Organic Chemistry
Organic chem terminology
organic chemistry = chemistry of carbon compounds
enabled by carbon’s ability to form several strong covalent bonds with itself
a functional group is a certain atom or group of atoms that give the molecule they’re part of certain physical, chemical properties
organic compounds with the same functional group AND general formula but a different number of carbon atoms belong to a homologous series
the start of an organic compound’s name is dictated by the number of carbon atoms it contains. the first 4 four are:
meth-
eth-
prop-
but-
remember with the anagram My Elephant Prefers Bananas.
there are different formulae you might be asked to present an answer as.
general: composition of any member of a whole homologous series
alkAnes: CnH2n+2
alkEnes: CnH2n
displayed: shows the spatial arrangement of all the atoms and bonds. when you’re drawing these, check that every carbon has 4 lines coming off of it.
molecular: number of each atom in a molecule
structural: presents the structure as letters, showing only double/triple bonds
e.g: butene
general formula | CnH2n |
displayed formula | 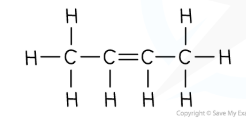 |
molecular formula | C4H8 |
structural formula | CH3CH2=CHCH3 |
Crude Oil
formed over millions of years as biomass was crushed by layers of sand and mud.
high pressure, temperature and lacking oxygen caused crude oil to form
crude oil is a mixture of different hydrocarbons of different sizes. hydrocarbons are compounds that contain ONLY carbon and hydrogen → you must say ‘only’ to get the mark
crude oil alone is not useful, but its fractions are. a fraction is a mixture of different hydrocarbons of a similar carbon chain length (and thus with similar properties)
we can seperate the fractions from crude oil via fractional distillation. this process is often a 6 marker.
crude oil is vaporised by heating to 350 degrees C and enters the fractionating column
crude oil vapours rise up the fractionating column, which has a negative temperature gradient (temp decreses as vapours move up)
the first fractions to condense are those with the longest chains = highest boiling points, e.g. bitumen
why? because longer chains have stronger intermolecular forces which take more energy to overcome
the last fractions to condense are those with shortest chains = lowest boiling points, e.g. liquid petroleum gas
the vapours of a certain fraction condense on a bubble tray at their boiling point, are collected and transported away for use
crude oil fractions are used as fuel + as feedstock for the petrochemical industry:
fraction - top → bottom of fractionating column | use |
liquified petroleum gas | domestic heating, cooking |
petrol | car fuel |
kerosene | airplane fuel |
diesel | diesel engines in cars, lorries. etc |
heavy fuel oil | ship fuel, power stations |
these are not all the fractions of crude oil, just the ones you need to know for the exam.
other products of the petrochemical industry, that use certain crude oil fractions, are solvents, lubricants, polymers and detergents
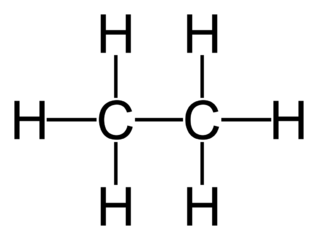
Alkanes
crude oil is formed primarily of alkanes
these are saturated hydrocarbons, meaning every possible bond is filled with hydrogen because they only have single bonds between carbon atoms
suffix is -ane
general formula: CnH2n+2
e.g: ethane, C2H6
properties of alkanes
as you change the length of the carbon chain, the properties of the alkane change
