Chapter 2 - Periodic Table
The Modern Periodic Table
- The periodic table is in order of increasing atomic number. Each symbol is in a separate box.
- Period - Each row of the periodic table.
- Group - Each column of the periodic table. Elements within groups have similar physical and chemical properties.
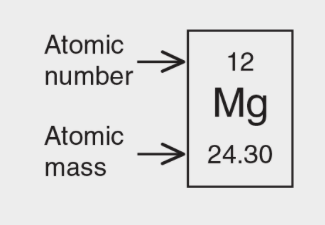
Atomic Symbols
- NOTE - Names and symbols for the elements must be memorized.

- A is the mass of an isotope
- Z is the atomic number
- Charge is the charge of the ion that says if the atom lost or gained electrons
- The subscript is used to indicate how many molecules of that atom are there.
Electrons, Protons, and Neutrons
Protons
- The number of protons is always equal to the atomic number of the element.

Electrons
- The number of electrons is equal to the atomic number.

- For ions, the number of electrons is the atomic number minus the charge of the ion.
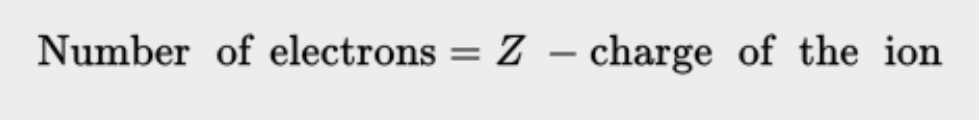
- Cation - Positive ion, lost electrons
- Anion - Negative ion, gained electrons
Neutrons
- The number of neutrons depend on the isotope of the element. If the isotope mass is known, neutrons can be calculated:

