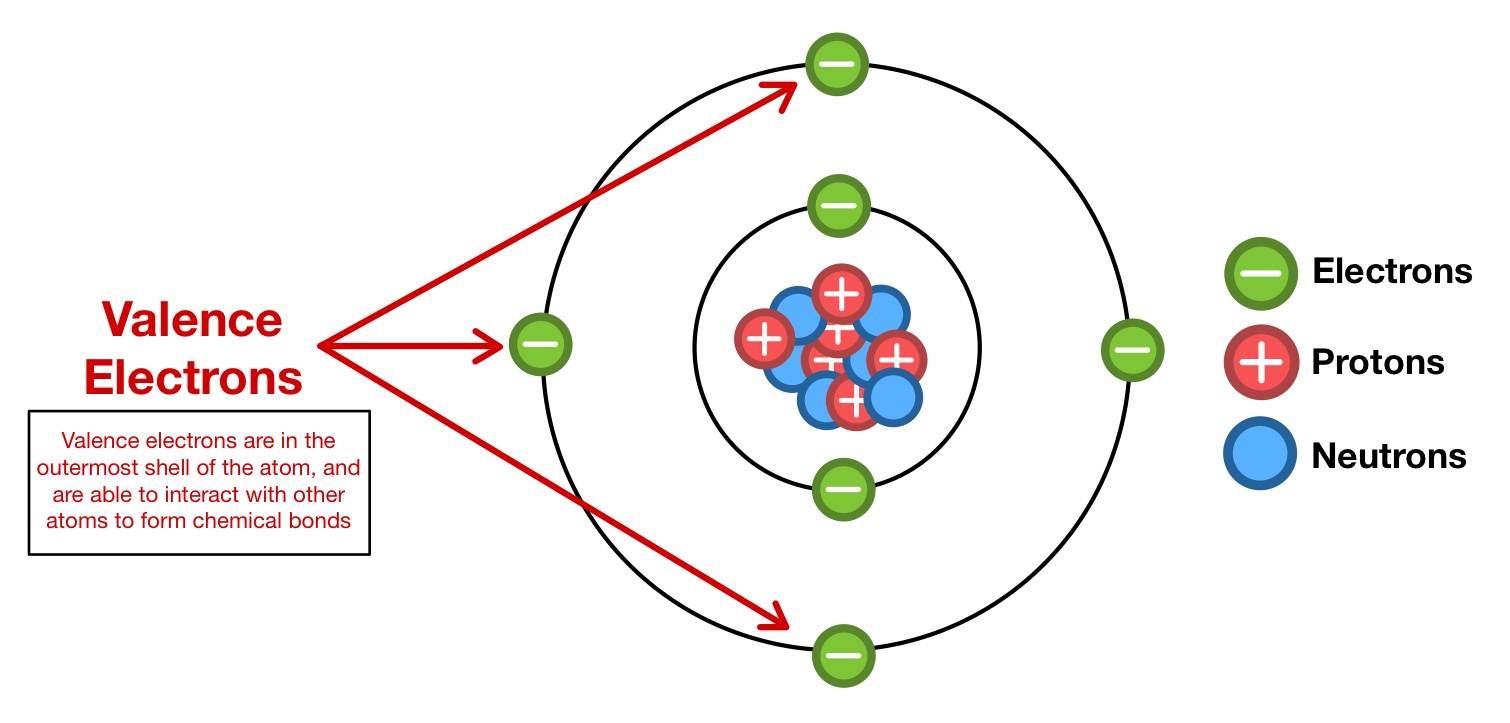
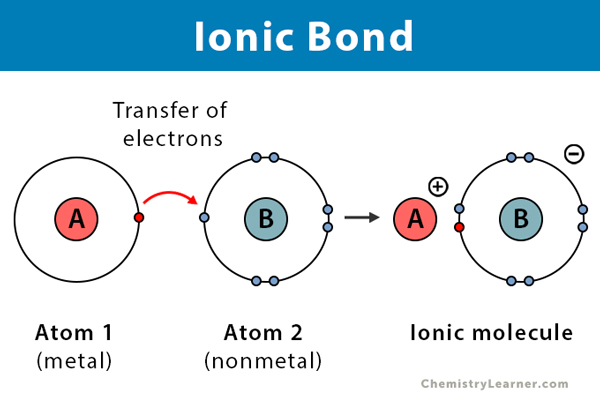
\ Ionic bonds are ==bonds that exist between a metal and a non-metal==. In ionic bonding, electrons are transferred from the metal to the non-metal. In other words, the metal gives away its electrons to the non-metal which is due to their respective electronegativities. Once this happens, the metal ions becomes a positively charged ion, and the non-metal becomes a negatively charged ion. The electrons that are transferred are the valence electrons of the metal. Valence electrons are the electrons located in the outermost energy level.
Examples
Example 1:
NaCl (sodium chloride)
Na (sodium) is the metal and has 1 valence electron
Cl (chlorine) is the non-metal and has 7 valence electrons
In this case, the sodium atom will give its 1 valence electron to the chlorine atom. Since sodium has only given 1 electron away, it will form . On the other hand, since chlorine gained only 1 electron, it will form Remember that electrons are negatively charged, that’s why when an atom gains electrons it charges decreases/becomes more negative–and the opposite is true.
Example 2:
MgS (magnesium sulfide)
Mg (magnesium) is the metal and has 2 valence electrons
S (sulfur) is the non-metal and has 6 valence electrons
In this case, the magnesium atom will give its 2 electrons to the sulfur atom. Therefore, magnesium will form a On the other hand, sulfur will recieve the 2 electrons and will form a
\ When the metal and non-metal form a bond, they form an ionic compound. Ionic compounds are compounds made of a metal and non-metal that are bonded by ionic bonding. Ionic compounds are electrically neutral–meaning, their overall charge is 0–because the positive charge of metal cancels the negative charge of the non-metal.
\ \ \ \ \ \