longer alkanes are:
less flammable (harder to light, burn with a smoky flame)
are more viscous (harder to pour, flow less easily)
less volatile (less likely to turn into a gas, because boiling points are higher due to stronger intermolecular forces)
vice versa for shorter alkanes.
reactions of alkanes
alkanes are generally unreactive but they do undergo combustion, can react with halogens in presence of light and can be cracked.
(complete) combustion:
alkane + oxygen → water + carbon dioxide gas
carbon and hydrogen are oxidised (because oxygen is gained)
often asked to balance these equations
if there isn’t enough oxygen to enable complete combustion of an alkane, unburnt hydrocarbons will be left over, causing environmental issues → see chemistry of the atmosphere notes. this is a problem with larger hydrocarbons e.g. octane (which incidentally is what petrol is mainly comprised of) because they need more oxygen to combust completely.
cracking:
shorter carbon chain hydrocarbons are in higher demand because they can be used as vehicle fuel
but crude oil is mostly long chains, which are less useful and of lower demand
so we crack excess long chain hydrocarbons down into smaller ones to meet demand
cracking produces a smaller alkane and also an alkene (usually ethene or propene)
when you’re writing cracking equations, make sure your reactant is an alkane and the products are a smaller alkane + an alkene. also ensure that the number of C and H atoms are the same on each side
there are 2 ways to crack molecules:
catalytic cracking
heating hydrocarbons to 470 to 550 degrees C → vaporises them
vapours pass over powdered aluminium oxide catalyst
covalent bonds in molecules broken down - thermal decomposition
steam/thermal cracking
higher temperatures than catalytic cracking
vapourised hydrocarbons mixed with steam and heated more → cracking
more alkenes and hydrogen formed due to higher temperature and pressure.
Alkenes
alkenes are unsaturated hydrocarbons, meaning not every bond is filled with a hydrogen atom because they have a double bond between 2 carbon atoms on their chain
suffix is -ene
functional group is C=C
general formula: CnH2n
e.g: ethene, C2H4
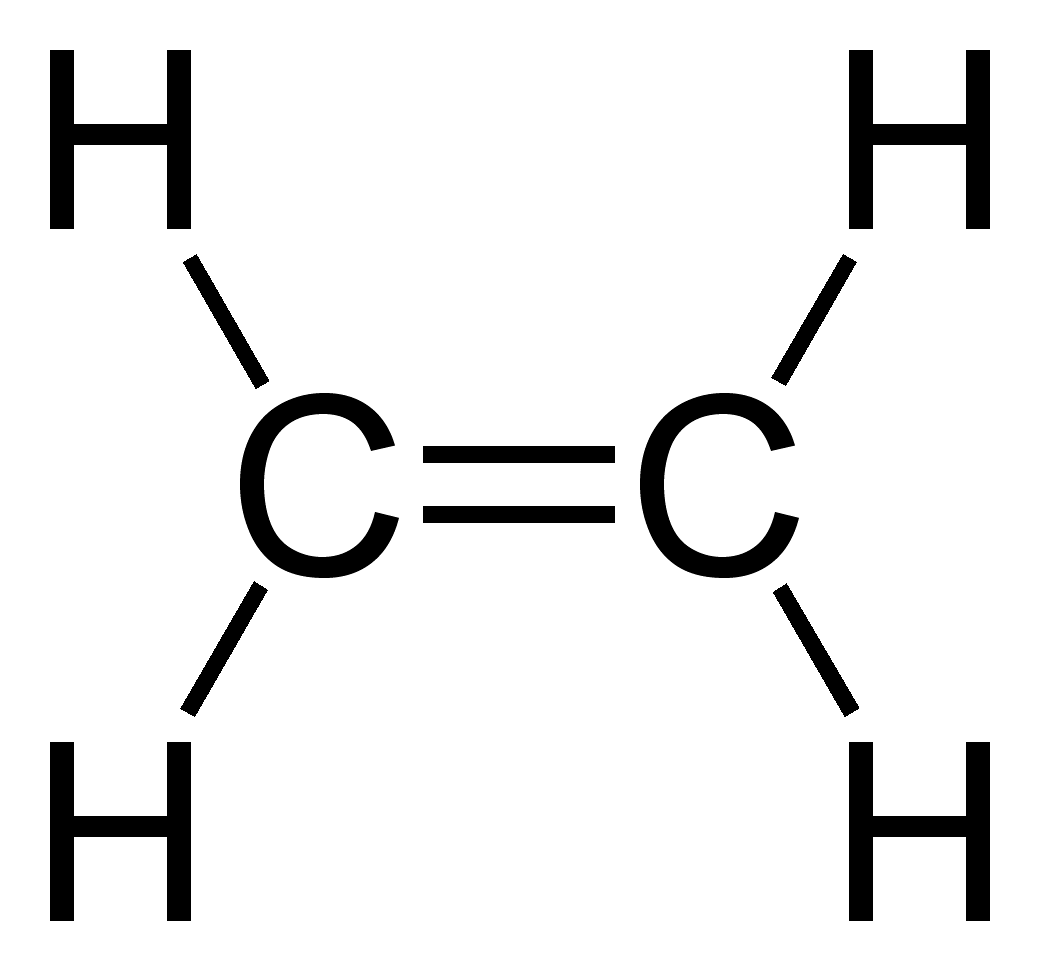
methene does not exist because there’s only 1 carbon atom; no double bond can be formed. so the 4th one you need to know is pentene.
reactions of alkenes
alkenes are very reactive because their C=C bond can open up to enable more bonds to be made with other atoms.
combustion:
alkenes are more susceptible to incomplete combustion than alkenes because of the higher carbon:hydrogen ratio
this produces smoky yellow flames
the possible products of incomplete combustion are CO (carbon monoxide) or C (as soot)
alkenes undergo addition reactions - where the simple molecule the alkene is reacting with is added across the C=C bond, making it into a C-C bond.
when you draw the display formulae for these reactions, you need to make sure you’re adding the new molecules where the C=C bond is being broken; not just anywhere in the molecule.
where the C=C bond is is denoted by a number in the middle of the name. e.g prop-2-ene has the C=C between the 2nd and 3rd C atoms.
with halogens (halogenation)
occurs at room temperature
reaction with bromine water used as a test for alkene - solution turns orange → colourless if it’s an alkene because the bromine gets added across C=C - no longer free in the solution