- A - isotope mass
- Z - atomic number
- Isotopes - Atoms with the same number of protons but different number of neutrons.
Radioactivity
- Radioactivity - A property of matter where an unstable nucleus spontaneously emits small particles and/or energy to attain a more stable nuclear state.
- Radioactive decay - The process.
- Radioactive isotope (Radioisotope) - An isotope that contains an unstable nucleus.
- Natural radioactive substances - Radioactive isotopes that exist in nature.
- Artificial radioactive isotopes - Isotopes created in a laboratory in nuclear experiments.
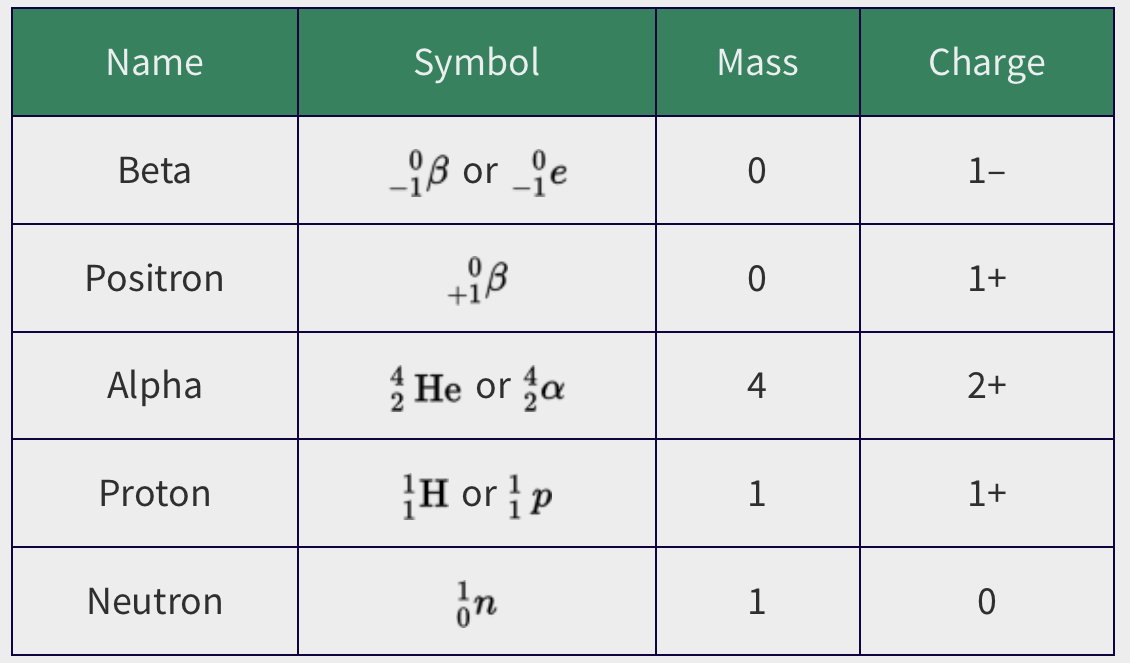
- Radioactive isotopes only emit a few subatomic particles as they disintegrate.
* Electron (beta particle)
* Neutron
* Helium nucleus (alpha particle)
* Positron
* When these are emitted, the nuclear mass and/or nuclear charge of the nucleus changes causing an isotope to turn into another one.
*
Mass Spectrometry
- In a mass spectrometer, tiny amounts of substances are vaporized and exposed to a beam of electrons to create ions.
- Molar mass based on mass spectra - The molar mass is the peak with the largest m/z ratio.
- Base peak - The largest peak used to define 100%.
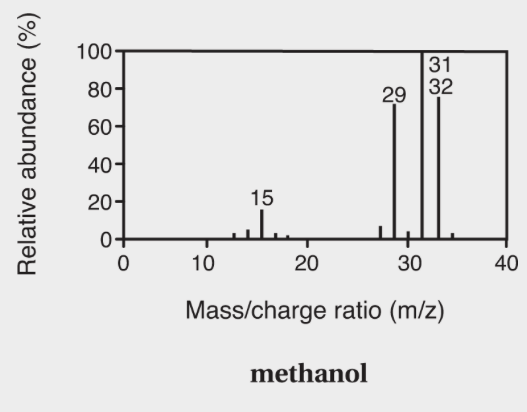
- Methanol’s molar mass is 32.
- The base peak is 31.
Atomic Masses
- The atomic masses on the periodic table is the weighted average of all the element’s isotopes.
- To calculate the weighted average:
* Add the multiplications of the masses of the isotope and their abundances divided by 100.
* ^^Ex. →^^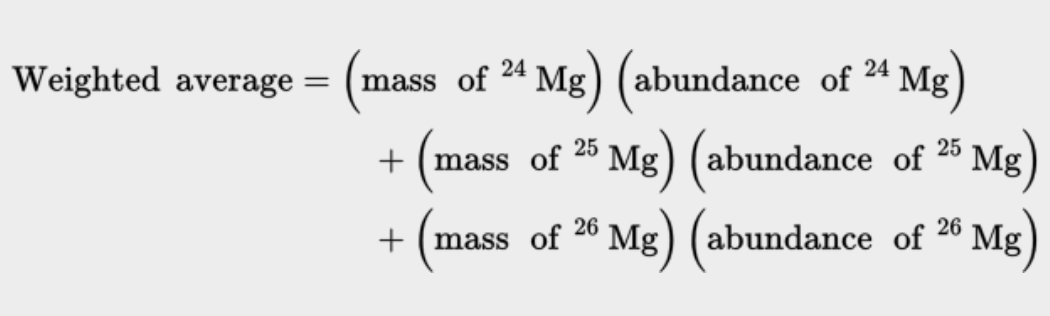
Periodic Properties of the Elements
- Chemical reactions occur when one atom collides with another. The reason why elements with similar electronic structure have similar chemical properties is because in the collisions, the outermost electrons are the ones that make the first contact between atoms.
- Differentiating electron - The electron in a neutral element that makes it different from the previous element.
- Isoelectronic - Atoms and ions that have identical electron configurations.
Physical Properties of the Elements
- Only two elements of the periodic table are liquid under normal conditions:
* Mercury
* Bromine - A few elements exist as a diatomic molecule
* H2
* O2
* N2
* Halogens
Metals and Metalloids
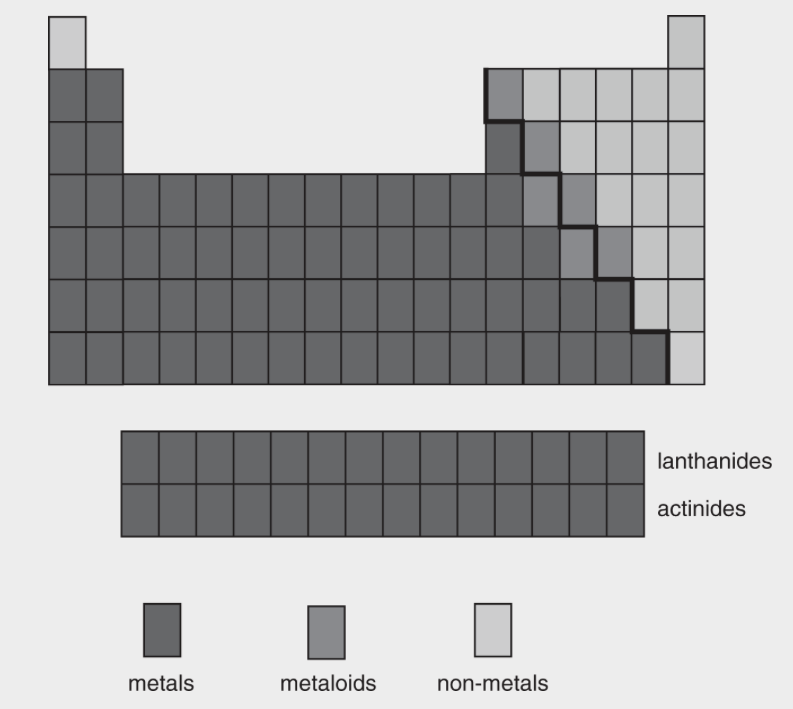
- Metalloids are along the staircase. They have some metal properties and some nonmetal properties.
- The metallic character of the elements increases from the top to the bottom of the periodic table.
Allotropes
- Allotropes - Elements that have two or more distinct sets of chemical and physical properties.
* Ex. → Oxygen (O2) and ozone (O3) are allotropes of oxygen.
* Graphite, diamond, and buckminsterfullerene are allotropes of carbon.
Variation of Physical Properties
- The melting and boiling points of metals tend to decrease from the top to the bottom of a group.
- The melting and boiling points of nonmetals increase from top to bottom.
Atomic Radii
- Atomic radius increases from the top to the bottom of a group because each increase in period number involves a larger energy level.
- When moving from left to right across a period, there’s a decrease in the atomic radius because of an increase in the effective nuclear charge.
Effective Nuclear Charge
- Core electrons (nonvalence electrons) shield the valence electrons from an equal amount of positive nuclear charge.
- Valence electrons don’t shield other valence electrons from the remaining nuclear charge.
* This causes an increase in nuclear charge from left to right in any period.
Ionization Energy
\
\
\
\
\
\
\
\
\
\
\
\
\
\
\
\
\