with hydrogen (hydrogenation)
nickel catalyst
at 150 degrees C
forms an alkane
e.g. ethEne + hydrogen → ethAne

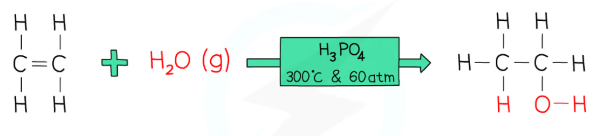
with water (hydration)
phosphoric acid catalyst
pressure of 60 atm
temperature of 300 degrees C - so the water is water vapour
forms an alcohol
reversible reaction
after the reaction, unreacted alkene is seperated by condensing the mixture because its boiling point is much lower
the left over water and the alcohol are seperated by fractional distillation
e.g: ethene + water vapour → ethanol
Alcohols
functional group OH
suffix -anol
general formula CnH2n+1OH
note that their molecular formula has the OH at the end - e.g. propanol = C3H7OH, NOT C3H8O. the OH is the functional group, so we don’t combine it with the other Hs.
alcohols are colourless, dissolve in water to form neutral solutions
first 4 are commonly used as fuels for lab equipment because they burn cleanly (no smoke) and without a strong odour. ethanol is used in car fuel
methanol and ethanol are often used as solvents because they can dissolve many substances water cannot (e.g. fats, oils) + the ones water can.
ethanol is the alcohol used in alcoholic drinks
producing ethanol
there are 2 ways to produce ethanol:
hydrating ethene. the ethene is gathered from cracking of long chain alkanes. for more detail on reaction see above
advantages | disadvantages |
fast, creates pure product | high tech equipment needed, which is expensive short term |
95% yield, other 5% is unreacted ethene which can be recycled | high cost to generate high temps+ pressures needed |
fermentation.
extract glucose form crops
add yeast to glucose; enzymes in the yeast act as a catalyst
ferment the glucose - 15-35 degrees C, absence of oxygen
glucose + enzymes → ethanol + carbon dioxide
batch process (stops and has to be started again) because once ethanol concentrated exceeds 15%, the yeast are killed off and have to be replenished
advantages | disadvantages |
sugars are a renewable source | very slow |
theoretically carbon neutral because the CO2 the plant took in to create glucose gets lets out again | ethanol produced is very impure so fractional distillation needed, which requires energy, so process not really carbon neutral |
reactions of alcohols
combustion: produce carbon dioxide and water; the alcohol is the fuel
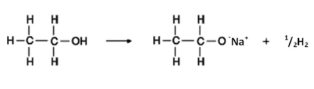
with sodium:
alcohol + sodium → sodium alkoxide + hydrogen
fizzing (H gas bubbles) and sodium decreases in size during the reaction
e.g. ethanol + sodium → sodium ethoxide + hydrogen
note that there’s no line between the O and the Na because they’re ionically bonded. The ½ in front of H2 is for balancing
with an oxidising agent:
oxidising agents induce oxidation in an alcohol
this can occur naturally (e.g. in the case of wine) or artificially (e.g. by using potassium dichromate)
redox reaction
alcohol + oxidising agent → carboxylic acid + water
e.g. ethanol + potassium dichromate → ethanoic acid + water

Carboxylic acids
functional group COOH
suffix -anoic
carboxylic acids are weak acids - they only partly ionise in water to produce weakly acidic solutions. this causes them to form aqueous solutions with high pHs of 3-7.
reactions of carboxylic acids
with water:
soluble in water, produce a weakly acidic solution
with carbonates:
carboxylic acid + carbonate → salt + water + carbon dioxide
like another other acid
e.g. propanoic acid + potassium carbonate → potassium propanoate + water + carbon dioxide
with bases:
like any other acid produce a salt and water
esterification
when a carboxylic acid reacts with an alcohol, an ester is formed. this is usually done in the presence of a concentrated sulfuric acid catalyst
ester functional group is R1-COO-R2 where R1 is the carboxylic acid and R2 the alcohol.
(glucose)are sweet smelling and oily, used in food flavouring or perfume. they’re volatile (vapourise easily)

Polymerisation
creating large molecules of high relative molecular mass from a lot of small molecules (monomers). each monomer is a repeat unit and is connected to adjacent repeat units via covalent bonds.
addition polymerisation
polymerisaiton of molecules with a C=C bond (not ONLY alkenes, but mainly alkenes.)
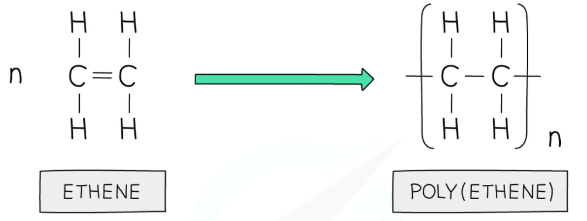
for monomers that’re longer than just one C=C, you need to be careful with how you condense it into the repeating unit:

condensation polymerisation
linking two monomers with different functional groups together by removing a small molecule, usually water. these means per linkage, one water molecule is formed.
polyesters
polyesters are formed by reacting a dicarboxylic acid with a dialcohol. (the di- prefix just means that the functional group is on both ends)

polyamides
polyamides can be formed by reacting a dicarboxylic acid with a diamine (functional group NH2)

polypeptides are specific type of polyamides formed when amino acids undergo condensation polymerisation. they can do this because they have the functional group of both carboxylic acids and of amines:

Naturally occuring polymers
DNA is a polymer essential for life.
it encodes genetic instructions for the development and functioning of organisms
they consist of 4 different monomers called nucleotides. the nucleotide consists of a pentose sugar, a phosphate and a base - A,T, C or G.
the nucleotides bind by polymerisation into strands that then intertwine to produce the double helix shape.
monomer | polymer |
amino acids | protein - condensation polymer |
sugar (glucose) | starch |
sugar (glucose) | cellulose |